Abstract
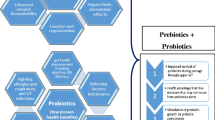
Nowadays, use of prebiotics as feed and food additives has received increasing interest because of the beneficial effects of prebiotics on the health of animals and humans. One of the beneficial effects of prebiotics is stimulation of immune system, which can be direct or indirect through increasing population of beneficial microbes or probiotics, especially lactic acid bacteria and bifidobacteria, in the gut. An important mechanism of action of probiotics and prebiotics, by which they can affect the immune system, is changing the expression of cytokines. The present review tried to summarize the findings of studies that investigated the effects of prebiotics on immune system with focusing on their effects on cytokine expression. Generally, most of reviewed studies indicated beneficial effects for prebiotics in terms of improving immune system, by increasing the expression of anti-inflammatory cytokines, while reducing the expressions of proinflammatory cytokines. However, most of studies mainly considered the indirect effects of prebiotics on the immune system (through changing the composition and population of gut microbiota), and their direct effects still need to be further studied using prebiotics with different degree of polymerization in different hosts.
Similar content being viewed by others
References
Badia R, Brufau MT, Guerrero-Zamora AM, Lizardo R, Dobrescu I, Martin-Venegas R, Ferrer R, Salmon H, Martínez P, Brufau J (2012) β-Galactomannan and Saccharomyces cerevisiae var. Boulardii modulate the immune response against Salmonella enterica serovar Typhimurium in porcine intestinal epithelial and dendritic cells. Clin Vaccine Immunol 19:368–376
Badia R, Zanello G, Chevaleyre C, Lizardo R, Meurens F, Martínez P, Brufau J, Salmon H (2012) Effect of Saccharomyces cerevisiae var. Boulardii and β-galactomannan oligosaccharide on porcine intestinal epithelial and dendritic cells challenged in vitro with Escherichia coli F4 (K88). Vet Res 43:4–15
Bandeira A, Mota-Santos T, Itohara S, Degermann S, Heusser C, Tonegawa S, Coutinho A (1990) Localization of gamma/delta T cells to the intestinal epithelium is independent of normal microbial colonization. J Exp Med 172:239–244
Brown AJ, Goldsworthy SM, Barnes AA, Eilert MM, Tcheang L, Daniels D, Muir AI, Wigglesworth MJ, Kinghorn I, Fraser NJ (2003) The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem 278:11312–11319
Cani PD, Possemiers S, Van De Wiele T, Guiot Y, Everard A, Rottier O, Geurts L, Naslain D, Neyrinck A, Lambert DM (2009) Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 58:1091–1103
Cavaglieri CR, Nishiyama A, Fernandes LC, Curi R, Miles EA, Calder PC (2003) Differential effects of short-chain fatty acids on proliferation and production of pro-and anti-inflammatory cytokines by cultured lymphocytes. Life Sci 73:1683–1690
Clark RSB, Kochanek P (eds) (2012) Brain injury. Springer Science & Business Media, Berlin
Dehghan P, Gargari BP, Jafar-Abadi MA (2014) Oligofructose-enriched inulin improves some inflammatory markers and metabolic endotoxemia in women with type 2 diabetes mellitus: a randomized controlled clinical trial. Nutrition 30:418–423
Denji KA, Mansour MR, Akrami R, Ghobadi S, Jafarpour S, Mirbeygi S (2015) Effect of dietary prebiotic mannan oligosaccharide (MOS) on growth performance, intestinal microflora, body composition, haematological and blood serum biochemical parameters of rainbow trout (Oncorhynchus mykiss) juveniles. J Fish Aquat Sci 10:255
Deshmane SL, Kremlev S, Amini S, Sawaya BE (2009) Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interferon Cytokine Res 29:313–326
Dinarello CA (2011) Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 117:3720–3732
Faghfoori Z, Navai L, Shakerhosseini R, Somi MH, Nikniaz Z, Norouzi MF (2011) Effects of an oral supplementation of germinated barley foodstuff on serum tumour necrosis factor-α, interleukin-6 and-8 in patients with ulcerative colitis. Ann Clin Biochem 48:233–237
