Abstract
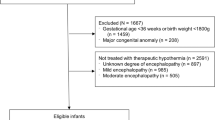
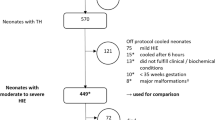
Initiation of therapeutic hypothermia (TH) within 6 h of life is a major concern for treating neonatal hypoxic ischemic encephalopathy (HIE). We aimed to determine clinical and healthcare organizational factors associated with delayed TH in a French population-based cohort of neonates with moderate/severe HIE. Time to reach a rectal temperature of 34 °C defines optimal and delayed (within and over 6 h, respectively) TH. Clinical and healthcare organizational factors associated with delayed TH were analysed among neonates born in cooling centres (CCs) and non-cooling centres (non-CCs). Among 629 neonates eligible for TH, 574 received treatment (91.3%). TH was delayed in 29.8% neonates and in 20.3% and 36.2% of those born in CCs and non-CCs, respectively. Neonates with moderate HIE were more exposed to delayed TH in both CCs and non-CCs. After adjustment for HIE severity, maternal and neonatal characteristics and circumstances of birth were not associated with increased risk of delayed TH. However, this risk was 2 to 5 times higher in maternities with < 1999 annual births, when the delay between birth and call for transfer (adjusted odds ratio [aOR] 2.47, 95% confidence interval [CI] [1.03 to 5.96]) or between call for transfer and admission (aOR 6.06, 95%CI [2.60 to 14.12]) was > 3 h and when an undesirable event occurred during transfer (aOR 2.66, 95%CI [1.11 to 6.37].
Conclusion: Increasing early identification of neonates who could benefit from TH and access to TH in non-CCs before transfer are modifiable factors that could improve care of neonates with HIE.
Trial registration: The trial was registered at ClinicalTrials.gov (NCT02676063).
What is Known: • International recommendations are to initiate therapeutic hypothermia before 6 h of life in neonates with moderate or severe hypoxic ischemic encephalopathy. | |
What is New: •In this French population-based cohort of infants with hypoxic ischemic encephalopathy, nearly one-third of neonates eligible for treatment did not have access to hypothermia in the therapeutic window of 6 h of life. . • Among infants born in non-cooling centres, healthcare organizational factors involved in delayed care were the small size of maternities (1999 annual births), a time interval of more than 3 h between birth and call for transfer and between call for transfer and admission in neonatology, and the occurrence of an undesirable event during transfer. |

Similar content being viewed by others
Abbreviations
- HIE:
-
Hypoxic ischemic encephalopathy
- TH:
-
Therapeutic hypothermia
- CC:
-
Cooling centre
- Non-CC:
-
Non-cooling centre
- WG:
-
Weeks’ gestation
- aOR:
-
Adjusted odds ratio
- CI:
-
Confidence interval
References
Ferlie EB, Shortell SM (2001) Improving the quality of health care in the United Kingdom and the United States: a framework for change. Milbank Q 79:281–315. https://doi.org/10.1111/1468-0009.00206
Diguisto C, Goffinet F, Lorthe E et al (2017) Providing active antenatal care depends on the place of birth for extremely preterm births: the EPIPAGE 2 cohort study. Arch Dis Child Fetal Neonatal Ed 102:F476–F482. https://doi.org/10.1136/archdischild-2016-312322
Perlbarg J, Ancel PY, Khoshnood B et al (2016) Delivery room management of extremely preterm infants: the EPIPAGE-2 study. Arch Dis Child Fetal Neonatal Ed. https://doi.org/10.1136/archdischild-2015-308728
Goodman DC, Fisher ES, Little GA et al (2002) The relation between the availability of neonatal intensive care and neonatal mortality. N Engl J Med 346:1538–1544. https://doi.org/10.1056/NEJMoa011921
Gupta N, Shipley L, Goel N et al (2019) Neurocritical care of high-risk infants during inter-hospital transport. Acta Paediatr Oslo Nor 108:1965–1971. https://doi.org/10.1111/apa.14940
Jacobs SE, Berg M, Hunt R et al (2013) Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev CD003311. https://doi.org/10.1002/14651858.CD003311.pub3
Saliba E, Debillon T (2010) Hypothermia for hypoxic-ischemic encephalopathy in fullterm newborns. Arch Pediatr Organe Off Soc Francaise Pediatr 17(Suppl 3):S67-77. https://doi.org/10.1016/S0929-693X(10)70904-0
Drury PP, Gunn ER, Bennet L, Gunn AJ (2014) Mechanisms of hypothermic neuroprotection. Clin Perinatol 41:161–175. https://doi.org/10.1016/j.clp.2013.10.005
Committee on Fetus and Newborn, Papile L-A, Baley JE et al (2014) Hypothermia and neonatal encephalopathy. Pediatrics 133:1146–1150. https://doi.org/10.1542/peds.2014-0899
Lemyre B, Chau V (2018) Hypothermia for newborns with hypoxic-ischemic encephalopathy. Paediatr Child Health 23:285–291. https://doi.org/10.1093/pch/pxy028
Wassink G, Davidson JO, Dhillon SK et al (2019) Therapeutic Hypothermia in Neonatal Hypoxic-Ischemic Encephalopathy. Curr Neurol Neurosci Rep 19:2. https://doi.org/10.1007/s11910-019-0916-0
Mistry A, Shipley L, Ojha S et al (2022) Availability of active therapeutic hypothermia at birth for neonatal hypoxic ischaemic encephalopathy: a UK population study from 2011 to 2018. Arch Dis Child Fetal Neonatal Ed fetalneonatal-2021-322906. https://doi.org/10.1136/archdischild-2021-322906
Chevallier M, Ego A, Cans C et al (2013) Adherence to hypothermia guidelines: a French multicenter study of fullterm neonates. PLoS ONE 8:e83742. https://doi.org/10.1371/journal.pone.0083742
