Abstract
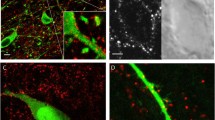
G-protein-coupled D2 autoreceptors expressed on dopamine neurons (D2Rs) inhibit transmitter release and cell firing at axonal endings and somatodendritic compartments. Mechanistic details of somatodendritic dopamine release remain unresolved, partly due to insufficient information on the subcellular distribution of D2Rs. Previous studies localizing D2Rs have been hindered by a dearth of antibodies validated for specificity in D2R knockout animals and have been limited by the small sampling areas imaged by electron microscopy. This study utilized sub-diffraction fluorescence microscopy and electron microscopy to examine D2 receptors in a superecliptic pHlourin GFP (SEP) epitope-tagged D2 receptor knockin mouse. Incubating live slices with an anti-SEP antibody achieved the selective labeling of plasma membrane-associated receptors for immunofluorescent imaging over a large area of the substantia nigra pars compacta (SNc). SEP-D2Rs appeared as puncta-like structures along the surface of dendrites and soma of dopamine neurons visualized by antibodies to tyrosine hydroxylase (TH). TH-associated SEP-D2Rs displayed a cell surface density of 0.66 puncta/µm2, which corresponds to an average frequency of 1 punctum every 1.50 µm. Separate ultrastructural experiments using silver-enhanced immunogold revealed that membrane-bound particles represented 28% of total D2Rs in putative dopamine cells within the SNc. Structures immediately adjacent to dendritic membrane gold particles were unmyelinated axons or axon varicosities (40%), astrocytes (19%), other dendrites (7%), or profiles unidentified (34%) in single sections. Some apposed profiles also expressed D2Rs. Fluorescent and ultrastructural analyses also provided the first visualization of membrane D2Rs at the axon initial segment, a compartment critical for action potential generation. The punctate appearance of anti-SEP staining indicates there is a population of D2Rs organized in discrete signaling sites along the plasma membrane, and for the first time, a quantitative estimate of spatial frequency is provided.







Similar content being viewed by others
Availability of data material
The datasets generated and/or analyzed during the current study are available from the corresponding author at reasonable request.
Code availability
Not applicable.
References
Bal A, Bachelot T, Savasta M, Manier M, Verna JM, Benabid AL, Feuerstein C (1994) Evidence for dopamine D2 receptor mRNA expression by striatal astrocytes in culture: in situ hybridization and polymerase chain reaction studies. Mol Brain Res 23:204–212. https://doi.org/10.1016/0169-328X(94)90227-5
Bayer VE, Pickel VM (1990) Ultrastructural localization of tyrosine hydroxylase in the rat ventral tegmental area: relationship between immunolabeling density and neuronal associations. J Neurosci 10:2996–3013. https://doi.org/10.1523/JNEUROSCI.10-09-02996.1990
Beaulieu J-M, Gainetdinov RR (2011) The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol Rev 63:182–217. https://doi.org/10.1124/pr.110.002642
Beckstead MJ, Grandy DK, Wickman K, Williams JT (2004) Vesicular dopamine release elicits an inhibitory postsynaptic current in midbrain dopamine neurons. Neuron 42:939–946. https://doi.org/10.1016/j.neuron.2004.05.019
