Abstract
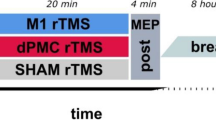
Implicit motor learning and memory involve complex cortical and subcortical networks. The induction of plasticity in these network components via non-invasive brain stimulation, including transcranial direct current stimulation (tDCS), has shown to improve motor learning. However, studies showing these effects are mostly restricted to stimulation of the primary motor cortex (M1) during the early stage of learning. Because of this, we aimed to explore the efficacy of anodal tDCS applied over the posterior parietal cortex (PPC), which is involved in memory processes, on serial reaction time task (SRTT) performance. Specifically, to evaluate the involvement of both motor learning network components, we compared the effects of tDCS applied over regions corresponding to M1 and PPC during the early and late stages of learning. The results revealed a selective improvement of reaction time (RT) during anodal stimulation over the PPC in the late stage of learning. These findings support the assumption that the PPC is relevant during specific phases of learning, at least for SRTT performance. The results also indicate that not only the target area (i.e., PPC), but also timing is crucial for achieving the effects of stimulation on motor learning.





Similar content being viewed by others
Data availability
Data and material are available under request.
References
Agboada D, Mosayebi Samani M, Jamil A et al (2019) Expanding the parameter space of anodal transcranial direct current stimulation of the primary motor cortex. Sci Rep. https://doi.org/10.1038/s41598-019-54621-0
Alam M, Truong DQ, Khadka N, Bikson M (2016) Spatial and polarity precision of concentric high-definition transcranial direct current stimulation (HD-tDCS). Phys Med Biol. https://doi.org/10.1088/0031-9155/61/12/4506
Allman C, Amadi U, Winkler AM et al (2016) Ipsilesional anodal tDCS enhances the functional benefits of rehabilitation in patients after stroke. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aad5651
Ammann C, Spampinato D, Márquez-Ruiz J (2016) Modulating motor learning through transcranial direct-current stimulation: an integrative view. Front Psychol. https://doi.org/10.3389/fpsyg.2016.01981
Antal A, Alekseichuk I, Bikson M et al (2017) Low intensity transcranial electric stimulation: safety, ethical, legal regulatory and application guidelines. Clin Neurophysiol 128:1774–1809
Berlot E, Popp NJ, Diedrichsen J (2020) A critical re-evaluation of fMRI signatures of motor sequence learning. bioRxiv. https://doi.org/10.1101/2020.01.08.899229
Bikson M, Grossman P, Thomas C, Zannou AL, Jiang J, Adnan T, Mourdoukoutas AP, Kronberg G, Truong D, Boggio P, Brunoni AR, Charvet L, Fregni F, Fritsch B, Gillick B, Hamilton RH, Hampstead BM, Jankord R, Kirton A, Knotkova H, Liebetanz D, Liu A, Loo C, Nitsche MA, Reis J, Richardson JD, Rotenberg A, Turkeltaub PE, Woods AJ (2016) Safety of transcranial direct current stimulation: evidence based update 2016. Brain Stimul 9(5):641–661. https://doi.org/10.1016/j.brs.2016.06.004
Boros K, Poreisz C, Münchau A et al (2008) Premotor transcranial direct current stimulation (tDCS) affects primary motor excitability in humans. Eur J Neurosci. https://doi.org/10.1111/j.1460-9568.2008.06090.x
Brennan S, McLoughlin DM, O’Connell R et al (2017) Anodal transcranial direct current stimulation of the left dorsolateral prefrontal cortex enhances emotion recognition in depressed patients and controls. J Clin Exp Neuropsychol 39:384–395. https://doi.org/10.1080/13803395.2016.1230595