Debillon T, Bednarek N, Ego A et al (2018) LyTONEPAL: long term outcome of neonatal hypoxic encephalopathy in the era of neuroprotective treatment with hypothermia: a French population-based cohort. BMC Pediatr 18:255. https://doi.org/10.1186/s12887-018-1232-6
Mamelle N, Munoz F, Grandjean H (1996) Fetal growth from the AUDIPOG study. I. Establishment of reference curves. J Gynecol Obstet Biol Reprod (Paris) 25:61–70
Groenendaal F, Casaer A, Dijkman KP et al (2013) Introduction of hypothermia for neonates with perinatal asphyxia in the Netherlands and Flanders. Neonatology 104:15–21. https://doi.org/10.1159/000348823
Shipley L, Mistry A, Sharkey D (2022) Outcomes of neonatal hypoxic-ischaemic encephalopathy in centres with and without active therapeutic hypothermia: a nationwide propensity score-matched analysis. Arch Dis Child Fetal Neonatal Ed 107:6–12. https://doi.org/10.1136/archdischild-2020-320966
Shipley L, Gale C, Sharkey D (2021) Trends in the incidence and management of hypoxic-ischaemic encephalopathy in the therapeutic hypothermia era: a national population study. Arch Dis Child Fetal Neonatal Ed 106:529–534. https://doi.org/10.1136/archdischild-2020-320902
Parmentier CEJ, De Vries LS, Toet MC et al (2020) Increased use of therapeutic hypothermia in infants with milder neonatal encephalopathy due to presumed perinatal asphyxia. Neonatology 117:488–494. https://doi.org/10.1159/000508710
Hage L, Jeyakumaran D, Dorling J et al (2021) Changing clinical characteristics of infants treated for hypoxic-ischaemic encephalopathy in England, Wales and Scotland: a population-based study using the national neonatal research database. Arch Dis Child Fetal Neonatal Ed 106:501–508. https://doi.org/10.1136/archdischild-2020-319685
Mathew JL, Kaur N, Dsouza JM (2022) Therapeutic hypothermia in neonatal hypoxic encephalopathy: A systematic review and meta-analysis. J Glob Health 12:04030. https://doi.org/10.7189/jogh.12.04030
Montaldo P, Lally PJ, Oliveira V et al (2019) Therapeutic hypothermia initiated within 6 hours of birth is associated with reduced brain injury on MR biomarkers in mild hypoxic-ischaemic encephalopathy: a non-randomised cohort study. Arch Dis Child Fetal Neonatal Ed 104:F515–F520. https://doi.org/10.1136/archdischild-2018-316040
Kariholu U, Montaldo P, Markati T et al (2020) Therapeutic hypothermia for mild neonatal encephalopathy: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed 105:225–228. https://doi.org/10.1136/archdischild-2018-315711
El-Dib M, Inder TE, Chalak LF et al (2019) Should therapeutic hypothermia be offered to babies with mild neonatal encephalopathy in the first 6 h after birth? Pediatr Res 85:442–448. https://doi.org/10.1038/s41390-019-0291-1
Laptook AR, Shankaran S, Tyson JE et al (2017) Effect of therapeutic hypothermia initiated after 6 hours of age on death or disability among newborns with hypoxic-ischemic encephalopathy: a randomized clinical trial. JAMA 318:1550–1560. https://doi.org/10.1001/jama.2017.14972
Oliveira V, Singhvi DP, Montaldo P et al (2018) Therapeutic hypothermia in mild neonatal encephalopathy: a national survey of practice in the UK. Arch Dis Child Fetal Neonatal Ed 103:F388–F390. https://doi.org/10.1136/archdischild-2017-313320
McDouall A, Wassink G, Bennet L et al (2021) Challenges in developing therapeutic strategies for mild neonatal encephalopathy. Neural Regen Res 17:277–282. https://doi.org/10.4103/1673-5374.317963
Boylan GB, Kharoshankaya L, Wusthoff CJ (2015) Seizures and hypothermia: importance of electroencephalographic monitoring and considerations for treatment. Semin Fetal Neonatal Med 20:103–108. https://doi.org/10.1016/j.siny.2015.01.001
Acknowledgements
We thank members of the LyTONEPAL Study Group, all the regional teams participating in the study, and all French maternity and neonatal units for their substantial contribution to acquisition of data; and Laura Smales for her helpful contribution for proofreading. We are grateful for the participation of all families of infants in the LyTONEPAL cohort study.
Funding
National Program for Clinical Research (PHRC-N-13–0327).
J.B has received support from The American Memorial Committee.
Author information
Authors and Affiliations
Contributions
JB, AV, TD, AE and PYA had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. JB, TD, VP, AE and PYA conceptualized the study and wrote the manuscript. JB and AV performed the statistical analysis. TD obtained funding and supervised the study. IG, GL and NB contributed to interpretation of the results and reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Informed consent was obtained from both parents. The study protocol was approved by the Advisory National Committee on the treatment of personal health data for research purposes (Comité Consultatif sur le Traitement de l’Information en matière de Recherche sur la Santé, reference no. 14.724). Authorizations were obtained from the National French data protection authority (Commission Nationale Informatique et Libertés, DR-2015–136) and the Regional Ethics committee CPP (Comité de Protection des Personnes Sud Est; Institutional Review Board no. 5891).
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Daniele De Luca
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Beck, J., Debillon, T., Guellec, I. et al. Healthcare organizational factors associated with delayed therapeutic hypothermia in neonatal hypoxic-ischemic encephalopathy: the LyTONEPAL cohort. Eur J Pediatr 182, 181–190 (2023). https://doi.org/10.1007/s00431-022-04666-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-022-04666-7