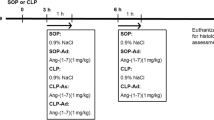
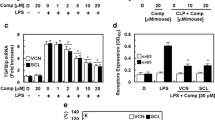
Abstract
Staphylococcus aureus is a Gram-positive bacteria described as an important causative agent of sepsis. The contact between host leukocytes and bacteria activates the innate immune response. Nitric oxide, tumor necrosis factor (TNF)-α and interleukin (IL)-1β play a key role in increasing microbicidal activity and controlling cell influx into infectious focus. Contrarily, IL-10 acts as an anti-inflammatory cytokine and bacterial killing suppressor. Immunoregulatory properties have also been attributed to hormones, including cholecystokinin (CCK). CCK protects cardiovascular function and inhibits the inflammatory response induced by lipopolysaccharide, product derived from Gram-negative bacteria. Nevertheless, the role of CCK during Gram-positive infection remains a literature gap. Our aims were to investigate whether CCK protects rats against bacterial dissemination during sepsis induced by S. aureus. We determined whether CCK modulates local and systemic inflammatory response, as well as the cell migration into the infectious focus and the bactericidal capacity of leukocytes. Our results revealed that proglumide (nonselective CCK receptor antagonist) pretreated rats showed higher bacterial counts in blood and peritoneal lavage fluid (PLF) and reduced TNF-α and IL-10 levels in PLF. Moreover, the dissemination of S. aureus may be related to the failure of neutrophil and macrophage migration into the peritoneal cavity. Also, CCK improved the phagocytic and bactericidal ability of these inflammatory cells. Noteworthy is that the adoptive transfer of CCK-treated neutrophils and macrophages in septic rats improved immune defense, reducing bacterial number in blood and PLF. All together, our study clearly demonstrates an important protective role of CCK against sepsis induced by S. aureus.






Similar content being viewed by others
References
Calandra T, Cohen J (2005) The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Crit Care Med 33(7):1538–1548
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, Cohen J, Opal SM, Vincent JL, Ramsay G (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International sepsis definitions conference. Intensive Care Med 29(4):530–538. doi:10.1007/s00134-003-1662-x
Naber CK (2009) Staphylococcus aureus bacteremia: epidemiology, pathophysiology, and management strategies. Clin Infect Dis 48(Suppl 4):S231–S237. doi:10.1086/598189
Martin GS, Mannino DM, Eaton S, Moss M (2003) The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 348(16):1546–1554. doi:10.1056/NEJMoa022139
Marra AR, Camargo LF, Pignatari AC, Sukiennik T, Behar PR, Medeiros EA, Ribeiro J, Girao E, Correa L, Guerra C, Brites C, Pereira CA, Carneiro I, Reis M, de Souza MA, Tranchesi R, Barata CU, Edmond MB (2011) Nosocomial bloodstream infections in Brazilian hospitals: analysis of 2,563 cases from a prospective nationwide surveillance study. J Clin Microbiol 49(5):1866–1871. doi:10.1128/JCM.00376-11
Vincent JL, Sakr Y, Sprung CL, Ranieri VM, Reinhart K, Gerlach H, Moreno R, Carlet J, Le Gall JR, Payen D (2006) Sepsis in European intensive care units: results of the SOAP study. Crit Care Med 34(2):344–353
Miyake K (2004) Innate recognition of lipopolysaccharide by Toll-like receptor 4-MD-2. Trends Microbiol 12(4):186–192. doi:10.1016/j.tim.2004.02.009
