Abstract
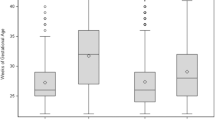
To assess whether lipid infusion could be a risk factor for parenteral nutrition-associated cholestasis (PNAC) in low birth weight neonates, 22 newborns with cholestasis (29.8 ± 1.6 weeks, 1298 ± 217 g) were compared with 22 without cholestasis (29.5 ± 1.7 weeks, 1286 ± 363 g). The mean level of peak direct bilirubin for the cholestasis group was 4.6 mg/dl compared to 1.2 mg/dl for the noncholestasis group. A univariate analysis revealed that PNAC was significantly related to duration of fasting (p = 0.008) and parenteral nutrition (p < 0.0001), days of antibiotics use (p = 0.025), positive C-reactive protein (p = 0.018) or gastric culture (p = 0.018), and feeding intolerance (p < 0.0001). Total amino acid amount (p < 0.0001), total lipid amount (p < 0.0001), and average daily lipid amount (p = 0.002) were significantly higher in the cholestasis group than in the noncholestasis group. Conversely, prenatal administration of dexamethasone was a significant protective factor of PNAC (p = 0.008). Logistic regression analysis revealed that the cumulative amount of lipid infusion was an independent risk factor for PNAC (p = 0.041; OR 1.174; CI 1.007–1.369). We suggest that decreasing the cumulative load of amino acids and intralipids with early trophic feeding, control of infection, and prenatal administration of dexamethasone could possibly attenuate the severity of PNAC.

Similar content being viewed by others
References
Aynsley-Green A (1983) Plasma hormone concentrations during enteral and parenteral nutrition in the human newborn. J Pediatr Gastroenterol Nutr 2(Suppl):108–112
Baird LL (2001) Protecting TPN and lipid infusions from light: reducing hydroperoxides in NICU patients. Neonatal Netw 20:17–22
Beath SV, Davies P, Papadopoulou A, Khan AR, Buick RG, Corkery JJ, Gornall P, Booth IW (1996) Parenteral nutrition-related cholestasis in postsurgical neonates: multivariate analysis of risk factors. J Pediatr Surg 31:604–606
Bindl L, Lutjohann D, Buderus S, Lentze MJ, von Bergmann K (2000) High plasma levels of phytosterols in patients on parenteral nutrition: a marker of liver dysfunction. J Pediatr Gastroenterol Nutr 31:313–316
Btaiche IF, Khalidi N (2002) Parenteral nutrition-associated liver complications in children. Pharmacotherapy 22:188–211
Bucuvalas JC, Goodrich AL, Blitzer BL, Suchy FJ (1985) Amino acids are potent inhibitors of bile acid uptake by liver plasma membrane vesicles isolated from suckling rats. Pediatr Res 19:1298–1304
Cavicchi M, Beau P, Crenn P, Degott C, Messing B (2000) Prevalence of liver disease and contributing factors in patients receiving home parenteral nutrition for permanent intestinal failure. Ann Intern Med 132:525–532
Colomb V, Jobert-Giraud A, Lacaille F, Goulet O, Fournet JC, Ricour C (2000) Role of lipid emulsions in cholestasis associated with long-term parenteral nutrition in children. J Parenter Enteral Nutr 24:345–350
Forchielli ML, Gura KM, Sandler R, Lo C (1995) Aminosyn PF or trophamine: which provides more protection from cholestasis associated with total parenteral nutrition? J Pediatr Gastroenterol Nutr 21:374–382
Goulet O, Girot R, Maier-Redelsperger M, Bougle D, Virelizier JL, Ricour C (1986) Hematologic disorders following prolonged use of intravenous fat emulsions in children. J Parenter Enteral Nutr 10:284–288
Haynes MJ, Stahlman MT, Bradley YB, Jacqueline CJ (2002) Hepatic pathology in extremely low birth weight baboons receiving long term intralipid. Pediatr Res 51:287A
Jacquemin E, Maurage C, Borderon JC, Gold F, Laugier J, Rolland JC (1995) Early cholestasis in premature infants receiving total parenteral nutrition: a possible consequence of shock and hypoxia. Eur J Pediatr Surg 5:259–261
