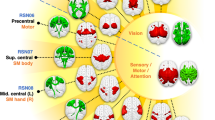
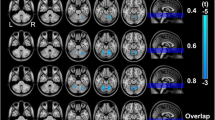
Abstract
For years, the cerebellum was left out of functional magnetic resonance imaging (fMRI) studies due to technological limitations. The advent of novel data acquisition and reconstruction strategies (e.g., whole-brain simultaneous multi-slice imaging) employing multi-channel array coils has overcome such limitations, ushering unprecedented improvements in temporal signal-to-noise ratio and spatiotemporal resolution. Here, we aim to provide a brief report on the deep cerebellar nuclei, specifically focusing on the dentate nuclei, the primary output nuclei, situated within both cognitive and motor cerebello-cerebral circuits. We highlight the importance of functional parcellation in refining our understanding of broad resting-state functional connectivity (RSFC) in both health and disease. First, we review work relevant to the functional topography of the dentate nuclei, including recent advances in functional parcellation. Next, we review RSFC studies using the dentate nuclei as seed regions of interest in neurological and psychiatric populations and discuss the potential benefits of applying functionally defined subdivisions. Finally, we discuss recent technological advances and underscore ultrahigh-field neuroimaging as a tool to potentiate functionally parcellated RSFC analyses in clinical populations.
Similar content being viewed by others
Data availability
As this work is a literature review, data sharing is not pertinent to this publication because no datasets were processed for the present work.
References
Akakin A, Peris-Celda M, Kilic T, Seker A, Gutierrez-Martin A, Rhoton A Jr (2014) The dentate nucleus and its projection system in the human cerebellum: the dentate nucleus microsurgical anatomical study. Neurosurgery 74(4):401–424. https://doi.org/10.1227/neu.0000000000000293. (discussion 424–405)
Andreasen NC, Paradiso S, O’Leary DS (1998) “Cognitive dysmetria” as an integrative theory of schizophrenia: a dysfunction in cortical-subcortical-cerebellar circuitry? Schizophr Bull 24(2):203–218. https://doi.org/10.1093/oxfordjournals.schbul.a033321
Anteraper SA, Guell X, Taylor HP, D’Mello A, Whitfield-Gabrieli S, Joshi G (2019) Intrinsic functional connectivity of dentate nuclei in autism spectrum disorder. Brain Connect 9(9):692–702. https://doi.org/10.1089/brain.2019.0692
Anteraper SA, Guell X, Collin G, Qi Z, Ren J, Nair A et al (2021) Abnormal function in dentate nuclei precedes the onset of psychosis: a resting-state fMRI study in high-risk individuals. Schizophr Bull 47(5):1421–1430. https://doi.org/10.1093/schbul/sbab038
Balsters JH, Cussans E, Diedrichsen J, Phillips KA, Preuss TM, Rilling JK, Ramnani N (2010) Evolution of the cerebellar cortex: the selective expansion of prefrontal-projecting cerebellar lobules. NeuroImage 49(3):2045–2052. https://doi.org/10.1016/j.neuroimage.2009.10.045
Balsters JH, Laird AR, Fox PT, Eickhoff SB (2014) Bridging the gap between functional and anatomical features of cortico-cerebellar circuits using meta-analytic connectivity modeling. Hum Brain Mapp 35(7):3152–3169. https://doi.org/10.1002/hbm.22392
Bernard JA, Peltier SJ, Benson BL, Wiggins JL, Jaeggi SM, Buschkuehl M et al (2014) Dissociable functional networks of the human dentate nucleus. Cereb Cortex 24(8):2151–2159. https://doi.org/10.1093/cercor/bht065
Bharti K, Khan M, Beaulieu C, Graham SJ, Briemberg H, Frayne R et al (2020) Involvement of the dentate nucleus in the pathophysiology of amyotrophic lateral sclerosis: a multi-center and multi-modal neuroimaging study. Neuroimage Clin 28:102385. https://doi.org/10.1016/j.nicl.2020.102385
Bond KM, Brinjikji W, Eckel LJ, Kallmes DF, McDonald RJ, Carr CM (2017) Dentate update: imaging features of entities that affect the dentate nucleus. AJNR Am J Neuroradiol 38(8):1467–1474. https://doi.org/10.3174/ajnr.A5138
Buckner RL (2013) The Cerebellum and Cognitive Function: 25 Years of Insight from Anatomy and Neuroimaging. Neuron 80(3):807–815. https://doi.org/10.1016/j.neuron.2013.10.044
