Abstract
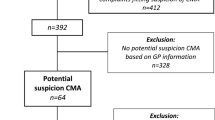
Cow’s milk allergy is a common food allergy in children with an incidence of 1–3%. The gold standard to diagnose cow’s milk allergy is a double-blinded placebo-controlled food challenge (DBPCFC). Previous studies show that reintroduction of cow’s milk is unsuccessful in 10–12% of patients. The aim of this retrospective study is to evaluate the effect of follow-up on the reintroduction of cow’s milk. We analyzed the data of patients with a negative DBPCFC for cow’s milk between 2014 and 2016 in three different departments. Questionnaires were used to compare the three ways of follow-up (no follow-up, follow-up in person or by telephone). Of the 336 children with a negative DBPCFC for cow’s milk, 128 questionnaires (41%) were returned. Reintroduction of cow’s milk was unsuccessful in 13.3% of the patients. There was no significant difference found between children with (73.8%) or without (26.2%) follow-up, or between follow-up by phone or personally. Whether this finding is caused by small numbers within this retrospective study should be investigated in future prospective studies.
Conclusion: Follow-up does not influence the reintroduction success rate of cow’s milk after a negative double-blinded placebo-controlled food challenge.
What is Known: Diagnosis of cow’s milk allergy by double-blinded placebo-controlled food challenge is the gold standard. • After a negative double-blinded placebo-controlled food challenge, reintroduction of cow’s milk in the child’s diet is unsuccessful in 10–12% of the children. • Recurrence of symptoms, aversion to the examined food, and fear are seen as the main reasons for unsuccessful introduction. | |
What is New: • Reintroduction after a negative double-blinded placebo-controlled food challenge is not influenced by different ways of follow-up. |


Similar content being viewed by others
Abbreviations
- CMA:
-
Cow’s milk allergy
- DBPCFC:
-
Double-blinded placebo-controlled food challenge
- CHC:
-
Child health care center
References
Dambacher WM, de Kort EH, Blom WM, Houben GF, de Vries E (2013) Double-blind placebo-controlled food challenges in children with alleged cow’s milk allergy: prevention of unnecessary elimination diets and determination of eliciting doses. Nutr J 12:22
Eigenmann PA, Caubet JC, Zamora SA (2006) Continuing food-avoidance diets after negative food challenges. Pediatr Allergy Immunol 17:601–605
Flammarion S, Santos C, Romero D, Thumerelle C, Deschildre A (2010 Jun 1) Changes in diet and life of children with food allergies after a negative food challenge. Allergy. 65(6):797–798
Gua J, Wang J (2017) Rate of food introduction after a negative oral food challenge in the paediatric population. J Allergy Clin Immunol Pract 5(2):475–476
Høst A (2002) Frequency of cow’s milk allergy in childhood. Ann Allergy Asthma Immunol 89(6 Suppl 1):33–37
Isolauri E, Sütas Y, Salo MK, Isosomppi R, Kaila M (1998) Elimination diet in cow’s milk allergy: risk for impaired growth in young children. J Pediatr 132:1004–1009
Koletzko S, Niggemann B, Arato A, Dias JA, Heuschkel R, Husby S, Mearin ML, Papadopoulou A, Ruemmele FM, Staiano A, Schäppi MG, Vandenplas Y (2012) Diagnostic approach and management of cow’s-milk protein allergy in infants and children: ESPGHAN GI committee practical guidelines. J Pediatr Gastroenterol Nutr 55:221–229
Mehta H, Groetch M, Wang J (2013) Growth and nutritional concerns in children with food allergy. Curr Opin Allergy Clin Immunol 13:275–279
Sampson HA (2001) Utility of food-specific IgE concentrations in predicting symptomatic food allergy. J Allergy Clin Immunol 107:891–896
van der Valk JP, Gerth van Wijk R, Vergouwe Y, de Jong NW (2015) Failure of introduction of food allergens after negative oral food challenge tests in children. Eur J Pediatr 174:1093–1099
Author information
Authors and Affiliations
Contributions
Author 1 collected the data, performed the analysis and wrote the paper. Author 2 and 3 conceived and designed the analysis. Author 4 conceived and designed the analysis and wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was submitted to the Medical Ethical Committee of the Maxima Medical Centre for approval. The Committee concluded that no Ethical Approval was required as it concerned a retrospective study.
Additional information
Communicated by Nicole Ritz
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schrijvers, M., Hendriks, T., Mars, H. et al. The effect of follow-up after a negative double-blinded placebo-controlled cow’s milk challenge on successful reintroduction. Eur J Pediatr 178, 1113–1117 (2019). https://doi.org/10.1007/s00431-019-03385-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-019-03385-w