Beeler JA, Dreyer JK (2019) Synchronicity: the role of midbrain dopamine in whole-brain coordination. eNeuro. https://doi.org/10.1523/ENEURO.0345-18.2019
Bender KJ, Ford CP, Trussell LO (2010) Dopaminergic modulation of axon initial segment calcium channels regulates action potential initiation. Neuron 68:500–511. https://doi.org/10.1016/j.neuron.2010.09.026
Bender KJ, Uebele VN, Renger JJ, Trussell LO (2012) Control of firing patterns through modulation of axon initial segment T-type calcium channels. J Physiol 590:109–118. https://doi.org/10.1113/jphysiol.2011.218768
Berghs S, Aggujaro D, Dirkx R, Maksimova E, Stabach P, Hermel J-M, Zhang J-P, Philbrick W, Slepnev V, Ort T, Solimena M (2000) βIV spectrin, a new spectrin localized at axon initial segments and nodes of Ranvier in the central and peripheral nervous system. J Cell Biol 151:985–1002
Björklund A, Dunnett SB (2007) Dopamine neuron systems in the brain: an update. Trends Neurosci Fifty Years Dopamine Res 30:194–202. https://doi.org/10.1016/j.tins.2007.03.006
Blythe SN, Wokosin D, Atherton JF, Bevan MD (2009) Cellular mechanisms underlying burst firing in substantia nigra dopamine neurons. J Neurosci 29:15531–15541. https://doi.org/10.1523/JNEUROSCI.2961-09.2009
Carlin RK, Grab DJ, Cohen RS, Siekevitz P (1980) Isolation and characterization of postsynaptic densities from various brain regions: enrichment of different types of postsynaptic densities. J Cell Biol 86:831–845. https://doi.org/10.1083/jcb.86.3.831
Condon AF, Robinson BG, Asad N, Dore TM, Tian L, Williams JT (2021) The residence of synaptically released dopamine on D2 autoreceptors. Cell Rep. https://doi.org/10.1016/j.celrep.2021.109465
Courtney NA, Ford CP (2014) The timing of dopamine- and noradrenaline-mediated transmission reflects underlying differences in the extent of spillover and pooling. J Neurosci 34:7645–7656. https://doi.org/10.1523/JNEUROSCI.0166-14.2014
Ford CP (2014) The role of D2-autoreceptors in regulating dopamine neuron activity and transmission. Neurosci Ventral Tegmentum Dopamine New Wave Divers 282:13–22. https://doi.org/10.1016/j.neuroscience.2014.01.025
Ford CP, Phillips PEM, Williams JT (2009) The time course of dopamine transmission in the ventral tegmental area. J Neurosci 29:13344–13352. https://doi.org/10.1523/JNEUROSCI.3546-09.2009
Ford CP, Gantz SC, Phillips PEM, Williams JT (2010) Control of extracellular dopamine at dendrite and axon terminals. J Neurosci 30:6975–6983. https://doi.org/10.1523/JNEUROSCI.1020-10.2010
Gantz SC, Bunzow JR, Williams JT (2013) Spontaneous inhibitory synaptic currents mediated by a G protein-coupled receptor. Neuron 78:807–812. https://doi.org/10.1016/j.neuron.2013.04.013
Gantz SC, Robinson BG, Buck DC, Bunzow JR, Neve RL, Williams JT, Neve KA (2015) Distinct regulation of dopamine D2S and D2L autoreceptor signaling by calcium. Elife 4:e09358. https://doi.org/10.7554/eLife.09358
Gantz SC, Ford CP, Morikawa H, Williams JT (2018) The evolving understanding of dopamine neurons in the substantia nigra and ventral tegmental area. Annu Rev Physiol 80:219–241. https://doi.org/10.1146/annurev-physiol-021317-121615
Garzón M, Duffy AM, Chan J, Lynch M-K, Mackie K, Pickel VM (2013) Dopamine D2 and acetylcholine α7 nicotinic receptors have subcellular distributions favoring mediation of convergent signaling in the mouse ventral tegmental area. Neuroscience 252:126–143. https://doi.org/10.1016/j.neuroscience.2013.08.008