Buckner RL, Krienen FM, Castellanos A, Diaz JC, Yeo BT (2011) The organization of the human cerebellum estimated by intrinsic functional connectivity. J Neurophysiol 106(5):2322–2345. https://doi.org/10.1152/jn.00339.2011
Deistung A, Schäfer A, Schweser F, Biedermann U, Güllmar D, Trampel R et al (2013) High-resolution MR imaging of the human brainstem in vivo at 7 Tesla. Front Hum Neurosci 7:710. https://doi.org/10.3389/fnhum.2013.00710
Deistung A, Stefanescu MR, Ernst TM, Schlamann M, Ladd ME, Reichenbach JR, Timmann D (2016) Structural and functional magnetic resonance imaging of the cerebellum: considerations for assessing cerebellar ataxias. Cerebellum 15(1):21–25. https://doi.org/10.1007/s12311-015-0738-9
Deshpande G, Zhao X, Robinson J (2022) Functional parcellation of the hippocampus based on its layer-specific connectivity with default mode and dorsal attention networks. Neuroimage 254:119078. https://doi.org/10.1016/j.neuroimage.2022.119078
Diedrichsen J, Maderwald S, Küper M, Thürling M, Rabe K, Gizewski ER et al (2011) Imaging the deep cerebellar nuclei: a probabilistic atlas and normalization procedure. Neuroimage 54(3):1786–1794. https://doi.org/10.1016/j.neuroimage.2010.10.035
Dum RP, Strick PL (2003) An unfolded map of the cerebellar dentate nucleus and its projections to the cerebral cortex. J Neurophysiol 89(1):634–639. https://doi.org/10.1152/jn.00626.2002
Grbatinić I, Milošević N (2016) Classification of adult human dentate nucleus border neurons: artificial neural networks and multidimensional approach. J Theor Biol 404:273–284. https://doi.org/10.1016/j.jtbi.2016.06.011
Guell X, Schmahmann J (2020) Cerebellar functional anatomy: a didactic summary based on human fMRI evidence. Cerebellum 19(1):1–5. https://doi.org/10.1007/s12311-019-01083-9
Guell X, Gabrieli JD, Schmahmann JD (2018) Triple representation of language working memory social and emotion processing in the cerebellum: convergent evidence from task and seed-based resting-state fMRI analyses in a single large cohort. NeuroImage 172:437–449. https://doi.org/10.1016/j.neuroimage.2018.01.082
Guell X, D’Mello AM, Hubbard NA, Romeo RR, Gabrieli JDE, Whitfield-Gabrieli S et al (2020) Functional territories of human dentate nucleus. Cereb Cortex 30(4):2401–2417. https://doi.org/10.1093/cercor/bhz247
Habas C (2010) Functional imaging of the deep cerebellar nuclei: a review. Cerebellum 9(1):22–28. https://doi.org/10.1007/s12311-009-0119-3
Hoppenbrouwers SS, Schutter DJ, Fitzgerald PB, Chen R, Daskalakis ZJ (2008) The role of the cerebellum in the pathophysiology and treatment of neuropsychiatric disorders: a review. Brain Res Rev 59(1):185–200. https://doi.org/10.1016/j.brainresrev.2008.07.005
Kebschull JM, Richman EB, Ringach N, Friedmann D, Albarran E, Kolluru SS et al (2020) Cerebellar nuclei evolved by repeatedly duplicating a conserved cell-type set. Science. https://doi.org/10.1126/science.abd5059
Koziol LF, Budding D, Andreasen N, D’Arrigo S, Bulgheroni S, Imamizu H et al (2014) Consensus paper: the cerebellum’s role in movement and cognition. Cerebellum (Lond, Engl) 13(1):151–177. https://doi.org/10.1007/s12311-013-0511-x
Kuper M, Dimitrova A, Thurling M, Maderwald S, Roths J, Elles HG et al (2011) Evidence for a motor and a non-motor domain in the human dentate nucleus—an fMRI study. Neuroimage 54(4):2612–2622. https://doi.org/10.1016/j.neuroimage.2010.11.028
Lee YJ, Guell X, Hubbard NA, Siless V, Frosch IR, Goncalves M et al (2021) Functional alterations in cerebellar functional connectivity in anxiety disorders. Cerebellum 20(3):392–401. https://doi.org/10.1007/s12311-020-01213-8
Margulies DS, Ghosh SS, Goulas A, Falkiewicz M, Huntenburg JM, Langs G et al (2016) Situating the default-mode network along a principal gradient of macroscale cortical organization. Proc Natl Acad Sci 113(44):12574–12579. https://doi.org/10.1073/pnas.1608282113
