Abstract
Purpose
The aim of this study was to evaluate the possibility of using a monoclonal antibody against exon 9 deleted E-cadherin (E-cad delta 9-1) for immunotherapy of gastric cancer.
Methods
Among nine human diffuse-type gastric cancer cell lines, we selected a cell line expressing exon 9 deleted E-cadherin (HSC-45M2) by direct sequencing. Tumor specificity and tumor specific in vivo targeting of E-cad delta 9-1 were evaluated in nude mouse bearing a tumor derived from HSC-45M2 cell line by immunohistochemical staining. The expression rate of E-cad delta 9-1 was evaluated in 299 gastric cancer patients, and in positive cases, the mutational status of E-cadherin exon 9 was examined.
Results
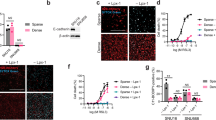
Immunohistochemical staining of various tissues from nude mice showed that only tumor tissue reacted with E-cad delta 9-1. However, immunohistochemical staining of the same tissues after systemic injection of E-cad delta 9-1 showed that reticuloendothelial and hypervascular organs reacted with E-cad delta 9-1, but tumor tissue showed only a slight reaction. Evaluation of the reactivity of 299 gastric cancer patients to E-cad delta 9-1 showed that 4.8% (9/187) of patients, who all had diffuse- or mixed-type gastric cancers, reacted positively, but none of the 112 intestinal-type gastric cancer patients reacted positively. Two of 9 patients (22%) with positive staining to E-cad delta 9-1 were confirmed to have mutant forms of E-cadherin exon 9.
Conclusion
Considering that E-cad delta 9-1 showed good tumor specificity and that some diffuse-type gastric cancers were immunopositive to it, this antibody could be a candidate therapeutic antibody against gastric cancers that express mutant E-cadherin.




Similar content being viewed by others
Abbreviations
- mAb:
-
Monoclonal antibody
- DGC:
-
Diffuse-type gastric cancer
References
Becker KF, Atkinson MJ, Reich U, Becker I, Nekarda H, Siewert JR, Hofler H (1994) E-cadherin gene mutations provide clues to diffuse type gastric carcinomas. Cancer Res 54:3845–3852
Becker KF, Kremmer E, Eulitz M, Becker I, Handschuh G, Schuhmacher C, Muller W, Gabbert HE, Ochiai A, Hirohashi S, Hofler H (1999) Analysis of E-cadherin in diffuse-type gastric cancer using a mutation-specific monoclonal antibody. Am J Pathol 155:1803–1809
Becker KF, Kremmer E, Eulitz M, Schulz S, Mages J, Handschuh G, Wheelock MJ, Cleton-Jansen AM, Hofler H, Becker I (2002) Functional allelic loss detected at the protein level in archival human tumours using allele-specific E-cadherin monoclonal antibodies. J Pathol 197:567–574
Berx G, Becker KF, Hofler H, van Roy F (1998) Mutations of the human E-cadherin (CDH1) gene. Hum Mutat 12:226–237
Carter P (2001) Improving the efficacy of antibody-based cancer therapies. Nat Rev Cancer 1:118–129
Central Cancer Registry in Korea (2002) Annual report of the central cancer registry in Korea. Ministry of health and welfare, Republic of Korea
Christiansen J, Rajasekaran AK (2004) Biological impediments to monoclonal antibody-based cancer immunotherapy. Mol Cancer Ther 3:1493–1501
Fukudome Y, Yanagihara K, Takeichi M, Ito F, Shibamoto S (2000) Characterization of a mutant E-cadherin protein encoded by a mutant gene frequently seen in diffuse-type human gastric carcinoma. Int J Cancer 88:579–583
Gamboa-Dominguez A, Dominguez-Fonseca C, Chavarri-Guerra Y, Vargas R, Reyes-Gutierrez E, Green D, Quintanilla-Martinez L, Luber B, Busch R, Becker KF, Becker I, Hofler H, Fend F (2005) E-cadherin expression in sporadic gastric cancer from Mexico: exon 8 and 9 deletions are infrequent events associated with poor survival. Hum Pathol 36:29–35
Graziano F, Humar B, Guilford P (2003) The role of the E-cadherin gene (CDH1) in diffuse gastric cancer susceptibility: from the laboratory to clinical practice. Ann Oncol 14:1705–1713
Graziano F, Arduini F, Ruzzo A, Bearzi I, Humar B, More H, Silva R, Muretto P, Guilford P, Testa E, Mari D, Magnani M, Cascinu S (2004) Prognostic analysis of E-cadherin gene promoter hypermethylation in patients with surgically resected, node-positive, diffuse gastric cancer. Clin Cancer Res 10:2784–2789