Brodt S, Gais S, Beck J et al (2018) Fast track to the neocortex: a memory engram in the posterior parietal cortex. Science 362:1045–1048. https://doi.org/10.1126/science.aau2528
Brunoni AR, Nitsche MA, Bolognini N, Bikson M, Wagner T, Merabet L, Edwards DJ, Valero-Cabre A, RotenbergA Pascual-Leone A, Ferrucci R, Priori A, Boggio PS, Fregni F (2012) Clinical research with transcranial direct current stimulation (tDCS): challenges and future directions. Brain Stimul 5(3):175–195. https://doi.org/10.1016/j.brs.2011.03.002
Catalan MJ, Honda M, Weeks RA et al (1998) The functional neuroanatomy of simple and complex sequential finger movements: a PET study. Brain 121:253–264. https://doi.org/10.1093/brain/121.2.253
Chen J, McCulloch A, Kim H et al (2020) Application of anodal tDCS at primary motor cortex immediately after practice of a motor sequence does not improve offline gain. Exp Brain Res. https://doi.org/10.1007/s00221-019-05697-7
Ciaramelli E, Moscovitch M (2020) The space for memory in posterior parietal cortex: re-analyses of bottom-up attention data. Neuropsychologia. https://doi.org/10.1016/j.neuropsychologia.2020.107551
Ciaramelli E, Grady CL, Moscovitch M (2008) Top-down and bottom-up attention to memory: a hypothesis (AtoM) on the role of the posterior parietal cortex in memory retrieval. Neuropsychologia 46:1828–1851
Clark D, Ivry RB (2010) Multiple systems for motor skill learning. Wiley Interdiscip Rev Cogn Sci 1:461–467. https://doi.org/10.1002/wcs.56
Cuypers K, Leenus DJF, van den Berg FE et al (2013) Is motor learning mediated by tDCS intensity? PLoS ONE. https://doi.org/10.1371/journal.pone.0067344
de Xivry JJO, Marko MK, Pekny SE et al (2011) Stimulation of the human motor cortex alters generalization patterns of motor learning. J Neurosci. https://doi.org/10.1523/JNEUROSCI.0273-11.2011
Deiber MP, Wise SP, Honda M et al (1997) Frontal and parietal networks for conditional motor learning: a positron emission tomography study. J Neurophysiol. https://doi.org/10.1152/jn.1997.78.2.977
Della-Maggiore V, Villalta JI, Kovacevic N, McIntosh AR (2017) Functional evidence for memory stabilization in sensorimotor adaptation: a 24-h resting-state fmri study. Cereb Cortex 27:1748–1757. https://doi.org/10.1093/cercor/bhv289
Dudai Y, Karni A, Born J (2015) The consolidation and transformation of memory. Neuron 88:20–32
Esmaeilpour Z, Marangolo P, Hampstead BM et al (2018) Incomplete evidence that increasing current intensity of tDCS boosts outcomes. Brain Stimul. https://doi.org/10.1016/j.brs.2017.12.002
Evans C, Bachmann C, Lee JSA et al (2020) Dose-controlled tDCS reduces electric field intensity variability at a cortical target site. Brain Stimul. https://doi.org/10.1016/j.brs.2019.10.004
Focke J, Kemmet S, Krause V et al (2017) Cathodal transcranial direct current stimulation (tDCS) applied to the left premotor cortex (PMC) stabilizes a newly learned motor sequence. Behav Brain Res 316:87–93. https://doi.org/10.1016/j.bbr.2016.08.032
Foerster Á, Yavari F, Farnad L et al (2019) Effects of electrode angle-orientation on the impact of transcranial direct current stimulation on motor cortex excitability. Brain Stimul. https://doi.org/10.1016/j.brs.2018.10.014
Fonteneau C, Mondino M, Arns M et al (2019) Sham tDCS: A hidden source of variability? Reflections for further blinded, controlled trials. Encephale 45:S76. https://doi.org/10.1016/j.encep.2019.04.033
Fregni F, Nitsche MA, Loo CK, Brunoni AR, Marangolo P, Leite J, Carvalho S, Bolognini N, Caumo W, Paik NJ, Simis M, Ueda K, Ekhitari H, Luu P, Tucker DM, Tyler WJ, Brunelin J, Datta A, Juan CH, Venkatasubramanian G, Boggio PS, Bikson M (2015) Regulatory considerations for the clinical and research use of transcranial direct current stimulation (tDCS): review and recommendations from an expert panel. Clin Res Regul Aff 32(1):22–35. https://doi.org/10.3109/10601333.2015.980944