Verdrengh M, Tarkowski A (1997) Role of neutrophils in experimental septicemia and septic arthritis induced by Staphylococcus aureus. Infect Immun 65(7):2517–2521
Ulloa L, Tracey KJ (2005) The “cytokine profile”: a code for sepsis. Trends Molec Med 11(2):56–63. doi:10.1016/j.molmed.2004.12.007
Bozza FA, Salluh JI, Japiassu AM, Soares M, Assis EF, Gomes RN, Bozza MT, Castro-Faria-Neto HC, Bozza PT (2007) Cytokine profiles as markers of disease severity in sepsis: a multiplex analysis. Crit Care 11(2):R49. doi:10.1186/cc5783
Chandra R, Liddle RA (2007) Cholecystokinin. Curr Opin Endocrinol Diabetes Obes 14(1):63–67. doi:10.1097/MED.0b013e3280122850
Dufresne M, Seva C, Fourmy D (2006) Cholecystokinin and gastrin receptors. Physiol Rev 86(3):805–847. doi:10.1152/physrev.00014.2005
Sacerdote P, Wiedermann CJ, Wahl LM, Pert CB, Ruff MR (1991) Visualization of cholecystokinin receptors on a subset of human monocytes and in rat spleen. Peptides 12(1):167–176
Cuq P, Gross A, Terraza A, Fourmy D, Clerc P, Dornand J, Magous R (1997) mRNAs encoding CCKB but not CCKA receptors are expressed in human T lymphocytes and Jurkat lymphoblastoid cells. Life Sci 61(5):543–555
Xu SJ, Gao WJ, Cong B, Yao YX, Gu ZY (2004) Effect of lipopolysaccharide on expression and characterization of cholecystokinin receptors in rat pulmonary interstitial macrophages. Acta Pharmacol Sin 25(10):1347–1353
Cong B, Li SJ, Yao YX, Zhu GJ, Ling YL (2002) Effect of cholecystokinin octapeptide on tumor necrosis factor alpha transcription and nuclear factor-kappaB activity induced by lipopolysaccharide in rat pulmonary interstitial macrophages. World J Gastroenterol 8(4):718–723
Saia RS, Bertozi G, Mestriner FL, Antunes-Rodrigues J, Queiroz Cunha F, Carnio EC (2013) Cardiovascular and inflammatory response to cholecystokinin during endotoxemic shock. Shock 39(1):104–113. doi:10.1097/SHK.0b013e3182793e2e
Li S, Ni Z, Cong B, Gao W, Xu S, Wang C, Yao Y, Ma C, Ling Y (2007) CCK-8 inhibits LPS-induced IL-1beta production in pulmonary interstitial macrophages by modulating PKA, p38, and NF-kappaB pathway. Shock 27(6):678–686. doi:10.1097/shk.0b013e3180ze26dd
De la Fuente M, Campos M, Del Rio M, Hernanz A (1995) Inhibition of murine peritoneal macrophage functions by sulfated cholecystokinin octapeptide. Regul Pept 55(1):47–56
Carrasco M, Del Rio M, Hernanz A, De la Fuente M (1997) Inhibition of human neutrophil functions by sulfated and nonsulfated cholecystokinin octapeptides. Peptides 18(3):415–422
Bozkurt A, Cakir B, Ercan F, Yegen BC (2003) Anti-inflammatory effects of leptin and cholecystokinin on acetic acid-induced colitis in rats: role of capsaicin-sensitive vagal afferent fibers. Regul Pept 116(1–3):109–118
Zhao XY, Ling YL, Li YG, Meng AH, Xing HY (2005) Cholecystokinin octapeptide improves cardiac function by activating cholecystokinin octapeptide receptor in endotoxic shock rats. World J Gastroenterol 11(22):3405–3410
Miyamoto S, Shikata K, Miyasaka K, Okada S, Sasaki M, Kodera R, Hirota D, Kajitani N, Takatsuka T, Kataoka HU, Nishishita S, Sato C, Funakoshi A, Nishimori H, Uchida HA, Ogawa D, Makino H (2012) Cholecystokinin plays a novel protective role in diabetic kidney through anti-inflammatory actions on macrophage: anti-inflammatory effect of cholecystokinin. Diabetes 61(4):897–907. doi:10.2337/db11-0402
