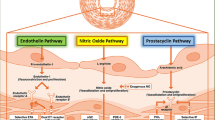
Abstract
Pulmonary arterial hypertension (PAH) is a rare and devastating disease of the pulmonary vasculature with a high morbidity and mortality rate in infants and children. Currently, treatment approaches are mostly based on adult guidelines and pediatrician clinical experience, focusing on specific pulmonary antihypertensive therapy and conventional supportive care. The advent of targeted drugs has led to significant advances in the treatment of PAH in children, including endothelin receptor antagonists, phosphodiesterase type 5 inhibitors, and prostacyclins, which have been studied and proven to improve hemodynamics and functional class in children PAH. A new targeted drug, riociguat, is assessing its safety and efficacy in clinical trials. However, more randomized controlled studies are needed to evaluate the combination of drugs, treatment strategies, and clinical endpoints of targeted therapy in children PAH. In this review, we summarize the research advances of PAH-targeted therapy in children over the last decade in order to provide a theoretical basis for future studies.
Conclusion: Pulmonary arterial hypertension (PAH) is a rare and devastating pulmonary vascular disease that is associated with a variety of diseases of any age in childhood onset.
What is Known: • Therapeutic strategies for targeted drugs for PAH in children are based almost exclusively on data from adult studies and clinical experience of pediatric specialists. • Due to the complex etiology of PAH in children and the relative lack of clinical trial data, the selection of appropriate targeted drug therapy remains difficult. | |
What is New: • We redefine the definition of pulmonary arterial hypertension in children and summarize the progress of targeted therapy of pulmonary arterial hypertension in children in the past ten years. • The dosage and adverse reactions were summarized, and the mechanism of action was drawn according to the available targeted drugs. It can provide theoretical support for the development of guidelines and treatment strategies for the diagnosis and treatment of pulmonary arterial hypertension in children. |

Similar content being viewed by others
References
Steven H. Abman, Mary P. Mullen, Lynn A et al (2021) Characterisation of pediatric pulmonary hypertensive vascular disease from the PPHNet Registry. Eur Respir J 59(1):2003337. https://doi.org/10.1183/13993003.03337-2020. PMID:34140292
van Loon RL, Roofthooft MT, Hillege HL et al (2011) Pediatric pulmonary hypertension in the Netherlands: epidemiology and characterization during the period 1991 to 2005. Circulation 124(16):1755–1764. https://doi.org/10.1161/circulationaha.110.969584 [published Online First: 2011/09/29]
Avitabile CM, Vorhies EE, Ivy DD (2020) Drug treatment of pulmonary hypertension in children. Paediatr Drugs 22(2):123–147. https://doi.org/10.1007/s40272-019-00374-2 [published Online First: 2020/01/22]
Hassoun PM, Mouthon L, Barbera JA et al (2009) Inflammation, growth factors, and pulmonary vascular remodeling. J Am Coll Cardiol 54(1 Suppl):S10–S19. https://doi.org/10.1016/j.jacc.2009.04.006 [published Online First: 2009/07/09]
Cappelleri JC, Hwang LJ, Mardekian J et al (2012) Assessment of measurement properties of peak VO(2) in children with pulmonary arterial hypertension. BMC Pulm Med 12:54. https://doi.org/10.1186/1471-2466-12-54 [published Online First: 2012/09/12]
Sun H, Stockbridge N, Ariagno RL et al (2016) Reliable and developmentally appropriate study end points are needed to achieve drug development for treatment of pediatric pulmonary arterial hypertension. J Perinatol: Off J Cal Perin Assoc 36(12):1029–1033. https://doi.org/10.1038/jp.2016.103 [published Online First: 2016/07/15]
Frank BS, Ivy DD (2018) Diagnosis, evaluation and treatment of pulmonary arterial hypertension in children. Children (Basel, Switzerland) 5(4). https://doi.org/10.3390/children5040044 [published Online First: 2018/03/24]