Geffen LB, Jessell TM, Cuello AC, Iversen LL (1976) Release of dopamine from dendrites in rat substantia nigra. Nature 260:258–260. https://doi.org/10.1038/260258a0
Gentet LJ, Williams SR (2007) Dopamine gates action potential backpropagation in midbrain dopaminergic neurons. J Neurosci 27:1892–1901. https://doi.org/10.1523/JNEUROSCI.5234-06.2007
Giguère N, Delignat-Lavaud B, Herborg F, Voisin A, Li Y, Jacquemet V, Anand-Srivastava M, Gether U, Giros B, Trudeau L-É (2019) Increased vulnerability of nigral dopamine neurons after expansion of their axonal arborization size through D2 dopamine receptor conditional knockout. PLOS Genet 15:e1008352. https://doi.org/10.1371/journal.pgen.1008352
González-Cabrera C, Meza R, Ulloa L, Merino-Sepúlveda P, Luco V, Sanhueza A, Oñate-Ponce A, Bolam JP, Henny P (2017) Characterization of the axon initial segment of mice substantia nigra dopaminergic neurons. J Comp Neurol 525:3529–3542. https://doi.org/10.1002/cne.24288
Grace AA, Bunney BS (1983a) Intracellular and extracellular electrophysiology of nigral dopaminergic neurons—1. Identification and Characterization. Neuroscience 10:301–315. https://doi.org/10.1016/0306-4522(83)90135-5
Grace AA, Bunney BS (1983b) Intracellular and extracellular electrophysiology of nigral dopaminergic neurons—2. Action potential generating mechanisms and morphological correlates. Neuroscience 10:317–331. https://doi.org/10.1016/0306-4522(83)90136-7
Groves PM, Linder JC (1983) Dendro-dendritic synapses in substantia nigra: descriptions based on analysis of serial sections. Exp Brain Res 49:209–217. https://doi.org/10.1007/BF00238581
Hage TA, Khaliq ZM (2015) Tonic firing rate controls dendritic Ca2+ signaling and synaptic gain in substantia nigra dopamine neurons. J Neurosci 35:5823–5836. https://doi.org/10.1523/JNEUROSCI.3904-14.2015
Harterink M, Vocking K, Pan X, Jerez EMS, Slenders L, Fréal A, Tas RP, van de Wetering WJ, Timmer K, Motshagen J, van Beuningen SFB, Kapitein LC, Geerts WJC, Post JA, Hoogenraad CC (2019) TRIM46 organizes microtubule fasciculation in the axon initial segment. J Neurosci 39:4864–4873. https://doi.org/10.1523/JNEUROSCI.3105-18.2019
Hedstrom KL, Ogawa Y, Rasband MN (2008) AnkyrinG is required for maintenance of the axon initial segment and neuronal polarity. J Cell Biol 183:635–640. https://doi.org/10.1083/jcb.200806112
Hikima T, Lee CR, Witkovsky P, Chesler J, Ichtchenko K, Rice ME (2021) Activity-dependent somatodendritic dopamine release in the substantia nigra autoinhibits the releasing neuron. Cell Rep. https://doi.org/10.1016/j.celrep.2021.108951
Hrabetova S, Cognet L, Rusakov DA, Nägerl UV (2018) Unveiling the extracellular space of the brain: from super-resolved microstructure to in vivo function. J Neurosci 38:9355–9363. https://doi.org/10.1523/JNEUROSCI.1664-18.2018
Khan ZU, Koulen P, Rubinstein M, Grandy DK, Goldman-Rakic PS (2001) An astroglia-linked dopamine D2-receptor action in prefrontal cortex. Proc Natl Acad Sci 98:1964–1969. https://doi.org/10.1073/pnas.98.4.1964
Kim O-J, Ariano MA, Namkung Y, Marinec P, Kim E, Han J, Sibley DR (2008) D2 dopamine receptor expression and trafficking is regulated through direct interactions with ZIP. J Neurochem 106:83–95. https://doi.org/10.1111/j.1471-4159.2008.05348.x
King AN, Manning CF, Trimmer JS (2014) A unique ion channel clustering domain on the axon initial segment of mammalian neurons. J Comp Neurol 522:2594–2608. https://doi.org/10.1002/cne.23551
