Abstract
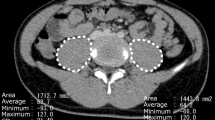
The clinical characteristics, cause, and risk factors of sarcopenia are unclear in children. The aim of this study was to describe the course of and identify the factors related to muscle mass change during chemotherapy in children with neuroblastoma. A total of 24 consecutive patients aged below 18 years with newly diagnosed high-risk neuroblastoma between 2010 and 2018 in our hospital were enrolled in a case-series study. The psoas muscle index (PMI) was calculated as a parameter of muscle mass based on computer tomography (CT) images of the psoas muscle. PMIs were evaluated at 4 time points (TPs): TP1, at the diagnosis of neuroblastoma; TP2, after the first cycle of chemotherapy; TP3, after the third cycle of chemotherapy; and TP4, at the end of the induction chemotherapy. PMI recovery was defined as an increase in PMI between TP2 and TP4. The mean PMI decreased by 15% between TP1 and TP2 (TP1 7.09 ± 0.99 vs. TP2 6.01 ± 0.98, P < 0.001) and by 10% between TP1 and TP4 (TP1 7.09 vs. TP4 6.35, P = 0.004). PMI recovery between TP1 and TP2 was observed in 7 (29%) patients. The median age of patients with PMI recovery was significantly lower (2 vs. 4 years, P = 0.028), and the proportion of boys was significantly higher in patients with PMI recovery (100% vs. 41%, P = 0.017).
Conclusion: This study demonstrated that prominent PMI reduction occurs during the early time of chemotherapy, and a younger age and male sex may be predictive factors for PMI recovery.
What is Known: • Sarcopenia is a common disorder in elderly people. • Several causes and risk factors have been reported in adults. • Children with previous hematological malignancies have decreased physical activity. What is New: • Prominent muscle mass loss was observed early in children with high-risk neuroblastoma during chemotherapy. • Age and sex were found to be potentially associated with muscle mass recovery. |


Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
N/A.
Abbreviations
- ALL:
-
Acute lymphoblastic leukemia
- BMI:
-
Body mass index
- BSA:
-
Body surface area
- CT:
-
Computed tomography
- ECOG:
-
Eastern Cooperative Oncology Group
- INPC:
-
International Neuroblastoma Pathology Classification
- INSS:
-
International Neuroblastoma Staging System
- MKI:
-
Mitosis-karyorrhexis index
- PS:
-
Performance status
- PMCSA:
-
Psoas muscle cross-sectional area
- QOL:
-
Quality of life
- TP:
-
Time point
References
San Juan F, Chamorro-Viña C, Maté-Muñoz L, Fernández del Valle M, Cardona C, Hernández M et al (2007) Functional capacity of children with leukemia. Int J Sports Med 28:1–5
Van Brussel M, Takken T, Lucia A, van der Net J, Helders PJ (2005) Is physical fitness decreased in survivors of childhood leukemia? A systematic review. Leukemia. 19:13–17
Bianco A, Patti A, Thomas E, Palma R, Cristina Maggio M, Paoli A et al (2014) Evaluation of fitness levels of children with a diagnosis of acute leukemia and lymphoma after completion of chemotherapy and autologous hematopoietic stem cell transplantation. Cancer Med 3:385–389
Stene GB, Helbostad JL, Balstad TR, Riphagen II, Kaasa S, Oldervoll LM (2013) Effect of physical exercise on muscle mass and strength in cancer patients during treatment--a systematic review. Crit Rev Oncol Hematol 88:573–593
Sung L, Zaoutis T, Ullrich NJ, Johnston D, Dupuis L, Ladas E, Children's Oncology Group Cancer Control and Supportive Care Committee (2013) Children's Oncology Group's 2013 blueprint for research: cancer control and supportive care. Pediatr Blood Cancer 60:1027–1030
Braam KI, van Dijk-Lokkart EM, Kaspers GJL, Takken T, Huisman J, Buffart LM, Bierings MB, Merks JHM, van den Heuvel-Eibrink MM, Veening MA, van Dulmen-den Broeder E (2018) Effects of a combined physical and psychosocial training for children with cancer: a randomized controlled trial. BMC Cancer 18:1289