Simonneau G, Montani D, Celermajer DS et al (2019) Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J 53(1). https://doi.org/10.1183/13993003.01913-2018 [published Online First: 2018/12/14]
Lammers AE, Apitz C (2021) Update from the World Symposium on Pulmonary Hypertension 2018: does the new hemodynamic definition of pediatric pulmonary hypertension have an impact on treatment strategies? Cardiovas Diagn Ther 11(4):1048–1051. https://doi.org/10.21037/cdt-20-412 [published Online First: 2021/09/17]
Rich S, Kaufmann E, Levy PS (1992) The effect of high doses of calcium-channel blockers on survival in primary pulmonary hypertension. N Engl J Med 327(2):76–81. https://doi.org/10.1056/nejm199207093270203 [published Online First: 1992/07/09]
Male C, Thom K, O’Brien SH (2019) Direct oral anticoagulants: what will be their role in children? Thromb Res 173:178–185. https://doi.org/10.1016/j.thromres.2018.06.021 [published Online First: 2018/07/19]
Gorenflo M, Ziesenitz VC (2021) Treatment of pulmonary arterial hypertension in children. Cardiovas Diagn Ther 11(4):1144–1159. https://doi.org/10.21037/cdt-20-912 [published Online First: 2021/09/17]
Haworth SG, Hislop AA (2009) Treatment and survival in children with pulmonary arterial hypertension: the UK Pulmonary Hypertension Service for Children 2001–2006. Heart 95(4):312–317. https://doi.org/10.1136/hrt.2008.150086 [published Online First: 2008/10/28]
Jeremiasen I, Naumburg E, Westöö C et al (2021) Vasodilator therapy for pulmonary hypertension in children: a national study of patient characteristics and current treatment strategies. Pulm Circ 11(4):20458940211057892. https://doi.org/10.1177/20458940211057891 [published Online First: 2021/12/21]
Barnes H, Brown Z, Burns A et al (2019) Phosphodiesterase 5 inhibitors for pulmonary hypertension. Cochrane Database System Rev 1(1):Cd012621. https://doi.org/10.1002/14651858.CD012621.pub2 [published Online First: 2019/02/01]
Simonca L, Tulloh R (2017) Sildenafil in Infants and Children. Children (Basel, Switzerland) 4(7). https://doi.org/10.3390/children4070060 [published Online First: 2017/07/25]
Sun X, Wang K, Wang W et al (2014) [Clinical study on sildenafil in treatment of pregnant women with pulmonary arterial hypertension]. Zhonghua fu chan ke za zhi 49(6):414–8. [published Online First: 2014/08/30]
Xia YL, Yan WX, Chen H (2014) [Efficacy and safety of sildenafil in the treatment of high altitude heart disease associated with severe pulmonary arterial hypertension in children: a preliminary evaluation]. Zhongguo dang dai er ke za zhi = Chinese J Contemp Pedia 16(7):745–8. [published Online First: 2014/07/11]
Beghetti M, Gorenflo M, Ivy DD et al (2019) Treatment of pediatric pulmonary arterial hypertension: a focus on the NO-sGC-cGMP pathway. Pediatr Pulmonol 54(10):1516–1526. https://doi.org/10.1002/ppul.24442 [published Online First: 2019/07/18]
Barst RJ, Ivy DD, Gaitan G et al (2012) A randomized, double-blind, placebo-controlled, dose-ranging study of oral sildenafil citrate in treatment-naive children with pulmonary arterial hypertension. Circulation 125(2):324–334. https://doi.org/10.1161/CIRCULATIONAHA.110.016667 [published Online First: 2011/12/01]
Barst RJ, Beghetti M, Pulido T et al (2014) STARTS-2: long-term survival with oral sildenafil monotherapy in treatment-naive pediatric pulmonary arterial hypertension. Circulation 129(19):1914–1923. https://doi.org/10.1161/circulationaha.113.005698 [published Online First: 2014/03/19]
Sable CA, Ivy DD, Beekman RH, 3rd et al (2017) ACC/AAP/AHA health policy statement on opportunities and challenges in pediatric drug development: learning from sildenafil. Circul Cardiovas Qual Outcomes 10(7). https://doi.org/10.1161/hcq.0000000000000026 [published Online First: 2017/07/01]