Kinney JP, Spacek J, Bartol TM, Bajaj CL, Harris KM, Sejnowski TJ (2013) Extracellular sheets and tunnels modulate glutamate diffusion in hippocampal neuropil. J Comp Neurol 521:448–464. https://doi.org/10.1002/cne.23181
Koga E, Momiyama T (2000) Presynaptic dopamine D2-like receptors inhibit excitatory transmission onto rat ventral tegmental dopaminergic neurones. J Physiol 523:163–173. https://doi.org/10.1111/j.1469-7793.2000.t01-2-00163.x
Labouèbe G, Lomazzi M, Cruz HG, Creton C, Luján R, Li M, Yanagawa Y, Obata K, Watanabe M, Wickman K, Boyer SB, Slesinger PA, Lüscher C (2007) RGS2 modulates coupling between GABAB receptors and GIRK channels in dopamine neurons of the ventral tegmental area. Nat Neurosci 10:1559–1568. https://doi.org/10.1038/nn2006
Lacas-Gervais S, Guo J, Strenzke N, Scarfone E, Kolpe M, Jahkel M, De Camilli P, Moser T, Rasband MN, Solimena M (2004) βIVΣ1 spectrin stabilizes the nodes of Ranvier and axon initial segments. J Cell Biol 166:983–990. https://doi.org/10.1083/jcb.200408007
Le Bras B, Fréal A, Czarnecki A, Legendre P, Bullier E, Komada M, Brophy PJ, Davenne M, Couraud F (2014) In vivo assembly of the axon initial segment in motor neurons. Brain Struct Funct 219:1433–1450. https://doi.org/10.1007/s00429-013-0578-7
Leterrier C (2018) The axon initial segment: an updated viewpoint. J Neurosci 38:2135–2145. https://doi.org/10.1523/JNEUROSCI.1922-17.2018
Liu C, Kershberg L, Wang J, Schneeberger S, Kaeser PS (2018) Dopamine secretion is mediated by sparse active zone-like release sites. Cell 172:706-718.e15. https://doi.org/10.1016/j.cell.2018.01.008
Lu X-Y, Behnam Ghasemzadeh M, Kalivas PW (1997) Expression of D1 receptor, D2 receptor, substance P and enkephalin messenger RNAs in the neurons projecting from the nucleus accumbens. Neuroscience 82:767–780. https://doi.org/10.1016/S0306-4522(97)00327-8
Lysakowski A, Gaboyard-Niay S, Calin-Jageman I, Chatlani S, Price SD, Eatock RA (2011) Molecular microdomains in a sensory terminal, the vestibular calyx ending. J Neurosci 31:10101–10114. https://doi.org/10.1523/JNEUROSCI.0521-11.2011
Mathiisen TM, Nagelhus EA, Jouleh B, Torp R, Frydenlund DS, Mylonakou M-N, Amiry-Moghaddam M, Covolan L, Utvik JK, Riber B, Gujord KM, Knutsen J, Skare Ø, Laake P, Davanger S, Haug F-M, Rinvik E, Ottersen OP (2006) Postembedding immunogold cytochemistry of membrane molecules and amino acid transmitters in the central nervous system. In: Zaborszky L, Wouterlood FG, Lanciego JL (eds) Neuroanatomical tract-tracing 3: molecules, neurons, and systems. Springer US, Boston, pp 72–108. https://doi.org/10.1007/0-387-28942-9_3
Mengual E, Pickel VM (2002) Ultrastructural immunocytochemical localization of the dopamine D2 receptor and tyrosine hydroxylase in the rat ventral pallidum. Synapse 43:151–162. https://doi.org/10.1002/syn.10033
Moubarak E, Engel D, Dufour MA, Tapia M, Tell F, Goaillard J-M (2019) Robustness to axon initial segment variation is explained by somatodendritic excitability in rat substantia nigra dopaminergic neurons. J Neurosci 39:5044–5063. https://doi.org/10.1523/JNEUROSCI.2781-18.2019
Peters A, Palay SL, de F. Webster H (1991) Fine structures of the nervous system: neurons and their supporting cells. Oxford University Press, Oxford