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M, European Working Group on Sarcopenia in Older People (2010) Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 39:412–423
Suzuki D, Kobayashi R, Sano H, Hori D, Kobayashi K (2018) Sarcopenia after induction therapy in childhood acute lymphoblastic leukemia: its clinical significance. Int J Hematol 107:486–489
Cruz-Jentoft AJ, Francesco L, Stéphane MS, Zúñiga C, Arai H, Boirie Y et al (2014) Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 43:748–759
Bischoff-Ferrari HA, Orav JE, Kanis JA, Rizzoli R, Schlögl M, Staehelin HB, Willett WC, Dawson-Hughes B (2015) Comparative performance of current definitions of sarcopenia against the prospective incidence of falls among community-dwelling seniors age 65 and older. Osteoporos Int 26:2793–2802
Beaudart C, Rizzoli R, Bruyere O, Reginster JY, Biver E (2014) Sarcopenia: Burden and challenges for Public Health. Arch Public Health 72:45
Rizzoli R, Jean-Yves R, Jean-François A, Bautmans I, Beaudart C, Bischoff-Ferrari H et al (2013) Quality of life in sarcopenia and frailty. Calcif Tissue Int 93:101–120
Lauretani F, Cosimo RR, Stefania B, Bartali B, Cavazzini C, Iorio AD et al (2003) Age-associated changes in skeletal muscles and their effect on mobility: an operational diagnosis of sarcopenia. J Appl Physiol 95:1851–1860
Visser M, Schaap LA (2011) Consequences of sarcopenia. Clin Geriatr Med 27:387–399
Lang T, Streeper T, Cawthon P, Baldwin K, Taaffe DR, Harris TB (2010) Sarcopenia: etiology, clinical consequences, intervention, and assessment. Osteoporos Int 21:543–559
Francesco L, Rosa L, Andrea R, Giovannini S, Tosato M, Capoluongo E et al (2012) Sarcopenia as a risk factor for falls in elderly individuals: results from the ilSIRENTE study. Clin Nutr 31:652–658
Kang YJ, Yoo JI, Ha YC (2019) Sarcopenia feature selection and risk prediction using machine learning: a cross-sectional study. Medicine (Baltimore) 98:e17699
Kamijo Y, Kanda E, Ishibashi Y, Yoshida M (2018) Sarcopenia and frailty in PD: impact on mortality, malnutrition, and inflammation. Perit Dial Int 38:447–454
Mager DR, Hager A, Ooi PH, Siminoski K, Gilmour SM, Yap JYK (2019) Persistence of sarcopenia after pediatric liver transplantation is associated with poorer growth and recurrent hospital admissions. JPEN J Parenter Enteral Nutr 43:271–280
Van den Heuvel M, Voskuijl W, Chidzalo K, Kerac M, Reijneveld SA, Bandsma R et al (2017) Developmental and behavioural problems in children with severe acute malnutrition in Malawi: a cross-sectional study. J Glob Health 7:020416
Dedhia PH, White Y, Dillman JR, Adler J, Jarboe MD, Teitelbaum DH, Hirschl RB, Gadepalli SK (2018) Reduced paraspinous muscle area is associated with post-colectomy complications in children with ulcerative colitis. J Pediatr Surg 53:477–482
Rayar M, Webber CE, Nayiager T, Sala A, Barr RD (2013) Sarcopenia in children with acute lymphoblastic leukemia. J Pediatr Hematol Oncol 35:98–102
Oh J, Shin WJ, Jeong D, Yun TJ, Park CS, Choi ES, Choi JM, Gwak M, Song IK (2019) Low muscle mass as a prognostic factor for early postoperative outcomes in pediatric patients undergoing the fontan operation: a retrospective cohort study. J Clin Med 8:1257
Kawakubo N, Kinoshita Y, Souzaki R, Koga Y, Oba U, Ohga S, Taguchi T (2019) The influence of sarcopenia on high-risk neuroblastoma. J Surg Res 236:101–105
London WB, Castleberry RP, Matthay KK, Look AT, Seeger RC, Shimada H, Thorner P, Brodeur G, Maris JM, Reynolds CP, Cohn SL (2005) Evidence for an age cutoff greater than 365 days for neuroblastoma risk group stratification in the Children's Oncology Group. J Clin Oncol 23:6459–6465