Maric D (2010) Qualitative and quantitative analysis of adult human dentate nucleus neurons morphology. University of Novi Sad, Belgrade, Serbia
Matano S (2001) Brief communication: proportions of the ventral half of the cerebellar dentate nucleus in humans and great apes. Am J Phys Anthropol 114(2):163–165. https://doi.org/10.1002/1096-8644(200102)114:2%3c163::AID-AJPA1016%3e3.0.CO;2-F
Middleton FA, Strick PL (1998) Cerebellar output: motor and cognitive channels. Trends Cogn Sci 2(9):348–354. https://doi.org/10.1016/S1364-6613(98)01220-0
Nemani A, Lowe MJ (2021) Seed-based test-retest reliability of resting state functional magnetic resonance imaging at 3T and 7T. Med Phys 48(10):5756–5764. https://doi.org/10.1002/mp.15210
Olivito G, Clausi S, Laghi F, Tedesco AM, Baiocco R, Mastropasqua C et al (2017) Resting-state functional connectivity changes between dentate nucleus and cortical social brain regions in autism spectrum disorders. Cerebellum 16(2):283–292. https://doi.org/10.1007/s12311-016-0795-8
Olivito G, Serra L, Marra C, Di Domenico C, Caltagirone C, Toniolo S et al (2020) Cerebellar dentate nucleus functional connectivity with cerebral cortex in Alzheimer’s disease and memory: a seed-based approach. Neurobiol Aging 89:32–40. https://doi.org/10.1016/j.neurobiolaging.2019.10.026
Palesi F, Ferrante M, Gaviraghi M, Misiti A, Savini G, Lascialfari A et al (2021) Motor and higher-order functions topography of the human dentate nuclei identified with tractography and clustering methods. Hum Brain Mapp 42(13):4348–4361. https://doi.org/10.1002/hbm.25551
Priovoulos N, Andersen M, Dumoulin SO, Boer VO, van der Zwaag W (2023) High-resolution motion-corrected 7.0-T MRI to derive morphologic measures from the human cerebellum in vivo. Radiology 307(2):220989. https://doi.org/10.1148/radiol.220989
Ramnani N (2006) The primate cortico-cerebellar system: anatomy and function. Nat Rev Neurosci 7(7):511–522. https://doi.org/10.1038/nrn1953
Raudel S-C, Agnès G, José MD-G (2007) The cerebellar interpositus nucleus and the dynamic control of learned motor responses. J Neurosci 27(25):6620. https://doi.org/10.1523/JNEUROSCI.0488-07.2007
Sbardella E, Upadhyay N, Tona F, Prosperini L, De Giglio L, Petsas N et al (2017) Dentate nucleus connectivity in adult patients with multiple sclerosis: functional changes at rest and correlation with clinical features. Mult Scler 23(4):546–555. https://doi.org/10.1177/1352458516657438
Schmahmann JD (1998) Dysmetria of thought: clinical consequences of cerebellar dysfunction on cognition and affect. Trends Cogn Sci 2(9):362–371. https://doi.org/10.1016/S1364-6613(98)01218-2
Seoane S, Modrono C, Gonzalez-Mora JL, Janssen N (2022) Medial temporal lobe contributions to resting-state networks. Brain Struct Funct 227(3):995–1012. https://doi.org/10.1007/s00429-021-02442-1
Sereno MI, Diedrichsen J, Tachrount M, Testa-Silva G, d’Arceuil H, De Zeeuw C (2020) The human cerebellum has almost 80% of the surface area of the neocortex. Proc Natl Acad Sci U S A 117(32):19538–19543. https://doi.org/10.1073/pnas.2002896117
Steele CJ, Anwander A, Bazin PL, Trampel R, Schaefer A, Turner R et al (2017) Human cerebellar sub-millimeter diffusion imaging reveals the motor and non-motor topography of the dentate nucleus. Cereb Cortex 27(9):4537–4548. https://doi.org/10.1093/cercor/bhw258
Stoodley CJ (2016) The cerebellum and neurodevelopmental disorders. Cerebellum 15(1):34–37. https://doi.org/10.1007/s12311-015-0715-3
Sultan F, Hamodeh S, Baizer JS (2010) The human dentate nucleus: a complex shape untangled. Neuroscience 167(4):965–968. https://doi.org/10.1016/j.neuroscience.2010.03.007
Thurling M, Kuper M, Stefanescu R, Maderwald S, Gizewski ER, Ladd ME, Timmann D (2011) Activation of the dentate nucleus in a verb generation task: a 7T MRI study. Neuroimage 57(3):1184–1191. https://doi.org/10.1016/j.neuroimage.2011.05.045
Tikoo S, Pietracupa S, Tommasin S, Bologna M, Petsas N, Bharti K et al (2020) Functional disconnection of the dentate nucleus in essential tremor. J Neurol 267(5):1358–1367. https://doi.org/10.1007/s00415-020-09711-9