Furrie E, Macfarlane S, Kennedy A, Cummings JH, Walsh SV, O’neil DA, Macfarlane GT (2005) Synbiotic therapy (Bifidobacterium longum/Synergy 1) initiates resolution of inflammation in patients with active ulcerative colitis: a randomised controlled pilot trial. Gut 54:242–249
Gibson GR, Probert HM, Van Loo J, Rastall RA, Roberfroid MB (2004) Dietary modulation of the human colonic microbiota: updating the concept of prebiotics. Nutr Res Rev 17:259–275
Gibson GR, Roberfroid MB (1995) Dietary modulation of the colonic microbiota: introducing the concept of prebiotics. J Nutr 125:1401–1412
Herfel TM, Jacobi SK, Lin X, Fellner V, Walker DC, Jouni ZE, Odle J (2011) Polydextrose enrichment of infant formula demonstrates prebiotic characteristics by altering intestinal microbiota, organic acid concentrations, and cytokine expression in suckling piglets. J Nutr 141:2139–2145
Hoentjen F, Tannock G, Mulder C, Dieleman L (2006) The prebiotic combination inulin/oligofructose prevents colitis in HLA-B27 rats by immunomodulation and changes in intestinal microflora. Eur J Gastroenterol Hepatol 18:A27
Hoentjen F, Welling GW, Harmsen HJ, Zhang X, Snart J, Tannock GW, Lien K, Churchill TA, Lupicki M, Dieleman LA (2005) Reduction of colitis by prebiotics in HLA-B27 transgenic rats is associated with microflora changes and immunomodulation. Inflamm Bowel Dis 11:977–985
Hosono A, Ozawa A, Kato R, Ohnishi Y, Nakanishi Y, Kimura T, Nakamura R (2003) Dietary fructooligosaccharides induce immunoregulation of intestinal IgA secretion by murine Peyer’s patch cells. Biosci Biotechnol Biochem 67:758–764
Huang RL, Deng ZY, Yang CB, Yin YL, Xie MY, Wu GY, Li TJ, Li LL, Tang ZR, Kang P (2007) Dietary oligochitosan supplementation enhances immune status of broilers. J Sci Food Agric 87:153–159
Ito H, Takemura N, Sonoyama K, Kawagishi H, Topping DL, Conlon MA, Morita T (2011) Degree of polymerization of inulin-type fructans differentially affects number of lactic acid bacteria, intestinal immune functions, and immunoglobulin A secretion in the rat cecum. J Agric Food Chem 59:5771–5778
Jahromi MF, Altaher YW, Shokryazdan P, Ebrahimi R, Ebrahimi M, Idrus Z, Goh YM, Tufarelli V, Liang JB (2015) Dietary supplementation of a mixture of Lactobacillus strains enhances performance of broiler chickens raised under heat stress conditions. Int J Biometeorol. doi:10.1007/s00484-015-1103-x
Jahromi MF, Liang JB, Shokryazdan P, Ebrahimi R, Navidshad B, Abdullah N (2015) Extraction and characterization of oligosaccharides from palm kernel cake. Bioresources 11:674–695
Khadka A (2014) Interleukins in therapeutics. PharmaTutor 2:67–72
Klatt NR, Canary LA, Sun X, Vinton CL, Funderburg NT, Morcock DR, Quiñones M, Deming CB, Perkins M, Hazuda DJ (2013) Probiotic/prebiotic supplementation of antiretrovirals improves gastrointestinal immunity in SIV-infected macaques. J Clin Invest 123:903–907
Kogut MH, Swaggerty CL (2012) Effects of prebiotics and probiotics on the host immune response. In: Callaway TR, Ricke SC (eds) Direct-fed microbials and prebiotics for animals. Springer, New York
Lackie J (2010) A dictionary of biomedicine. Oxford University Press, Oxford
Langen LV, Mirjam A, Dieleman LA (2009) Prebiotics in chronic intestinal inflammation. Inflamm Bowel Dis 15:454–462
Lecerf J-M, Dépeint F, Clerc E, Dugenet Y, Niamba CN, Rhazi L, Cayzeele A, Abdelnour G, Jaruga A, Younes H (2012) Xylo-oligosaccharide (XOS) in combination with inulin modulates both the intestinal environment and immune status in healthy subjects, while XOS alone only shows prebiotic properties. Br J Nutr 108:1847–1858
Macdonald TT, Monteleone G (2001) IL-12 and Th1 immune responses in human Peyer’s patches. Trends Immunol 22:244–247
Martin R, Nauta A, Ben Amor K, Knippels L, Knol J, Garssen J (2010) Early life: gut microbiota and immune development in infancy. Benef Microbes 1:367–382
Mccarville J (2012) Effects of prebiotic fibre diets on rat mucosal intestinal and systemic immunity and in vitro mechanistic analysis of anti-inflammatory effects of Lactobacillus strains on rat and human intestinal epithelial cells. http://hdl.handle.net/10155/275