Dodgen AL, Hill KD (2015) Safety and tolerability considerations in the use of sildenafil for children with pulmonary arterial hypertension. Drug Healthcare Patient Safety 7:175–183. https://doi.org/10.2147/dhps.S65571 [published Online First: 2016/01/01]
Beghetti M, Rudzinski A, Zhang M (2017) Efficacy and safety of oral sildenafil in children with Down syndrome and pulmonary hypertension. BMC Cardiovasc Disord 17(1):177. https://doi.org/10.1186/s12872-017-0569-3 [published Online First: 2017/07/06]
Issapour A, Frank B, Crook S et al (2022) Safety and tolerability of combination therapy with ambrisentan and tadalafil for the treatment of pulmonary arterial hypertension in children: real-world experience. Pediatr Pulmonol 57(3):724–733. https://doi.org/10.1002/ppul.25796 [published Online First: 2021/12/19]
Sabri MR, Bigdelian H, Hosseinzadeh M et al (2017) Comparison of the therapeutic effects and side effects of tadalafil and sildenafil after surgery in young infants with pulmonary arterial hypertension due to systemic-to-pulmonary shunts. Cardiol Young 27(9):1686–1693. https://doi.org/10.1017/s1047951117000981 [published Online First: 2017/10/11]
Magee AG, Makhecha S, Bentley S (2015) Risk-benefit considerations when prescribing phosphodiesterase-5 inhibitors in children. Expert Opin Drug Saf 14(5):633–642. https://doi.org/10.1517/14740338.2015.1022527 [published Online First: 2015/03/10]
Belge C, Delcroix M (2019) Treatment of pulmonary arterial hypertension with the dual endothelin receptor antagonist macitentan: clinical evidence and experience. Ther Adv Respir Dis 13:1753466618823440. https://doi.org/10.1177/1753466618823440 [published Online First: 2019/02/10]
Ivy D, Frank BS (2021) Update on pediatric pulmonary arterial hypertension. Curr Opin Cardiol 36(1):67–79. https://doi.org/10.1097/hco.0000000000000822 [published Online First: 2020/11/03]
Berger RM, Haworth SG, Bonnet D et al (2016) FUTURE-2: results from an open-label, long-term safety and tolerability extension study using the pediatric FormUlation of bosenTan in pUlmonary arterial hypeRtEnsion. Int J Cardiol 202:52–58. https://doi.org/10.1016/j.ijcard.2015.08.080 [published Online First: 2015/09/21]
Berger RMF, Gehin M, Beghetti M et al (2017) A bosentan pharmacokinetic study to investigate dosing regimens in paediatric patients with pulmonary arterial hypertension: FUTURE-3. Br J Clin Pharmacol 83(8):1734–1744. https://doi.org/10.1111/bcp.13267 [published Online First: 2017/02/19]
Beghetti M, Haworth SG, Bonnet D et al (2009) Pharmacokinetic and clinical profile of a novel formulation of bosentan in children with pulmonary arterial hypertension: the FUTURE-1 study. Br J Clin Pharmacol 68(6):948–955. https://doi.org/10.1111/j.1365-2125.2009.03532.x [published Online First: 2009/12/17]
Galie N, Olschewski H, Oudiz RJ et al (2008) Ambrisentan for the treatment of pulmonary arterial hypertension: results of the ambrisentan in pulmonary arterial hypertension, randomized, double-blind, placebo-controlled, multicenter, efficacy (ARIES) study 1 and 2. Circulation 117(23):3010–3019. https://doi.org/10.1161/CIRCULATIONAHA.107.742510 [published Online First: 2008/05/29]
Hill KD, Maharaj AR, Li JS et al (2020) A Randomized, controlled pharmacokinetic and pharmacodynamics trial of ambrisentan after fontan surgery. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 21(9):e795–e803. https://doi.org/10.1097/pcc.0000000000002410 [published Online First: 2020/07/09]
Preston IR, Burger CD, Bartolome S et al (2020) Ambrisentan in portopulmonary hypertension: a multicenter, open-label trial. J Heart Lung Transplant 39(5):464–472. https://doi.org/10.1016/j.healun.2019.12.008 [published Online First: 2020/02/06]
Martynyuk TV, Nakonechnikov SN, Chazova IY (2018) New horizons for the use of the second generation of endothelin receptor antagonist macitentan in patients with pulmonary hypertension. Ter Arkh 90(4):72–80. https://doi.org/10.26442/terarkh201890472-80 [published Online First: 2019/02/01]
