Abstract
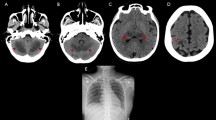
Oral polio vaccine (OPV) has been used safely and efficiently for more than 40 years in preventive medicine. Vaccine-associated paralytic poliomyelitis (VAPP) is a rare adverse event of OPV due to reversion of the vaccine strain virus to a neurovirulent strain. VAPP can occur in healthy recipients or their close contacts. However, persons with primary humoral immunodeficiencies are at a much higher risk. X-linked agammaglobulinemia (XLA) is a prototypic humoral deficiency caused by mutations in the Bruton’s tyrosine kinase (BTK) gene. In addition to susceptibility to bacterial infections, patients with XLA are especially prone to enteroviruses. Here, we describe the occurrence of VAPP in a 15-month old Iranian boy. The child had received four doses of OPV, administered at birth, 2, 4, and 6 months of age. The patient’s infectious history was unremarkable. Laboratory evaluation revealed low levels of immunoglobulin G and CD19+ B cells of less than 1% of the lymphocyte population. A novel insertion (c.685_686insTTAC) in the SH3 domain of the BTK gene was detected as the underlying cause. Immunodeficient recipients of OPV can excrete poliovirus vaccine strains for a long period and are at risk of developing flaccid paralysis. They could also serve as a source of reverted virulent poliovirus to be reintroduced into the general population. This patient presented for the first time with VAPP, without any history of other major infections in 15 months. This suggests that a negative history for recurrent infections does not exclude the presence of a primary defect in the immune system.

Similar content being viewed by others
Abbreviations
- AFP:
-
acute flaccid paralysis
- BTK:
-
Bruton’s tyrosine kinase
- IPV:
-
inactivated poliovirus vaccine
- iVDPV:
-
immunodeficiency-associated vaccine-derived polioviruses
- OPV:
-
oral polio vaccine
- PBMCs:
-
peripheral blood mononuclear cells
- TLR-8:
-
toll-like receptor 8
- VAPP:
-
vaccine-associated paralytic poliomyelitis
- VP1:
-
viral capsid protein 1
- XLA:
-
X-linked agammaglobulinemia
References
Alexander JP Jr, Gary HE Jr, Pallansch MA (1997) Duration of poliovirus excretion and its implications for acute flaccid paralysis surveillance: a review of the literature. J Infect Dis 175(Suppl 1):S176–S182
Centers for Disease Control and Prevention (2003) Progress toward poliomyelitis eradication—Ethiopia, Somalia, and Sudan, January 2001–October 2002. JAMA 289:1095–1097
Davis LE, Bodian D, Price D, Butler IJ, Vickers JH (1977) Chronic progressive poliomyelitis secondary to vaccination of an immunodeficient child. N Engl J Med 297:241–245
Kew OM, Sutter RW, de Gourville EM, Dowdle WR, Pallansch MA (2005) Vaccine-derived polioviruses and the endgame strategy for global polio eradication. Annu Rev Microbiol 59:587–635
Khetsuriani N, Prevots DR, Quick L, Elder ME, Pallansch M, Kew O, Sutter RW (2003) Persistence of vaccine-derived polioviruses among immunodeficient persons with vaccine-associated paralytic poliomyelitis. J Infect Dis 188:1845–1852
Lee WI, Torgerson TR, Schumacher MJ, Yel L, Zhu Q, Ochs HD (2005) Molecular analysis of a large cohort of patients with the hyper immunoglobulin M (IgM) syndrome. Blood 105:1881–1890
Parvaneh N, Shahmahmoudi S, Tabatabai H, Zahraei M, Mousavi T, Esteghamati AR, Gooya MM, Mamishi S, Nategh R, Kew OM (2007) Vaccine-associated paralytic poliomyelitis in a patient with MHC class II deficiency. J Clin Virol 39:145–148
Sochorová K, Horváth R, Rozková D, Litzman J, Bartunková J, Sedivá A, Spísek R (2007) Impaired Toll-like receptor 8-mediated IL-6 and TNF-alpha production in antigen-presenting cells from patients with X-linked agammaglobulinemia. Blood 109:2553–2556
Tsukada S, Saffran DC, Rawlings DJ, Parolini O, Allen RC, Klisak I, Sparkes RS, Kubagawa H, Mohandas T, Quan S, Belmont JW, Cooper MD, Conley ME, Witte ON (1993) Deficient expression of a B cell cytoplasmic tyrosine kinase in human X-linked agammaglobulinemia. Cell 72:279–290
Vetrie D, Vorechovský I, Sideras P, Holland J, Davies A, Flinter F, Hammarström L, Kinnon C, Levinsky R, Bobrow M, Smith CIE, Bentley DR (1993) The gene involved in X-linked agammaglobulinaemia is a member of the src family of protein-tyrosine kinases. Nature 361:226–233
Weibel RE, Benor DE (1996) Reporting vaccine-associated paralytic poliomyelitis: concordance between the CDC and the National Vaccine Injury Compensation Program. Am J Public Health 86:734–737
Winkelstein JA, Marino MC, Lederman HM, Jones SM, Sullivan K, Burks AW, Conley ME, Cunningham-Rundles C, Ochs HD (2006) X-linked agammaglobulinemia: report on a United States registry of 201 patients. Medicine (Baltimore) 85:193–202
Wright PF, Hatch MH, Kasselberg AG, Lowry SP, Wadlington WB, Karzon DT (1977) Vaccine-associated poliomyelitis in a child with sex-linked agammaglobulinemia. J Pediatr 91:408–412
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mamishi, S., Shahmahmoudi, S., Tabatabaie, H. et al. Novel BTK mutation presenting with vaccine-associated paralytic poliomyelitis. Eur J Pediatr 167, 1335–1338 (2008). https://doi.org/10.1007/s00431-008-0674-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-008-0674-5