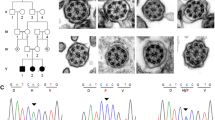
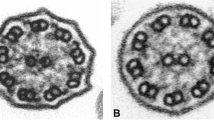
Abstract
Though PCD usually presents after birth in term neonates, diagnosing PCD during the neonatal and infancy stages is uncommon, particularly in children who do not exhibit laterality defects. We report our recent experience with the diagnosis of PCD in the neonatal and early infantile period in a highly consanguine population. This was achieved by implementing a novel genetic-based diagnostic approach based on direct testing for recognized regional genetic variants. We conducted a retrospective analysis of children diagnosed with PCD at Soroka University Medical Center during the neonatal or early infantile period between 2020 and 2023. We included children under 3 months of age who had a genetic confirmation of PCD, as evidenced by the presence of two pathogenic variants in recognized genes. Genetic testing targeted regional genetic variants in previously identified PCD genes. Eight patients were included. The median age at diagnosis was 12.5 days. Three (38%) were born prematurely < 34 weeks gestational age. All patients were presented with respiratory distress and hypoxemia after birth. The median duration of oxygen support was 23 days, and upper lobe atelectasis was present in five patients (63%). Congenital cardiac malformation was present in four patients. Organ laterality defects were present in four patients. Genetic mutations identified were in the DNAAF5, DNAL1, DNAAF3, and DNAH1 genes.
Conclusion: Neonatal diagnosis of PCD is uncommon, especially in atypical presentations such as children without laterality defects or preterms. Focusing on a genetic diagnosis of the local tribal pathogenic variants promotes a potential cost-efficient test leading to earlier diagnosis. There is a need for a standardized protocol for earlier diagnosis of PCD in high-consanguinity areas.
What is Known: • Primary ciliary dyskinesia (PCD) typically presents after birth in term neonates. • Diagnosing PCD during neonatal and infancy stages is challenging, particularly in children without laterality defects. | |
What is New: • A novel genetic-based diagnostic approach was implemented on the neonatal population in a highly consanguine community, focusing on direct testing for regional genetic variants, leading to early and rapid diagnosis of PCD. |

Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Rubbo B, Lucas JS (2017) Clinical care for primary ciliary dyskinesia: current challenges and future directions. Eur Respir Rev 26(145):170023
Ferkol TW, Puffenberger EG, Lie H et al (2013) Primary ciliary dyskinesia-causing mutations in Amish and Mennonite communities. J Pediatr 163:383–387
Casey JP, McGettigan PA, Healy F et al (2015) Unexpected genetic heterogeneity for primary ciliary dyskinesia in the Irish Traveller population. Eur J Hum Genet 23:210–217
Lucas JS, Davis SD, Omran H, Shoemark A (2020) Primary ciliary dyskinesia in the genomics age. Lancet Respir Med 8(2):202–216. https://doi.org/10.1016/S2213-2600(19)30374-1. Epub 2019 Oct 14 PMID: 31624012
Hannah WB, Seifert BA, Truty R, Zariwala MA, Ameel K, Zhao Y, Nykamp K, Gaston B (2022) The global prevalence and ethnic heterogeneity of primary ciliary dyskinesia gene variants: a genetic database analysis. Lancet Respir Med 10(5):459–468
Horani A, Ferkol TW (2021) Understanding primary ciliary dyskinesia and other ciliopathies. J Pediatr 230:15–22.e1. https://doi.org/10.1016/j.jpeds.2020.11.040. Epub 2020 Nov 23. PMID: 33242470; PMCID: PMC8690631
Leigh MW, Ferkol TW, Davis SD et al (2016) Clinical features and associated likelihood of primary ciliary dyskinesia in children and adolescents. Ann Am Thorac Soc 13(8):1305–1313
Kuehni CE, Frischer T, Strippoli MP et al (2010) ERS Task Force on primary ciliary dyskinesia in children. Factors influencing age at diagnosis of primary ciliary dyskinesia in European children. Eur Respir J 36(6):1248–58
Knowles MR, Daniels LA, Davis SD, Zariwala MA, Leigh MW (2013) Primary ciliary dyskinesia. Recent advances in diagnostics, genetics, and characterization of clinical disease. Am J Respir Crit Care Med 188(8):913–22. https://doi.org/10.1164/rccm.201301-0059CI. PMID: 23796196; PMCID: PMC3826280
Coren ME, Meeks M, Morrison I, Buchdahl RM, Bush A (2002) Primary ciliary dyskinesia: age at diagnosis and symptom history. Acta Paediatr 91(6):667–669
Ferkol T, Leigh M (2006) Primary ciliary dyskinesia and newborn respiratory distress. Semin Perinatol 30(6):335–340
Na’amnih W, Romano-Zelekha O, Kabaha A, Rubin LP, Bilenko N, Jaber L, Honovich M, Shohat T (2014) Prevalence of consanguineous marriages and associated factors among Israeli Bedouins. J Community Genet 5(4):395–8. https://doi.org/10.1007/s12687-014-0188-y. Epub 2014 May 10. PMID: 24816554; PMCID: PMC4159474
Gatt D, Golan Tripto I, Levanon E, Arwas N, Hazan G, Alkrinawi S, Goldbart AD, Aviram M (2024) Stepwise genetic approach for the diagnosis of primary ciliary dyskinesia in highly consanguineous populations. Arch Dis Child archdischild-2023–325921. https://doi.org/10.1136/archdischild-2023-325921. Epub ahead of print. PMID: 38296613
Edwards MO, Kotecha SJ, Kotecha S (2013) Respiratory distress of the term newborn infant. Paediatr Respir Rev 14(1):29–36; quiz 36–7. https://doi.org/10.1016/j.prrv.2012.02.002. Epub 2012 Mar 2. PMID: 23347658
Hossain T, Kappelman M, Perez-Atayde A et al (2003) Primary ciliary dyskinesia as a cause of neonatal respiratory distress: implications for the neonatologist. J Perinatol 23:684–687
Goutaki M, Halbeisen FS, Barbato A et al (2020) Late diagnosis of infants with PCD and neonatal respiratory distress. J Clin Med 9:2871
Ellerman A, Bisgaard H (1997) Longitudinal study of lung function in a cohort of primary ciliary dyskinesia. Eur Respir J 10(10):2376–2379
Noone PG, Leigh MW, Sannuti A, Minnix SL, Carson JL, Hazucha M, Zariwala MA, Knowles MR (2004) Primary ciliary dyskinesia: diagnostic and phenotypic features. Am J Respir Crit Care Med 169(4):459–467
Acknowledgements
We extend our appreciation to Dr. Shirly Amar, Ph.D., for her invaluable assistance in obtaining the genetic data for this study.
Author information
Authors and Affiliations
Contributions
I.G, D.G and I.A conceptualized the article and revised the manuscript, N.A wrote the main manuscript text and M.A, R.A., G.H and A.G provided patients data and revised the manuscript. All authors reviewed the manuscript
Corresponding author
Ethics declarations
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Soroka University Medical Center.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Peter de Winter
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arwas, N., Gatt, D., Aviram, M. et al. Neonatal diagnosis of primary ciliary dyskinesia in a high consanguinity population: a single tertiary center experience. Eur J Pediatr (2024). https://doi.org/10.1007/s00431-024-05574-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00431-024-05574-8