Pulido T, Adzerikho I, Channick RN et al (2013) Macitentan and morbidity and mortality in pulmonary arterial hypertension. N Engl J Med 369(9):809–818. https://doi.org/10.1056/NEJMoa1213917 [published Online First: 2013/08/30]
Wu Y, Liu HM, Gu L et al (2022) Prostacyclins and pulmonary arterial hypertension in children. Eur Rev Med Pharmacol Sci 26(1):37–45. https://doi.org/10.26355/eurrev_202201_27745 [published Online First: 2022/01/21]
Douwes JM, Zijlstra WMH, Rosenzweig EB et al (2022) Parenteral prostanoids in pediatric pulmonary arterial hypertension: start early, dose high, combine. Ann Am Thorac Soc 19(2):227–237. https://doi.org/10.1513/AnnalsATS.202012-1563OC [published Online First: 2021/06/29]
Mulligan C, Beghetti M (2012) Inhaled iloprost for the control of acute pulmonary hypertension in children: a systematic review. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 13(4):472–480. https://doi.org/10.1097/PCC.0b013e31822f192b [published Online First: 2011/09/20]
Gavotto A, Thomas F, Werner O et al (2020) Use of treprostinil in pediatric pulmonary hypertension: case reports and review of the literature. J Cardiovasc Pharmacol 76(1):23–31. https://doi.org/10.1097/fjc.0000000000000820 [published Online First: 2020/03/14]
Hopper RK, Wang Y, DeMatteo V et al (2018) Right ventricular function mirrors clinical improvement with use of prostacyclin analogues in pediatric pulmonary hypertension. Pulm Circ 8(2):2045894018759247. https://doi.org/10.1177/2045894018759247 [published Online First: 2018/02/27]
McIntyre CM, Hanna BD, Rintoul N et al (2013) Safety of epoprostenol and treprostinil in children less than 12 months of age. Pulm Circ 3(4):862–869. https://doi.org/10.1086/674762 [published Online First: 2014/07/10]
Hopper RK, Ivy DD, Yung D et al (2020) Pharmacokinetics of oral treprostinil in children with pulmonary arterial hypertension. J Cardiovasc Pharmacol 76(1):94–100. https://doi.org/10.1097/fjc.0000000000000842 [published Online First: 2020/05/14]
Krishnan U, Takatsuki S, Ivy DD et al (2012) Effectiveness and safety of inhaled treprostinil for the treatment of pulmonary arterial hypertension in children. Am J Cardiol 110(11):1704–1709. https://doi.org/10.1016/j.amjcard.2012.07.037 [published Online First: 2012/08/25]
Ablonczy L, Tordas D, Kis E et al (2018) Use of subcutaneous treprostinil in pediatric pulmonary arterial hypertension-Bridge-to-transplant or long-term treatment? Pediatr Transplant 22(2). https://doi.org/10.1111/petr.13106 [published Online First: 2017/12/22]
Levy M, Celermajer DS, Bourges-Petit E et al (2011) Add-on therapy with subcutaneous treprostinil for refractory pediatric pulmonary hypertension. J Pediatr 158(4):584–588. https://doi.org/10.1016/j.jpeds.2010.09.025
Levy M, Del Cerro MJ, Nadaud S et al (2018) Safety, efficacy and management of subcutaneous treprostinil infusions in the treatment of severe pediatric pulmonary hypertension. Int J Cardiol 264:153–157. https://doi.org/10.1016/j.ijcard.2018.03.067 [published Online First: 2018/04/14]
Jiang H, Yu X, Zhang L et al (2021) Effects of treprostinil on pulmonary arterial hypertension during surgery for congenital heart disease complicated with severe pulmonary arterial hypertension. Minerva Cardio Angiol 69(2):154–160. https://doi.org/10.23736/s2724-5683.20.05085-9 [published Online First: 2020/03/07]
Barnes H, Yeoh HL, Fothergill T et al (2019) Prostacyclin for pulmonary arterial hypertension. Cochrane Database Syst Rev 5(5):Cd012785. https://doi.org/10.1002/14651858.CD012785.pub2 [published Online First: 2019/05/02]
Takatsuki S, Nakayama T, Shimizu Y et al (2022) Clinical efficacy and safety of selexipag in children and young adults with idiopathic and heritable pulmonary arterial hypertension. Cardiol Young 1–5. https://doi.org/10.1017/s1047951122000415 [published Online First: 2022/04/07]