Tikoo S, Suppa A, Tommasin S, Gianni C, Conte G, Mirabella G et al (2022) The cerebellum in drug-naive children with tourette syndrome and obsessive-compulsive disorder. Cerebellum 21(6):867–878. https://doi.org/10.1007/s12311-021-01327-7
Tona F, De Giglio L, Petsas N, Sbardella E, Prosperini L, Upadhyay N et al (2018) Role of cerebellar dentate functional connectivity in balance deficits in patients with multiple sclerosis. Radiology 287(1):267–275. https://doi.org/10.1148/radiol.2017170311
Uddin LQ, Yeo BTT, Spreng RN (2019) Towards a universal taxonomy of macro-scale functional human brain networks. Brain Topogr 32(6):926–942. https://doi.org/10.1007/s10548-019-00744-6
Upadhyay N, Suppa A, Piattella MC, Gianni C, Bologna M, Di Stasio F et al (2017) Functional disconnection of thalamic and cerebellar dentate nucleus networks in progressive supranuclear palsy and corticobasal syndrome. Parkinsonism Relat Disord 39:52–57. https://doi.org/10.1016/j.parkreldis.2017.03.008
van Oort ESB, Mennes M, Navarro Schroder T, Kumar VJ, Zaragoza Jimenez NI, Grodd W et al (2018) Functional parcellation using time courses of instantaneous connectivity. Neuroimage 170:31–40. https://doi.org/10.1016/j.neuroimage.2017.07.027
Van Overwalle F, Manto M, Cattaneo Z, Clausi S, Ferrari C, Gabrieli JDE et al (2020) Consensus paper: cerebellum and social cognition. Cerebellum 19(6):833–868. https://doi.org/10.1007/s12311-020-01155-1
Vu AT, Jamison K, Glasser MF, Smith SM, Coalson T, Moeller S et al (2017) Tradeoffs in pushing the spatial resolution of fMRI for the 7T human connectome project. Neuroimage 154:23–32. https://doi.org/10.1016/j.neuroimage.2016.11.049
Xie Y, Xi Y, Cui LB, Li C, Xu Y, Zhang Y et al (2021a) Altered functional connectivity of the dentate nuclei in patients with schizophrenia. Schizophr Res 233:16–23. https://doi.org/10.1016/j.schres.2021.06.035
Xie YJ, Xi YB, Cui LB, Guan MZ, Li C, Wang ZH et al (2021b) Functional connectivity of cerebellar dentate nucleus and cognitive impairments in patients with drug-naive and first-episode schizophrenia. Psychiatry Res 300:113937. https://doi.org/10.1016/j.psychres.2021.113937
Yang H, Wang N, Luo X, Lv H, Liu H, Fan G (2019) Altered functional connectivity of dentate nucleus in parkinsonian and cerebellar variants of multiple system atrophy. Brain Imaging Behav 13(6):1733–1745. https://doi.org/10.1007/s11682-019-00097-5
Yoshida J, Onate M, Khatami L, Vera J, Nadim F, Khodakhah K (2022) Cerebellar contributions to the basal ganglia influence motor coordination, reward processing, and movement vigor. J Neurosci 42(45):8406–8415. https://doi.org/10.1523/JNEUROSCI.1535-22.2022
Zhang X-Y, Wang J-J, Zhu J-N (2016) Cerebellar fastigial nucleus: from anatomic construction to physiological functions. Cerebellum Ataxias 3(1):9. https://doi.org/10.1186/s40673-016-0047-1
Funding
Research reported in this publication was supported by the National Institute of Mental Health of the National Institutes of Health under award number R03MH121879.
Author information
Authors and Affiliations
Contributions
SA conceptulaized the presented idea, conducted the literature review and wrote the first draft along with MK and JK. SA provided supervision to MK and KP for literature review and for preparing the tables. SA and XG performed the data analysis and verified the analytical methods for the four publications that are highlighted in this brief report. JK supervised MK during writing, review and editing. All authors contributed to writing and editing of the manuscript and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no financial or non-financial interests, and no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kulkarni, M., Kent, J.S., Park, K. et al. Resting-state functional connectivity-based parcellation of the human dentate nucleus: new findings and clinical relevance. Brain Struct Funct 228, 1799–1810 (2023). https://doi.org/10.1007/s00429-023-02665-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-023-02665-4