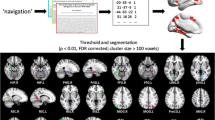
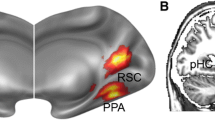
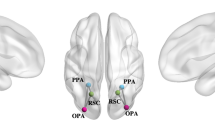
Abstract
Social navigation is a dynamic and complex process that requires the collaboration of multiple brain regions. However, the neural networks for navigation in a social space remain largely unknown. This study aimed to investigate the role of hippocampal circuit in social navigation from a resting-state fMRI data. Here, resting-state fMRI data were acquired before and after participants performed a social navigation task. By taking the anterior and posterior hippocampus (HPC) as the seeds, we calculated their connectivity with the whole brain using the seed-based static functional connectivity (sFC) and dynamic FC (dFC) approaches. We found that the sFC and dFC between the anterior HPC and supramarginal gyrus, sFC or dFC between posterior HPC and middle cingulate cortex, inferior parietal gyrus, angular gyrus, posterior cerebellum, medial superior frontal gyrus were increased after the social navigation task. These alterations were related to social cognition of tracking location in the social navigation. Moreover, participants who had more social support or less neuroticism showed a greater increase in hippocampal connectivity. These findings may highlight a more important role of the posterior hippocampal circuit in the social navigation, which is crucial for social cognition.





Similar content being viewed by others
Data availability
The datasets generated and analyzed in the current study are available from the corresponding author upon reasonable request.
References
Albert NB, Robertson EM, Miall RC (2009) The resting human brain and motor learning. Curr Biol 19(12):1023–1027. https://doi.org/10.1016/j.cub.2009.04.028
Aronov D, Nevers R, Tank DW (2017) Mapping of a non-spatial dimension by the hippocampal-entorhinal circuit. Nature 543(7647):719–722. https://doi.org/10.1038/nature21692
Barra J, Marquer A, Joassin R, Reymond C, Metge L, Chauvineau V, Perennou D (2010) Humans use internal models to construct and update a sense of verticality. Brain 133(Pt 12):3552–3563. https://doi.org/10.1093/brain/awq311
Basyouni R, Parkinson C (2022) Mapping the social landscape: tracking patterns of interpersonal relationships. Trends Cogn Sci 26(3):204–221. https://doi.org/10.1016/j.tics.2021.12.006
Bellmund JLS, Gardenfors P, Moser EI, Doeller CF (2018) Navigating cognition: spatial codes for human thinking. Science. https://doi.org/10.1126/science.aat6766
Berkes P, Orbán G, Lengyel M, Fiser J (2011) Spontaneous cortical activity reveals hallmarks of an optimal internal model of the environment. Science 331(6013):83–87. https://doi.org/10.1126/science.1195870
Biswal B, Yetkin FZ, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34(4):537–541. https://doi.org/10.1002/mrm.1910340409
Bourdieu P (2018) Social space and the genesis of appropriated physical space. Int J Urban Reg Res 42(1):106–114. https://doi.org/10.1111/1468-2427.12534
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129(Pt 3):564–583. https://doi.org/10.1093/brain/awl004
Chen PA, Jolly E, Cheong JH, Chang LJ (2020) Intersubject representational similarity analysis reveals individual variations in affective experience when watching erotic movies. Neuroimage 216:116851. https://doi.org/10.1016/j.neuroimage.2020.116851
Cohen S, Mermelstein R, Kamarck T, Hoberman HM (1985) Measuring the functional components of social support. In: Sarason IG, Sarason BR (eds) Social support: theory, research, and applications, pp 73–94. https://doi.org/10.1007/978-94-009-5115-0_5
Cohen S, Hoberman HM (1983) Positive events and social supports as buffers of life change stress. J Appl Soc Psychol 13:99–125. https://doi.org/10.1111/j.1559-1816.1983.tb02325.x
Cole MW, Ito T, Bassett DS, Schultz DH (2016) Activity flow over resting-state networks shapes cognitive task activations. Nat Neurosci 19(12):1718–1726. https://doi.org/10.1038/nn.4406
Cona G, Scarpazza C (2019) Where is the “where” in the brain? A meta-analysis of neuroimaging studies on spatial cognition. Hum Brain Mapp 40(6):1867–1886. https://doi.org/10.1002/hbm.24496
Doeller CF, King JA, Burgess N (2008) Parallel striatal and hippocampal systems for landmarks and boundaries in spatial memory. Proc Natl Acad Sci U S A 105(15):5915–5920. https://doi.org/10.1073/pnas.0801489105