Ghasemian-Shirvan E, Farnad L, Mosayebi-Samani M et al (2020) Age-related differences of motor cortex plasticity in adults: a transcranial direct current stimulation study. Brain Stimul 13:1588–1599. https://doi.org/10.1016/j.brs.2020.09.004
Giordano J, Bikson M, Kappenman ES, Clark VP, Coslett HB, Hamblin MR, Hamilton R, Jankord R, Kozumbo WJ, McKinley RA, Nitsche MA, Reilly JP, Richardson J, Wurzman R, Calabrese E (2017) Mechanisms and effects of transcranial direct current stimulation. Dose Response 15(1):1559325816685467. https://doi.org/10.1177/1559325816685467
Gonzalez CC, Burke MR (2018) Motor sequence learning in the brain: the long and short of it. Neuroscience 389:85–98. https://doi.org/10.1016/j.neuroscience.2018.01.061
Gonzalez CC, Billington J, Burke MR (2016) The involvement of the fronto-parietal brain network in oculomotor sequence learning using fMRI. Neuropsychologia. https://doi.org/10.1016/j.neuropsychologia.2016.04.021
Grafton ST, Hazeltine E, Ivry R (1995) Functional mapping of sequence learning in normal humans. J Cogn Neurosci. https://doi.org/10.1162/jocn.1995.7.4.497
Grafton ST, Hazeltine E, Ivry RB (1998) Abstract and effector-specific representations of motor sequences identified with pet. J Neurosci. https://doi.org/10.1523/jneurosci.18-22-09420.1998
Grafton ST, Hazeltine E, Ivry RB (2002) Motor sequence learning with the nondominant left hand: a PET functional imaging study. Exp Brain Res. https://doi.org/10.1007/s00221-002-1181-y
Hamano YH, Sugawara SK, Yoshimoto T, Sadato N (2020) The motor engram as a dynamic change of the cortical network during early sequence learning: an fMRI study. Neurosci Res 153:27–39. https://doi.org/10.1016/j.neures.2019.03.004
Hashemirad F, Zoghi M, Fitzgerald PB, Jaberzadeh S (2016) The effect of anodal transcranial direct current stimulation on motor sequence learning in healthy individuals: a systematic review and meta-analysis. Brain Cogn. https://doi.org/10.1016/j.bandc.2015.11.005
Hashemirad F, Fitzgerald PB, Zoghi M, Jaberzadeh S (2017) Single-session anodal tDCS with small-size stimulating electrodes over frontoparietal superficial sites does not affect motor sequence learning. Front Hum Neurosci. https://doi.org/10.3389/fnhum.2017.00153
Herwig U, Satrapi P, Schönfeldt-Lecuona C (2003) Using the international 10–20 EEG system for positioning of transcranial magnetic stimulation. Brain Topogr 16(2):95–99. https://doi.org/10.1023/b:brat.0000006333.93597.9d
Himmer L, Schönauer M, Heib DPJ et al (2019) Rehearsal initiates systems memory consolidation, sleep makes it last. Sci Adv. https://doi.org/10.1126/sciadv.aav1695
Honda M, Deiber MP, Ibáñez V et al (1998) Dynamic cortical involvement in implicit and explicit motor sequence learning. A PET Study Brain 121:2159–2173. https://doi.org/10.1093/brain/121.11.2159
Hutchinson JB, Uncapher MR, Wagner AD (2009) Posterior parietal cortex and episodic retrieval: Convergent and divergent effects of attention and memory. Learn Mem 16:343–356
Jackson MP, Bikson M, Liebetanz D, Nitsche M (2017a) Toward comprehensive tDCS safety standards. Brain Behav Immun 66:413. https://doi.org/10.1016/j.bbi.2017.08.007
Jackson MP, Bikson M, Liebetanz D, Nitsche M (2017b) How to consider animal data in tDCS safety standards. Brain Stimul 10(6):1141–1142. https://doi.org/10.1016/j.brs.2017.08.004
Jongkees BJ, Immink MA, Boer OD et al (2019) The effect of cerebellar tDCS on sequential motor response selection. Cerebellum. https://doi.org/10.1007/s12311-019-01029-1