Alvarez A, Ibiza S, Hernandez C, Alvarez-Barrientos A, Esplugues JV, Calatayud S (2006) Gastrin induces leukocyte-endothelial cell interactions in vivo and contributes to the inflammation caused by Helicobacter pylori. FASEB J 20(13):2396–2398. doi:10.1096/fj.05-5696fje
Saia RS, Oliveira-Pelegrin GR, da Silva ME, Aguila FA, Antunes-Rodrigues J, Rocha MJ, Carnio EC (2011) Neonatal endotoxin exposure changes neuroendocrine, cardiovascular function and mortality during polymicrobial sepsis in adult rats. Regul Pept 169(1–3):21–30. doi:10.1016/j.regpep.2011.04.009
Sidiq T, Khajuria A, Suden P, Sharma R, Singh S, Suri KA, Satti NK, Johri RK (2011) Possible role of macrophages induced by an irridoid glycoside (RLJ-NE-299A) in host defense mechanism. Int Immunopharmacol 11(1):128–135. doi:10.1016/j.intimp.2010.10.017
Crosara-Alberto DP, Darini AL, Inoue RY, Silva JS, Ferreira SH, Cunha FQ (2002) Involvement of NO in the failure of neutrophil migration in sepsis induced by Staphylococcus aureus. Br J Pharmacol 136(5):645–658. doi:10.1038/sj.bjp.0704734
Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB (2004) Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 39(3):309–317. doi:10.1086/421946
Cosgrove SE (2006) The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis 42(Suppl 2):S82–S89. doi:10.1086/499406
Weiland TJ, Kent S, Voudouris NJ, Shulkes A (2005) The effect of lipopolysaccharide on cholecystokinin in murine plasma and tissue. Peptides 26(3):447–455. doi:10.1016/j.peptides.2004.10.006
Bercik P, Verdu EF, Foster JA, Lu J, Scharringa A, Kean I, Wang L, Blennerhassett P, Collins SM (2009) Role of gut-brain axis in persistent abnormal feeding behavior in mice following eradication of Helicobacter pylori infection. Am J Physiol 296(3):R587–R594. doi:10.1152/ajpregu.90752.2008
Ovington KS, Bacarese-Hamilton AJ, Bloom SR (1985) Nippostrongylus brasiliensis: changes in plasma levels of gastrointestinal hormones in the infected rat. Exp Parasitol 60(3):276–284
Ling YL, Meng AH, Zhao XY, Shan BE, Zhang JL, Zhang XP (2001) Effect of cholecystokinin on cytokines during endotoxic shock in rats. World J Gastroenterol 7(5):667–671
Grivennikov SI, Tumanov AV, Liepinsh DJ, Kruglov AA, Marakusha BI, Shakhov AN, Murakami T, Drutskaya LN, Forster I, Clausen BE, Tessarollo L, Ryffel B, Kuprash DV, Nedospasov SA (2005) Distinct and nonredundant in vivo functions of TNF produced by t cells and macrophages/neutrophils: protective and deleterious effects. Immunity 22(1):93–104. doi:10.1016/j.immuni.2004.11.016
van den Berg S, Laman JD, Boon L, ten Kate MT, de Knegt GJ, Verdijk RM, Verbrugh HA, Nouwen JL, Bakker-Woudenberg IA (2013) Distinctive cytokines as biomarkers predicting fatal outcome of severe Staphylococcus aureus bacteremia in mice. PLoS ONE 8(3):e59107. doi:10.1371/journal.pone.0059107
van der Poll T, Marchant A, Buurman WA, Berman L, Keogh CV, Lazarus DD, Nguyen L, Goldman M, Moldawer LL, Lowry SF (1995) Endogenous IL-10 protects mice from death during septic peritonitis. J Immunol 155(11):5397–5401
Howard M, Muchamuel T, Andrade S, Menon S (1993) Interleukin 10 protects mice from lethal endotoxemia. J Exp Med 177(4):1205–1208
Friedman G, Jankowski S, Marchant A, Goldman M, Kahn RJ, Vincent JL (1997) Blood interleukin 10 levels parallel the severity of septic shock. J Crit Care 12(4):183–187
Berg DJ, Kuhn R, Rajewsky K, Muller W, Menon S, Davidson N, Grunig G, Rennick D (1995) Interleukin-10 is a central regulator of the response to LPS in murine models of endotoxic shock and the Shwartzman reaction but not endotoxin tolerance. J Clin Invest 96(5):2339–2347. doi:10.1172/JCI118290