Medzhitov R (2007) Recognition of microorganisms and activation of the immune response. Nature 449:819–826
Murarolli VDA, Burbarelli MFC, Polycarpo GV, Ribeiro PAP, Moro MEG, Albuquerque R (2014) Prebiotic, probiotic and symbiotic as alternative to antibiotics on the performance and Immune response of broiler chickens. Rev Bras Cienc Avic 16:279–284
Nagura T, Hachimura S, Hashiguchi M, Ueda Y, Kanno T, Kikuchi H, Sayama K, Kaminogawa S (2002) Suppressive effect of dietary raffinose on T-helper 2 cell-mediated immunity. Br J Nutr 88:421–426
Olivier JG, Van Aardenne JA, Dentener FJ, Pagliari V, Ganzeveld LN, Peters JA (2005) Recent trends in global greenhouse gas emissions: regional trends 1970–2000 and spatial distribution of key sources in 2000. Environ Sci 2:81–99
Oyofo BA, Deloach JR, Corrier DE, Norman JO, Ziprin RL, Mollenhauer HH (1989) Prevention of Salmonella typhimurium colonization of broilers with d-mannose. Poult Sci 68:1357–1360
Rakoff-Nahoum S, Paglino J, Eslami-Varzaneh F, Edberg S, Medzhitov R (2004) Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118:229–241
Rodríguez-Cabezas ME, Camuesco D, Arribas B, Garrido-Mesa N, Comalada M, Bailón E, Cueto-Sola M, Utrilla P, Guerra-Hernández E, Pérez-Roca C (2010) The combination of fructooligosaccharides and resistant starch shows prebiotic additive effects in rats. Clin Nutr 29:832–839
Roller M, Rechkemmer G, Watzl B (2004) Prebiotic inulin enriched with oligofructose in combination with the probiotics Lactobacillus rhamnosus and Bifidobacterium lactis modulates intestinal immune functions in rats. J Nutr 134:153–156
Seifert S, Watzl B (2007) Inulin and oligofructose: review of experimental data on immune modulation. J Nutr 137:2563–2567
Shokryazdan P, Sieo CC, Kalavathy R, Liang JB, Alitheen NB, Faseleh Jahromi M, Ho YW (2014) Probiotic potential of Lactobacillus strains with antimicrobial activity against some human pathogenic strains. Biomed Res Int. doi:10.1155/2014/927268
Sims JE, March CJ, Cosman D, Widmer MB, Macdonald HR, Mcmahan CJ, Grubin CE, Wignall JM, Jackson JL, Call SM (1988) cDNA expression cloning of the IL-1 receptor, a member of the immunoglobulin superfamily. Science 241:585–589
Steed H, Macfarlane S (2009) Mechanisms of prebiotic impact on health. In: Charalampopoulos D, Rastall RA (eds) Prebiotics and probiotics science and technology. Springer Science & Business Media, Berlin
Takahashí T, Nakagawa E, Nara T, Yajima T, Kuwata T (1998) Effects of orally ingested Bifidobacterium longum on the mucosal IgA response of mice to dietary antigens. Biosci Biotechnol Biochem 62:10–15
Vance RE, Isberg RR, Portnoy DA (2009) Patterns of pathogenesis: discrimination of pathogenic and nonpathogenic microbes by the innate immune system. Cell Host Microbe 6:10–21
Vulevic J, Drakoularakou A, Yaqoob P, Tzortzis G, Gibson GR (2008) Modulation of the fecal microflora profile and immune function by a novel trans-galactooligosaccharide mixture (B-GOS) in healthy elderly volunteers. Am J Clin Nutr 88:1438–1446
Wilson KH, Blitchington RB (1996) Human colonic biota studied by ribosomal DNA sequence analysis. Appl Environ Microbiol 62:2273–2278
Younger P (2007) Stedman’s medical dictionary. Ref Rev 21:41–42
Zhang J-M, An J (2007) Cytokines, inflammation and pain. Int Anesthesiol Clin 45:27–37
Acknowledgments
The present study was supported by the Ministry of Higher Education Malaysia under the LRGS Fasa 1/2012 grant, UPM/700-1/3/LRGS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest with respect to the authorship and/or publication of this article.
Rights and permissions
About this article
Cite this article
Shokryazdan, P., Faseleh Jahromi, M., Navidshad, B. et al. Effects of prebiotics on immune system and cytokine expression. Med Microbiol Immunol 206, 1–9 (2017). https://doi.org/10.1007/s00430-016-0481-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-016-0481-y