Du M, Parkinson C (2021) Navigating social knowledge. Nat Neurosci 24(9):1195–1197. https://doi.org/10.1038/s41593-021-00893-7
Duarte IC, Castelhano J, Sales F, Castelo-Branco M (2016) The anterior versus posterior hippocampal oscillations debate in human spatial navigation: evidence from an electrocorticographic case study. Brain Behav 6(9):e00507. https://doi.org/10.1002/brb3.507
Dunbar RIM (2018) The anatomy of friendship. Trends Cogn Sci 22(1):32–51. https://doi.org/10.1016/j.tics.2017.10.004
Eichenbaum H (2015) The hippocampus as a cognitive map of social space. Neuron 87(1):9–11. https://doi.org/10.1016/j.neuron.2015.06.013
Fan L, Li H, Zhuo J, Zhang Y, Wang J, Chen L et al (2016) The human brainnetome atlas: a new brain atlas based on connectional architecture. Cereb Cortex 26(8):3508–3526. https://doi.org/10.1093/cercor/bhw157
Fox MD, Raichle ME (2007) Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci 8(9):700–711. https://doi.org/10.1038/nrn2201
Friston KJ (2011) Functional and effective connectivity: a review. Brain Connect 1(1):13–36. https://doi.org/10.1089/brain.2011.0008
Friston KJ, Williams S, Howard R, Frackowiak RS, Turner R (1996) Movement-related effects in fMRI time-series. Magn Reson Med 35(3):346–355. https://doi.org/10.1002/mrm.1910350312
Gilmore AW, Nelson SM, McDermott KB (2015) A parietal memory network revealed by multiple MRI methods. Trends Cogn Sci 19(9):534–543. https://doi.org/10.1016/j.tics.2015.07.004
Goldfarb EV, Chun MM, Phelps EA (2016) Memory-guided attention: independent contributions of the hippocampus and striatum. Neuron 89(2):317–324. https://doi.org/10.1016/j.neuron.2015.12.014
Goldfarb EV, Rosenberg MD, Seo D, Constable RT, Sinha R (2020) Hippocampal seed connectome-based modeling predicts the feeling of stress. Nat Commun 11(1):2650. https://doi.org/10.1038/s41467-020-16492-2
Hao X, Huang T, Song Y, Kong X, Liu J (2021) Development of navigation network revealed by resting-state and task-state functional connectivity. Neuroimage 243:118515. https://doi.org/10.1016/j.neuroimage.2021.118515
Hutchison RM, Womelsdorf T, Allen EA (2013) Dynamic functional connectivity: promise, issues, and interpretations. Neuroimage 80:360–378. https://doi.org/10.1016/j.neuroimage.2013.05.079
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17(2):825–841. https://doi.org/10.1006/nimg.2002.1132
Kraus BT, Perez D, Ladwig Z, Seitzman BA, Dworetsky A, Petersen SE, Gratton C (2021) Network variants are similar between task and rest states. Neuroimage 229:117743. https://doi.org/10.1016/j.neuroimage.2021.117743
Li J, Zhang R, Liu S, Liang Q, Zheng S, He X, Huang R (2021a) Human spatial navigation: neural representations of spatial scales and reference frames obtained from an ALE meta-analysis. Neuroimage 238:118264. https://doi.org/10.1016/j.neuroimage.2021.118264
Li M, Ma Q, Baetens K, Pu M, Deroost N, Baeken C et al (2021b) Social cerebellum in goal-directed navigation. Soc Neurosci 16(5):467–485. https://doi.org/10.1080/17470919.2021.1970017
Liao W, Wu GR, Xu Q, Ji GJ, Zhang Z, Zang YF, Lu G (2014) DynamicBC: a MATLAB toolbox for dynamic brain connectome analysis. Brain Connect 4(10):780–790. https://doi.org/10.1089/brain.2014.0253
Mackey AP, Miller Singley AT, Bunge SA (2013) Intensive reasoning training alters patterns of brain connectivity at rest. J Neurosci 33(11):4796–4803. https://doi.org/10.1523/JNEUROSCI.4141-12.2013
Maguire EA, Gadian DG, Johnsrude IS, Good CD, Ashburner J, Frackowiak RS, Frith CD (2000) Navigation-related structural change in the hippocampi of taxi drivers. Proc Natl Acad Sci U S A 97(8):4398–4403. https://doi.org/10.1073/pnas.070039597
McDermott KB, Gilmore AW, Nelson SM, Watson JM, Ojemann JG (2017) The parietal memory network activates similarly for true and associative false recognition elicited via the DRM procedure. Cortex 87:96–107. https://doi.org/10.1016/j.cortex.2016.09.008
Minderer M, Brown KD, Harvey CD (2019) The spatial structure of neural encoding in mouse posterior cortex during navigation. Neuron 102(1):232-248 e211. https://doi.org/10.1016/j.neuron.2019.01.029