Kelly DA (2006) Intestinal failure-associated liver disease: what do we know today? Gastroenterology 130 (2 Suppl 1):S70–S77
Koletzko B, Goulet O, Hunt J, Krohn K, Shamir R (2005) Guidelines on paediatric parenteral nutrition of the European Society of Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) and the European Society for Clinical Nutrition and Metabolism (ESPEN), supported by the European Society of Paediatric Research (ESPR). J Pediatr Gastroenterol Nutr 41(Suppl 2):S1–S87
La Scala GC, Le Coultre C, Roche BG, Bugmann P, Belli DC (1993) The addition of lipids increases the TPN-associated cholestasis in the rat. Eur J Pediatr Surg 3:224–227
Lima L, Obregon F, Cubillos S, Fazzino F, Jaimes I (2001) Taurine as a micronutrient in development and regeneration of the central nervous system. Nutr Neurosci 4:439–443
Militante JD, Lombardini JB (2002) Taurine: evidence of physiological function in the retina. Nutr Neurosci 5:75–90
Ogle CK, Zuo L, Mao JX, Alexander JW, Fischer JE, Nussbaum MS (1995) Differential expression of intestinal and splenic cytokines after parenteral nutrition. Arch Surg 130:1307–1308
Owings E, Georgeson K (2000) Management of cholestasis in infants with very low birth weight. Semin Pediatr Surg 9:96–102
Sondheimer JM, Asturias E, Cadnapaphornchai M (1998) Infection and cholestasis in neonates with intestinal resection and long-term parenteral nutrition. J Pediatr Gastroenterol Nutr 27:131–137
Spear ML, Spear M, Cohen AR, Pereira GR (1990) Effect of fat infusions on platelet concentration in premature infants. J Parenter Enteral Nutr 14:165–168
Spencer AU, Yu S, Tracy TF, Aouthmany MM, Llanos A, Brown MB, Brown M, Shulman RJ, Hirschl RB, Derusso PA, Cox J, Dahlgren J, Strouse PJ, Groner JI, Teitelbaum DH (2005) Parenteral nutrition-associated cholestasis in neonates: multivariate analysis of the potential protective effect of taurine. J Parenter Enteral Nutr 29:337–343
Suchy FJ, Bucuvalas JC, Goodrich AL, Moyer MS, Blitzer BL (1986) Taurocholate transport and Na-K ATPase activity in fetal and neonatal rat liver plasma membrane vesicles. Am J Physiol 251:G665–G673
Thureen PJ, Hay WW Jr (2001) Early aggressive nutrition in preterm infants. Semin Neonatol 6:403–415
Vileisis RA, Inwood RJ, Hunt CE (1980) Prospective controlled study of parenteral nutrition-associated cholestatic jaundice: effect of protein intake. J Pediatr 96:893–897
Walsh MC, Kliegman RM (1986) Necrotizing enterocolitis: treatment based on staging criteria. Pediatr Clin North Am 33:179–201
Watkins JB, Szczepanik P, Gould JB, Klein P, Lester R (1975) Bile salt metabolism in the human premature infant. Preliminary observations of pool size and synthesis rate following prenatal administration of dexamethasone and phenobarbital. Gastroenterology 69:706–713
Wright K, Ernst KD, Gaylord MS, Dawson JP, Burnette TM (2003) Increased incidence of parenteral nutrition-associated cholestasis with aminosyn PF compared to trophamine. J Perinatol 23:444–450
Ziegler EE, Thureen PJ, Carlson SJ (2002) Aggressive nutrition of the very low birthweight infant. Clin Perinatol 29:225–244
Acknowledgement
A part of this study was poster presented in the European Society for Paediatric Research (ESPR) meeting in Bilbao, Spain from 27–30 September 2003.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shin, J.I., Namgung, R., Park, M.S. et al. Could lipid infusion be a risk for parenteral nutrition-associated cholestasis in low birth weight neonates?. Eur J Pediatr 167, 197–202 (2008). https://doi.org/10.1007/s00431-007-0454-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-007-0454-7