Latifi SQ, O’Riordan MA, Levine AD (2002) Interleukin-10 controls the onset of irreversible septic shock. Infect Immun 70(8):4441–4446
Alves-Filho JC, Spiller F, Cunha FQ (2010) Neutrophil paralysis in sepsis. Shock 34(Suppl 1):15–21. doi:10.1097/SHK.0b013e3181e7e61b
Benjamim CF, Ferreira SH, Cunha FQ (2000) Role of nitric oxide in the failure of neutrophil migration in sepsis. J Infect Dis 182(1):214–223. doi:10.1086/315682
Tavares-Murta BM, Cunha FQ, Ferreira SH (1998) The intravenous administration of tumor necrosis factor alpha, interleukin 8 and macrophage-derived neutrophil chemotactic factor inhibits neutrophil migration by stimulating nitric oxide production. Br J Pharmacol 124(7):1369–1374. doi:10.1038/sj.bjp.0701965
Ajuebor MN, Das AM, Virag L, Flower RJ, Szabo C, Perretti M (1999) Role of resident peritoneal macrophages and mast cells in chemokine production and neutrophil migration in acute inflammation: evidence for an inhibitory loop involving endogenous IL-10. J Immunol 162(3):1685–1691
Schroder JM (2000) Chemoattractants as mediators of neutrophilic tissue recruitment. Clin Dermatol 18(3):245–263
Henderson RB, Hobbs JA, Mathies M, Hogg N (2003) Rapid recruitment of inflammatory monocytes is independent of neutrophil migration. Blood 102(1):328–335. doi:10.1182/blood-2002-10-3228
Cailhier JF, Partolina M, Vuthoori S, Wu S, Ko K, Watson S, Savill J, Hughes J, Lang RA (2005) Conditional macrophage ablation demonstrates that resident macrophages initiate acute peritoneal inflammation. J Immunol 174(4):2336–2342
Issekutz AC, Issekutz TB (1993) Quantitation and kinetics of blood monocyte migration to acute inflammatory reactions, and IL-1 alpha, tumor necrosis factor-alpha, and IFN-gamma. J Immunol 151(4):2105–2115
Sacerdote P, Ruff MR, Pert CB (1988) Cholecystokinin and the immune system: receptor-mediated chemotaxis of human and rat monocytes. Peptides 9(Suppl 1):29–34
de la Fuente M, Drummond J, del Rio M, Carrasco M, Hernanz A (1996) Modulation of murine peritoneal macrophage functions by gastrin. Peptides 17(2):219–224
Pierangeli SS, Sonnenfeld G (1993) Treatment of murine macrophages with murine interferon-gamma and tumour necrosis factor-alpha enhances uptake and intracellular killing of Pseudomonas aeruginosa. Clin Exp Immunol 93(2):165–171
Nizet V (2007) Understanding how leading bacterial pathogens subvert innate immunity to reveal novel therapeutic targets. J Allergy Clin Immunol 120(1):13–22. doi:10.1016/j.jaci.2007.06.005
McMillen MA, Ferrara A, Adrian TE, Margolis DS, Schaefer HC, Zucker KA (1995) Cholecystokinin effect on human lymphocyte ionized calcium and mitogenesis. J Surg Res 58(2):149–158. doi:10.1006/jsre1995.1024
Acknowledgments
We would like to thank Marcelo Eduardo Batalhão for the technical assistance. The authors gratefully acknowledge the English grammar review by Renata Brancaleoni Mitchell and Ian Mitchell. This study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP).
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zuelli, F.M.G.C., Cárnio, E.C. & Saia, R.S. Cholecystokinin protects rats against sepsis induced by Staphylococcus aureus . Med Microbiol Immunol 203, 165–176 (2014). https://doi.org/10.1007/s00430-014-0328-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-014-0328-3