Kandel ER, Dudai Y, Mayford MR (2014) The molecular and systems biology of memory. Cell 157:163–186
Kantak SS, Mummidisetty CK, Stinear JW (2012) Primary motor and premotor cortex in implicit sequence learning—evidence for competition between implicit and explicit human motor memory systems. Eur J Neurosci 36:2710–2715. https://doi.org/10.1111/j.1460-9568.2012.08175.x
Karabanov A, Jin SH, Joutsen A et al (2012) Timing-dependent modulation of the posterior parietal cortex-primary motor cortex pathway by sensorimotor training. J Neurophysiol. https://doi.org/10.1152/jn.01049.2011
Klem GH, Lüders HO, Jasper HH, Elger C (1999) The ten-twenty electrode system of the International Federation. The International Federation of Clinical Neurophysiology. Electroencephalogr Clin Neurophysiol Suppl 52:3–6. https://doi.org/10.1016/0013-4694(58)90053-1
Klinzing JG, Niethard N, Born J (2019) Mechanisms of systems memory consolidation during sleep. Nat Neurosci 22(10):1598–1610. https://doi.org/10.1038/s41593-019-0467-3
Koch G, Rothwell JC (2009) TMS investigations into the task-dependent functional interplay between human posterior parietal and motor cortex. Behav Brain Res 202(2):147–152. https://doi.org/10.1016/j.bbr.2009.03.023
Kumar N, Manning TF, Ostry DJ (2019) Somatosensory cortex participates in the consolidation of human motor memory. PLoS Biol. https://doi.org/10.1371/journal.pbio.3000469
Kumar A, Panthi G, Divakar R, Mutha PK (2020) Mechanistic determinants of effector-independent motor memory encoding. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.2001179117
Kuo MF, Unger M, Liebetanz D et al (2008) Limited impact of homeostatic plasticity on motor learning in humans. Neuropsychologia. https://doi.org/10.1016/j.neuropsychologia.2008.02.023
Lin C, Ho J, Chiang MC, Wu AD et al (2012) Enhanced motor learning in older adults is accompanied by increased bilateral frontal and fronto-parietal connectivity. Brain Connect. https://doi.org/10.1089/brain.2011.0059
Maquet P, Laureys S, Peigneux P et al (2000) Experience-dependent changes in changes in cerebral activation during human REM sleep. Nat Neurosci. https://doi.org/10.1038/77744
Miller KE, Gehrman PR (2019) REM sleep: what is it good for? Curr Biol 29(16):R806–R807. https://doi.org/10.1016/j.cub.2019.06.074
Neri F, Mencarelli L, Menardi A et al (2020) A novel tDCS sham approach based on model-driven controlled shunting. Brain Stimul 13:507–516. https://doi.org/10.1016/j.brs.2019.11.004
Nissen MJ, Bullemer P (1987) Attentional requirements of learning: evidence from performance measures. Cogn Psychol. https://doi.org/10.1016/0010-0285(87)90002-8
Nitsche MA, Bikson M (2017) Extending the parameter range for tDCS: safety and tolerability of 4 mA stimulation. Brain Stimul 10(3):541–542. https://doi.org/10.1016/j.brs.2017.03.002
Nitsche MA, Paulus W (2000a) Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol 527:633–639. https://doi.org/10.1111/j.1469-7793.2000.t01-1-00633.x
Nitsche MA, Paulus W (2000b) Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. https://doi.org/10.1111/j.1469-7793.2000.t01-1-00633.x
Nitsche MA, Paulus W (2001) Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology 57:1899–1901. https://doi.org/10.1212/WNL.57.10.1899
Nitsche MA, Liebetanz D, Antal A et al (2003a) Chapter 27 modulation of cortical excitability by weak direct current stimulation—technical, safety and functional aspects. Suppl Clin Neurophysiol 56:255–276. https://doi.org/10.1016/S1567-424X(09)70230-2
Nitsche MA, Schauenburg A, Lang N et al (2003b) Facilitation of implicit motor learning by weak transcranial direct current stimulation of the primary motor cortex in the human. J Cogn Neurosci. https://doi.org/10.1162/089892903321662994