Moser EI, Moser MB, McNaughton BL (2017) Spatial representation in the hippocampal formation: a history. Nat Neurosci 20(11):1448–1464. https://doi.org/10.1038/nn.4653
O’Keefe J, Nadel L (1979) The hippocampus as a cognitive map. Behav Brain Sci 2(4):487–494. https://doi.org/10.1017/s0140525x00063949
Omer DB, Maimon SR, Las L, Ulanovsky N (2018) Social place-cells in the bat hippocampus. Science 359(6372):218–224. https://doi.org/10.1126/science.aao3474
Park SA, Miller DS, Nili H, Ranganath C, Boorman ED (2020) Map making: constructing, combining, and inferring on abstract cognitive maps. Neuron 107(6):1226-1238 e1228. https://doi.org/10.1016/j.neuron.2020.06.030
Park SA, Miller DS, Boorman ED (2021) Inferences on a multidimensional social hierarchy use a grid-like code. Nat Neurosci 24(9):1292–1301. https://doi.org/10.1038/s41593-021-00916-3
Pezzulo G, Zorzi M, Corbetta M (2021) The secret life of predictive brains: what’s spontaneous activity for? Trends Cogn Sci 25(9):730–743. https://doi.org/10.1016/j.tics.2021.05.007
Poon CS, Merfeld DM (2005) Internal models: the state of the art. J Neural Eng. https://doi.org/10.1088/1741-2552/2/3/E01
Poppenk J, Evensmoen HR, Moscovitch M, Nadel L (2013) Long-axis specialization of the human hippocampus. Trends Cogn Sci 17(5):230–240. https://doi.org/10.1016/j.tics.2013.03.005
Power JD, Fair DA, Schlaggar BL, Petersen SE (2010) The development of human functional brain networks. Neuron 67(5):735–748. https://doi.org/10.1016/j.neuron.2010.08.017
Raichle ME, Mintun MA (2006) Brain work and brain imaging. Annu Rev Neurosci 29:449–476. https://doi.org/10.1146/annurev.neuro.29.051605.112819
Rolls ET, Wirth S (2018) Spatial representations in the primate hippocampus, and their functions in memory and navigation. Prog Neurobiol 171:90–113. https://doi.org/10.1016/j.pneurobio.2018.09.004
Rolls ET, Joliot M, Tzourio-Mazoyer N (2015) Implementation of a new parcellation of the orbitofrontal cortex in the automated anatomical labeling atlas. Neuroimage 122:1–5. https://doi.org/10.1016/j.neuroimage.2015.07.075
Rubinstein DY, Camarillo-Rodriguez L, Serruya MD, Herweg NA, Waldman ZJ, Wanda PA et al (2021) Contribution of left supramarginal and angular gyri to episodic memory encoding: an intracranial EEG study. Neuroimage 225:117514. https://doi.org/10.1016/j.neuroimage.2020.117514
Schafer M, Schiller D (2018) Navigating social space. Neuron 100(2):476–489. https://doi.org/10.1016/j.neuron.2018.10.006
Schuck NW, Niv Y (2019) Sequential replay of nonspatial task states in the human hippocampus. Science 364(6447):eaaw5181. https://doi.org/10.1126/science.aaw5181
Shirer WR, Ryali S, Rykhlevskaia E, Menon V, Greicius MD (2012) Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb Cortex 22(1):158–165. https://doi.org/10.1093/cercor/bhr099
Snyder AZ (2015) Intrinsic brain activity and resting state networks. 1–52. https://doi.org/10.1007/978-1-4614-6434-1_133-1
Solomon EA, Lega BC, Sperling MR, Kahana MJ (2019) Hippocampal theta codes for distances in semantic and temporal spaces. Proc Natl Acad Sci 116(48):24343–24352. https://doi.org/10.1073/pnas.1906729116
Spagna A, Hajhajate D, Liu J, Bartolomeo P (2021) Visual mental imagery engages the left fusiform gyrus, but not the early visual cortex: a meta-analysis of neuroimaging evidence. Neurosci Biobehav Rev 122:201–217. https://doi.org/10.1016/j.neubiorev.2020.12.029
Spaniol J, Davidson PS, Kim AS, Han H, Moscovitch M, Grady CL (2009) Event-related fMRI studies of episodic encoding and retrieval: meta-analyses using activation likelihood estimation. Neuropsychologia 47(8–9):1765–1779. https://doi.org/10.1016/j.neuropsychologia.2009.02.028
Tavares RM, Mendelsohn A, Grossman Y, Williams CH, Shapiro M, Trope Y, Schiller D (2015) A map for social navigation in the human brain. Neuron 87(1):231–243. https://doi.org/10.1016/j.neuron.2015.06.011
Tavor I, Parker Jones O, Mars RB, Smith SM, Behrens TE, Jbabdi S (2016) Task-free MRI predicts individual differences in brain activity during task performance. Science 352(6282):216–220. https://doi.org/10.1126/science.aad8127
Theves S, Fernandez G, Doeller CF (2019) The hippocampus encodes distances in multidimensional feature space. Curr Biol 29(7):1226–1231. https://doi.org/10.1016/j.cub.2019.02.035