Boehler M, Bruderer S, Ulč I et al (2018) Biocomparison study of adult and paediatric dose strengths of the prostacyclin receptor agonist selexipag. Eur J Drug Metab Pharmacokinet 43(1):115–120. https://doi.org/10.1007/s13318-017-0424-z [published Online First: 2017/06/24]
Sandner P, Becker-Pelster EM, Stasch JP (2018) Discovery and development of sGC stimulators for the treatment of pulmonary hypertension and rare diseases. Nitric Oxide 77:88–95. https://doi.org/10.1016/j.niox.2018.05.001 [published Online First: 2018/05/09]
Ghofrani HA, Galie N, Grimminger F et al (2013) Riociguat for the treatment of pulmonary arterial hypertension. N Engl J Med 369(4):330–340. https://doi.org/10.1056/NEJMoa1209655 [published Online First: 2013/07/26]
Ghofrani HA, D’Armini AM, Grimminger F et al (2013) Riociguat for the treatment of chronic thromboembolic pulmonary hypertension. N Engl J Med 369(4):319–329. https://doi.org/10.1056/NEJMoa1209657 [published Online First: 2013/07/26]
Wang C, Jing ZC, Huang YG et al (2016) Riociguat for the treatment of pulmonary hypertension: Chinese subgroup analyses and comparison. Heart Asia 8(1):74–82. https://doi.org/10.1136/heartasia-2015-010712 [published Online First: 2016/06/22]
Spreemann T, Bertram H, Happel CM et al (2018) First-in-child use of the oral soluble guanylate cyclase stimulator riociguat in pulmonary arterial hypertension. Pulm Circ 8(1):2045893217743123. https://doi.org/10.1177/2045893217743123 [published Online First: 2017/11/04]
Takatsuki S, Ivy DD (2013) Current challenges in pediatric pulmonary hypertension. Sem Resp Crit Care Med 34(5):627–644. https://doi.org/10.1055/s-0033-1356461 [published Online First: 2013/09/17]
Barst RJ, McGoon MD, Elliott CG et al (2012) Survival in childhood pulmonary arterial hypertension: insights from the registry to evaluate early and long-term pulmonary arterial hypertension disease management. Circulation 125(1):113–122. https://doi.org/10.1161/CIRCULATIONAHA.111.026591 [published Online First: 2011/11/17]
Melnick L, Barst RJ, Rowan CA et al (2010) Effectiveness of transition from intravenous epoprostenol to oral/inhaled targeted pulmonary arterial hypertension therapy in pediatric idiopathic and familial pulmonary arterial hypertension. Am J Cardiol 105(10):1485–1489. https://doi.org/10.1016/j.amjcard.2009.12.075 [published Online First: 2010/05/11]
Haarman MG, Levy M, Roofthooft MTR et al (2021) Upfront triple combination therapy in severe paediatric pulmonary arterial hypertension. Eur Respir J 57(1). https://doi.org/10.1183/13993003.01120-2020 [published Online First: 2020/08/29]
Li Li, Xinyu Zhu, Xiaojie Chen et al (2022) Advances in targeted therapy for pulmonary arterial hypertension in children. Authorea July 05, 2022
Funding
This work was supported by the Research Projects of Gansu Provincial Hospital (20GSSY5-2, 21GSSYB-8), the Gansu Provincial Youth Science and Technology Fund (20JR10RA415), the Natural Science Foundation of Gansu Province (21JR11RA186), and the Guiding Plan for Scientific and Technological Development of Lanzhou (2019-ZD-102).
Author information
Authors and Affiliations
Contributions
Shixun Ma and Min Zhang: study design, methodological advice, drafting, and revising the manuscript. Li Li: study design, collecting data, drafting, and revising the manuscript. Xinyu Zhu and Xiaojie Chen: collecting data, data entry, data cleaning and drafting the manuscript. Jieyun Gao and Chunchun Ding: interpreting of data and drafting and revising the manuscript. The author(s) read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Peter de Winter
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A preprint has previously been published [Li Li, Xinyu Zhu, Xiaojie Chen, et.al. July 05, 2022].
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, L., Zhu, X., Chen, X. et al. Advances in targeted therapy for pulmonary arterial hypertension in children. Eur J Pediatr 182, 2067–2076 (2023). https://doi.org/10.1007/s00431-022-04750-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-022-04750-y