Nitsche MA, Seeber A, Frommann K et al (2005) Modulating parameters of excitability during and after transcranial direct current stimulation of the human motor cortex. J Physiol. https://doi.org/10.1113/jphysiol.2005.092429
Nitsche MA, Doemkes S, Karakose T et al (2007) Shaping the effects of transcranial direct current stimulation of the human motor cortex shaping the effects of transcranial direct current stimulation of the human motor cortex. J Neurophysiol 97:3109–3117. https://doi.org/10.1152/jn.01312.2006
Nitsche MA, Jakoubkova M, Thirugnanasambandam N et al (2010) Contribution of the premotor cortex to consolidation of motor sequence learning in humans during sleep. J Neurophysiol. https://doi.org/10.1152/jn.00611.2010
O’Shea J, Boudrias MH, Stagg CJ et al (2014) Predicting behavioural response to TDCS in chronic motor stroke. Neuroimage. https://doi.org/10.1016/j.neuroimage.2013.05.096
Parvizi J, Wagner AD (2018) Memory, numbers, and action decision in human posterior parietal cortex. Neuron 97:7–10
Pollok B, Keitel A, Foerster M et al (2020) The posterior parietal cortex mediates early offline-rather than online-motor sequence learning. Neuropsychologia. https://doi.org/10.1016/j.neuropsychologia.2020.107555
Pollok B, Schmitz-Justen C, Krause V (2021) Cathodal transcranial direct current stimulation (Tdcs) applied to the left premotor cortex interferes with explicit reproduction of a motor sequence. Brain Sci 11:1–18. https://doi.org/10.3390/brainsci11020207
Qin Y-L, Mcnaughton BL, Skaggs WE, Barnes CA (1997) Memory reprocessing in corticocortical and hippocampocortical neuronal ensembles. Phil Transact R Soc B 352:1525–1533
Reis J, Fritsch B (2011) Modulation of motor performance and motor learning by transcranial direct current stimulation. Curr Opin Neurol 24(6):590–596. https://doi.org/10.1097/WCO.0b013e32834c3db0
Reis J, Schambra HM, Cohen LG et al (2009) Noninvasive cortical stimulation enhances motor skill acquisition over multiple days through an effect on consolidation. Proc Natl Acad Sci USA 106:1590–1595. https://doi.org/10.1073/pnas.0805413106
Rioult-Pedotti MS, Friedman D, Donoghue JP (2000) Learning-induced LTP in neocortex. Science 290:533–536. https://doi.org/10.1126/science.290.5491.533
Rivera-Urbina GN, Batsikadze G, Molero-Chamizo A et al (2015) Parietal transcranial direct current stimulation modulates primary motor cortex excitability. Eur J Neurosci. https://doi.org/10.1111/ejn.12840
Sakurada T, Hirai M, Watanabe E (2019) Individual optimal attentional strategy during implicit motor learning boosts frontoparietal neural processing efficiency: a functional near-infrared spectroscopy study. Brain Behav. https://doi.org/10.1002/brb3.1183
Sami S, Robertson EM, Chris Miall R (2014) The time course of task-specific memory consolidation effects in resting state networks. J Neurosci. https://doi.org/10.1523/JNEUROSCI.4341-13.2014
Santos Ferreira I, Teixeira Costa B, Lima Ramos C, Lucena P, Thibaut A, Fregni F (2019) Searching for the optimal tDCS target for motor rehabilitation. J Neuroeng Rehabil 16(1):90. https://doi.org/10.1186/s12984-019-0561-5
Saturnino GB, Antunes A, Thielscher A (2015) On the importance of electrode parameters for shaping electric field patterns generated by tDCS. Neuroimage 120:25–35. https://doi.org/10.1016/j.neuroimage.2015.06.067
Saturnino GB, Siebner HR, Thielscher A, Madsen KH (2019) Accessibility of cortical regions to focal TES: dependence on spatial position, safety, and practical constraints. Neuroimage. https://doi.org/10.1016/j.neuroimage.2019.116183