Theves S, Fernandez G, Doeller CF (2020) The hippocampus maps concept space, not feature space. J Neurosci 40(38):7318–7325. https://doi.org/10.1523/JNEUROSCI.0494-20.2020
Tian L, Li Q, Wang C, Yu J (2018) Changes in dynamic functional connections with aging. Neuroimage 172:31–39. https://doi.org/10.1016/j.neuroimage.2018.01.040
Tolman E (1948) Cognitive maps in rats and men. Psychol Rev 55(4):189–208. https://doi.org/10.1037/h0061626
Tompson SH, Kahn AE, Falk EB, Vettel JM, Bassett DS (2020) Functional brain network architecture supporting the learning of social networks in humans. Neuroimage 210:116498. https://doi.org/10.1016/j.neuroimage.2019.116498
Vigano S, Piazza M (2021) The hippocampal-entorhinal system represents nested hierarchical relations between words during concept learning. Hippocampus 31(6):557–568. https://doi.org/10.1002/hipo.23320
Wang M, Dai X (2011) Development of the Chinese Big Five Personality Inventory (CBF-PI): psychometric properties of CBF-PI brief version. Chin J Clin Psychol 19(4):454–457. https://doi.org/10.16128/j.cnki.1005-3611.2011.04.004
Wang C, Ong JL, Patanaik A, Zhou J, Chee MW (2016) Spontaneous eyelid closures link vigilance fluctuation with fMRI dynamic connectivity states. Proc Natl Acad Sci U S A 113(34):9653–9658. https://doi.org/10.1073/pnas.1523980113
Wang M, Huang J, Liu M, Zhang D (2021) Modeling dynamic characteristics of brain functional connectivity networks using resting-state functional MRI. Med Image Anal 71:102063. https://doi.org/10.1016/j.media.2021.102063
Weiner KS, Zilles K (2016) The anatomical and functional specialization of the fusiform gyrus. Neuropsychologia 83:48–62. https://doi.org/10.1016/j.neuropsychologia.2015.06.033
Wolpert DM, Ghahramani Z, Jordan MI (1995) An internal model for sensorimotor integration. Science 269(5232):1880–1882. https://doi.org/10.1126/science.7569931
Wu X, Yu W, Hu H, Su Y, Liang Z, Bai Z et al (2021) Dynamic network topological properties for classifying primary dysmenorrhoea in the pain-free phase. Eur J Pain 25(9):1912–1924. https://doi.org/10.1002/ejp.1808
Yan CG, Wang XD, Zuo XN, Zang YF (2016) DPABI: data processing & analysis for (resting-state) brain imaging. Neuroinformatics 14(3):339–351. https://doi.org/10.1007/s12021-016-9299-4
Ye Z, Shi L, Li A, Chen C, Xue G (2020) Retrieval practice facilitates memory updating by enhancing and differentiating medial prefrontal cortex representations. Elife. https://doi.org/10.7554/eLife.57023
Zhang X, Wang MC, He L, Jie L, Deng J (2019) The development and psychometric evaluation of the Chinese Big Five Personality Inventory-15. PLoS ONE 14(8):e0221621. https://doi.org/10.1371/journal.pone.0221621
Zhang L, Chen P, Schafer M, Zheng S, Chen L, Wang S et al (2022) A specific brain network for a social map in the human brain. Sci Rep 12(1):1773. https://doi.org/10.1038/s41598-022-05601-4
Zheng A, Montez DF, Marek S, Gilmorec AW, Newbold DJ, Laumann TO, Kay BP (2021) Parallel hippocampal-parietal circuits for self- and goal-oriented processing. Proc Natl Acad Sci U S A 118(34):e2101743118. https://doi.org/10.1073/pnas.2101743118
Funding
The study was supported by grants from the National Natural Science Foundation of China (Grant numbers: 82171914 and 81871338), Guangdong Natural Science Foundation (2022A1515011022), and the National Key Research and Development Program of China (2018YFC1705006).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Experimental material preparation and data collection were performed by XW, LZ, BL, JL and YQ. XW conducted data analysis and wrote the first draft of the manuscript. RH contributed to guidance, funding acquisition. All authors commented on previous versions of the manuscript, read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of South China Normal University (Ethics number: 255).
Consent to participant
Informed consent was obtained from all individual participants included in the study.
Consent to publish
No individual person’s data was included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, X., Zhang, L., Liu, B. et al. Social navigation modulates the anterior and posterior hippocampal circuits in the resting brain. Brain Struct Funct 228, 799–813 (2023). https://doi.org/10.1007/s00429-023-02622-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-023-02622-1