Handschuh G, Candidus S, Luber B, Reich U, Schott C, Oswald S, Becke H, Hutzler P, Birchmeier W, Hofler H, Becker KF (1999) Tumour-associated E-cadherin mutations alter cellular morphology, decrease cellular adhesion and increase cellular motility. Oncogene 18:4301–4312
Hirohashi S (1998) Inactivation of the E-cadherin-mediated cell adhesion system in human cancers. Am J Pathol 153:333–339
Huber R, Seidl C, Schmid E, Seidenschwang S, Becker KF, Schuhmacher C, Apostolidis C, Nikula T, Kremmer E, Schwaiger M, Senekowitsch-Schmidtke R (2003) Locoregional alpha-radioimmunotherapy of intraperitoneal tumor cell dissemination using a tumor-specific monoclonal antibody. Clin Cancer Res 9:3922S-3928S
Iida S, Akiyama Y, Ichikawa W, Yamashita T, Nomizu T, Nihei Z, Sugihara K, Yuasa Y (1999) Infrequent germ-line mutation of the E-cadherin gene in Japanese familial gastric cancer kindreds. Clin Cancer Res 5:1445–1447
Janunger KG, Hafstrom L, Nygren P, Glimelius B (2001) A systematic overview of chemotherapy effects in gastric cancer. Acta Oncol 40:309–326
Kaneko S, Yoshimura T (2001) Time trend analysis of gastric cancer incidence in Japan by histological types, 1975–1989. Br J Cancer 84:400–405
Kim HC, Wheeler JM, Kim JC, Ilyas M, Beck NE, Kim BS, Park KC, Bodmer WF (2000) The E-cadherin gene (CDH1) variants T340A and L599V in gastric and colorectal cancer patients in Korea. Gut 47:262–267
Korean Gastric Cancer Association (2002) Nationwide gastric cancer report in Korea. J Korean Gastric Cancer Assoc 2:105–114
Lee HK, Yang HK, Kim WH, Lee KU, Choe KJ, Kim JP (2001) Influence of the number of lymph nodes examined on staging of gastric cancer. Br J Surg 88:1408–1412
Ludwig DL, Pereira DS, Zhu Z, Hicklin DJ, Bohlen P (2003) Monoclonal antibody therapeutics and apoptosis. Oncogene 22:9097–9106
Muta H, Noguchi M, Kanai Y, Ochiai A, Nawata H, Hirohashi S (1996) E-cadherin gene mutations in signet ring cell carcinoma of the stomach. Jpn J Cancer Res 87:843–848
Park JG, Frucht H, LaRocca RV, Bliss DP Jr, Kurita Y, Chen TR, Henslee JG, Trepel JB, Jensen RT, Johnson BE (1990) Characteristics of cell lines established from human gastric carcinoma. Cancer Res 50:2773–2780
Park JG, Yang HK, Kim WH, Chung JK, Kang MS, Lee JH, Oh JH, Park HS, Yeo KS, Kang SH, Song SY, Kang YK, Bang YJ, Kim YH, Kim JP (1997) Establishment and characterization of human gastric carcinoma cell lines. Int J Cancer 70:443–449
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Senekowitsch-Schmidtke R, Schuhmacher C, Becker KF, Nikula TK, Seidl C, Becker I, Miederer M, Apostolidis C, Adam C, Huber R, Kremmer E, Fischer K, Schwaiger M (2001) Highly specific tumor binding of a 213Bi-labeled monoclonal antibody against mutant E-cadherin suggests its usefulness for locoregional alpha-radioimmunotherapy of diffuse-type gastric cancer. Cancer Res 61:2804–2808
Shimada Y, Yamasaki S, Hashimoto Y, Ito T, Kawamura J, Soma T, Ino Y, Nakanishi Y, Sakamoto M, Hirohashi S, Imamura M (2004) Clinical significance of dysadherin expression in gastric cancer patients. Clin Cancer Res 10:2818–2823
Tamura G, Sakata K, Nishizuka S, Maesawa C, Suzuki Y, Iwaya T, Terashima M, Saito K, Satodate R (1996) Inactivation of the E-cadherin gene in primary gastric carcinomas and gastric carcinoma cell lines. Jpn J Cancer Res 87:1153–1159
Yanagihara K, Ito A, Toge T, Numoto M (1993) Antiproliferative effects of isoflavones on human cancer cell lines established from the gastrointestinal tract. Cancer Res 53:5815–5821
Acknowledgment
This study was supported by grant no. 04-2004-036 from the Seoul National University Hospital Research Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, HJ., Lee, H.S., Hur, K. et al. Tumor specificity and in vivo targeting of an antibody against exon 9 deleted E-cadherin in gastric cancer. J Cancer Res Clin Oncol 133, 987–994 (2007). https://doi.org/10.1007/s00432-007-0246-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-007-0246-5