Shadmehr R, Holcomb HH (1997) Neural correlates of motor memory consolidation. Science 277:821–825. https://doi.org/10.1126/science.277.5327.821
Stagg CJ, Jayaram G, Pastor D et al (2011) Polarity and timing-dependent effects of transcranial direct current stimulation in explicit motor learning. Neuropsychologia. https://doi.org/10.1016/j.neuropsychologia.2011.02.009
Stagg CJ, Bachtiar V, O’Shea J et al (2012) Cortical activation changes underlying stimulation-induced behavioural gains in chronic stroke. Brain. https://doi.org/10.1093/brain/awr313
Vignaud P, Mondino M, Poulet E et al (2018) Duration but not intensity influences transcranial direct current stimulation (tDCS) after-effects on cortical excitability. Neurophysiol Clin. https://doi.org/10.1016/j.neucli.2018.02.001
Wagner AD, Shannon BJ, Kahn I, Buckner RL (2005) Parietal lobe contributions to episodic memory retrieval. Trends Cogn Sci 9:445–453
Wang Y, Zhou H, Li Y, Liu W (2018) Impact of electrode number on the performance of high-definition transcranial direct current stimulation (HD-tDCS). In: Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBS
Wang S, Itthipuripat S, Ku Y (2019) Electrical stimulation over human posterior parietal cortex selectively enhances the capacity of visual short-term memory. J Neurosci 39:528–536. https://doi.org/10.1523/JNEUROSCI.1959-18.2018
Wexler A (2015) A pragmatic analysis of the regulation of consumer transcranial direct current stimulation (TDCS) devices in the United States. J Law Biosci 2:669–696. https://doi.org/10.1093/jlb/lsv039
Woods AJ, Antal A, Bikson M et al (2016) A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin Neurophysiol 127:1031–1048
Zanatta MS, Quillfeldt JH, Schaeffer E et al (1997) Involvement of the hippocampus, amygdala, entorhinal cortex and posterior parietal cortex in memory consolidation. Brazilian J Med Biol Res 30:235–240. https://doi.org/10.1590/S0100-879X1997000200012
Acknowledgements
The authors thank doctors G. Yáñez Téllez and J.R. Alameda for their respective contributions in recruiting participants and designing the task.
Funding
Michael A. Nitsche receives support by the EC Horizon 2020 Program, FET Grant, 686764-LUMINOUS, grants from the German Ministry of Research and Education (GCBS grant 01EE1403C, TRAINSTIM grant 01GQ1424E), and is member of the scientific advisory boards of Neuroelectrics, and NeuroDevice. The other authors declare that they have no conflict of interest. This research did not receive any specific grant from funding agencies in the public, commercial, or non-for-profit sectors. G. Nathzidy Rivera-Urbina receives support from the Mexican Government, Grant UABC-PTC-610 DSA/103.5/16/1024 (PRODEP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Michael A. Nitsche is a member of the scientific advisory boards of Neuroelectrics and NeuroDevice. The other authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Ethics Committee of the University of Huelva (05/2014) and conforms to the principles of the World Medical Association Declaration of Helsinki.
Consent to participate
All the subjects signed informed consent to participate in the study and were informed of all the procedure.
Consent for publication
All the participants signed informed consent for the publication of research data, excluding personal identification data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rivera-Urbina, G.N., Molero-Chamizo, A. & Nitsche, M.A. Discernible effects of tDCS over the primary motor and posterior parietal cortex on different stages of motor learning. Brain Struct Funct 227, 1115–1131 (2022). https://doi.org/10.1007/s00429-021-02451-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02451-0