Shimada H, Umehara S, Monobe Y, Hachitanda Y, Nakagawa A, Goto S, Gerbing RB, Stram DO, Lukens JN, Matthay KK (2001) International neuroblastoma pathology classification for prognostic evaluation of patients with peripheral neuroblastic tumors: a report from the Children's Cancer Group. Cancer. 92:2451–2461
Hishiki T, Matsumoto K, Ohira M, Kamijo T, Shichino H, Kuroda T et al (2018) Results of a phase II trial for high-risk neuroblastoma treatment protocol JN-H-07: a report from the Japan Childhood Cancer Group Neuroblastoma Committee (JNBSG). Int J Clin Oncol 23:965–973
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5:649–655
Davis MP, Panikkar R (2019) Sarcopenia associated with chemotherapy and targeted agents for cancer therapy. Ann Palliat Med 8:86–101
Metzger G, Sebastiao Y, Carsel A, Nishimura L, Fisher G, Deans K et al (2021) Establishing reference values for lean muscle mass in the pediatric patient. J Pediatr Gastroenterol Nutr 72:316–323
Lurz E, Patel H, Lebovic G, Quammie C, Woolfson J, Perez M et al (2020) Paediatric reference values for total psoas muscle area. J Cachexia Sarcopenia Muscle 11:405–414
Arrieta H, Hervás G, Rezola-Pardo C, Ruiz-Litago F, Iturburu M, Yanguas JJ et al (2019) Serum myostatin levels are higher in fitter, more active, and non-frail long-term nursing home residents and increase after a physical exercise intervention. Gerontology 65:229–239
Adams S, Segal R, McKenzie D, Vallerand J, Morielli A, Mackey J et al (2016) Impact of resistance and aerobic exercise on sarcopenia and dynapenia in breast cancer patients receiving adjuvant chemotherapy: a multicenter randomized controlled trial. Breast Cancer Res Treat 158:497–507
Mishra I, Scherer W, Snyder C, Geigle M, Berlanstein R, Topaloglu O (2012) Exercise interventions on health-related quality of life for people with cancer during active treatment. Cochrane Database Syst Rev 2012:CD008465
Hashida R, Kawaguchi T, Koya S, Hirota K, Goshima N, Yoshiyama T et al (2020) Impact of cancer rehabilitation on the prognosis of patients with hepatocellular carcinoma. Oncol Lett 19:2355–2367
Freerk TB, Wilhelm B, Julia B (2013) Clinical exercise interventions in pediatric oncology: a systematic review. Pediatr Res 74:366–374
Stössel S, Neu MA, Wingerter A, Bloch W, Zimmer P, Paret C, Malki KE, Baumann FT, Russo A, Henninger N, Lehmann N, Otto H, Faber J (2020) Benefits of exercise training for children and adolescents undergoing cancer treatment: results from the randomized controlled MUCKI trial. Front Pediatr 8:243
Author information
Authors and Affiliations
Contributions
NN conceptualized and designed the study, drafted the initial manuscript, and reviewed and revised the manuscript. KK conceptualized and designed the study, carried out the initial analyses, drafted the initial manuscript, and critically reviewed the manuscript for important intellectual content. TI, SN, AT, AK, and AS collected data, and reviewed and revised the manuscript. DH and YK conceptualized and designed the study, coordinated and supervised data collection, and reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Institutional Review Board of Kobe Children’s Hospital (approval number: R30-18).
Consent to participate
N/A.
Consent for publication
N/A.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Daniele De Luca
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nakamura, N., Kishimoto, K., Ishida, T. et al. Muscle mass change during chemotherapy in children with high-risk neuroblastoma: a retrospective case series of 24 patients. Eur J Pediatr 180, 3265–3271 (2021). https://doi.org/10.1007/s00431-021-04106-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-021-04106-y