Pickel VM, Chan J, Nirenberg MJ (2002) Region-specific targeting of dopamine D2-receptors and somatodendritic vesicular monoamine transporter 2 (VMAT2) within ventral tegmental area subdivisions. Synapse 45:113–124. https://doi.org/10.1002/syn.10092
Poulin J-F, Gaertner Z, Moreno-Ramos OA, Awatramani R (2020) Classification of midbrain dopamine neurons using single-cell gene expression profiling approaches. Trends Neurosci. https://doi.org/10.1016/j.tins.2020.01.004
Prou D, Gu W-J, Le Crom S, Vincent J-D, Salamero J, Vernier P (2001) Intracellular retention of the two isoforms of the D2 dopamine receptor promotes endoplasmic reticulum disruption. J Cell Sci 114:3517–3527. https://doi.org/10.1242/jcs.114.19.3517
Robinson BG, Bunzow JR, Grimm JB, Lavis LD, Dudman JT, Brown J, Neve KA, Williams JT (2017) Desensitized D2 autoreceptors are resistant to trafficking. Sci Rep. https://doi.org/10.1038/s41598-017-04728-z
Robinson BG, Cai X, Wang J, Bunzow JR, Williams JT, Kaeser PS (2019) RIM is essential for stimulated but not spontaneous somatodendritic dopamine release in the midbrain. Elife 8:e47972. https://doi.org/10.7554/eLife.47972
Roeper J (2013) Dissecting the diversity of midbrain dopamine neurons. Trends Neurosci 36:336–342. https://doi.org/10.1016/j.tins.2013.03.003
Schneider Gasser EM, Straub CJ, Panzanelli P, Weinmann O, Sassoè-Pognetto M, Fritschy J-M (2006) Immunofluorescence in brain sections: simultaneous detection of presynaptic and postsynaptic proteins in identified neurons. Nat Protoc 1:1887–1897. https://doi.org/10.1038/nprot.2006.265
Sesack SR, Aoki C, Pickel VM (1994) Ultrastructural localization of D2 receptor-like immunoreactivity in midbrain dopamine neurons and their striatal targets. J Neurosci 14:88–106. https://doi.org/10.1523/JNEUROSCI.14-01-00088.1994
Sesack SR, Miner LH, Omelchenko N (2006) Preembedding immunoelectron microscopy: applications for studies of the nervous system. In: Zaborszky L, Wouterlood FG, Lanciego JL (eds) Neuroanatomical tract-tracing 3. Springer US, Boston, pp 6–71. https://doi.org/10.1007/0-387-28942-9_2
Sherpa AD, Xiao F, Joseph N, Aoki C, Hrabetova S (2016) Activation of β-adrenergic receptors in rat visual cortex expands astrocytic processes and reduces extracellular space volume. Synapse 70:307–316. https://doi.org/10.1002/syn.21908
Spacek J, Harris KM (1997) Three-dimensional organization of smooth endoplasmic reticulum in hippocampal CA1 dendrites and dendritic spines of the immature and mature rat. J Neurosci 17:190–203. https://doi.org/10.1523/JNEUROSCI.17-01-00190.1997
Takeuchi Y, Fukunaga K (2003) Differential subcellular localization of two dopamine D2 receptor isoforms in transfected NG108-15 cells. J Neurochem 85:1064–1074. https://doi.org/10.1046/j.1471-4159.2003.01763.x
Torres GE (2006) The dopamine transporter proteome. J Neurochem 97:3–10. https://doi.org/10.1111/j.1471-4159.2006.03719.x
van der Weijden MCM, Rodriguez-Contreras D, Delnooz CCS, Robinson BG, Condon AF, Kielhold ML, Stormezand GN, Ma KY, Dufke C, Williams JT, Neve KA, Tijssen MAJ, Verbeek DS (2020) A gain-of-function variant in dopamine D2 receptor and progressive chorea and dystonia phenotype. Mov Disord. https://doi.org/10.1002/mds.28385
Wang H, Pickel VM (2002) Dopamine D2 receptors are present in prefrontal cortical afferents and their targets in patches of the rat caudate-putamen nucleus. J Comp Neurol 442:392–404. https://doi.org/10.1002/cne.10086
Wassef M, Berod A, Sotelo C (1981) Dopaminergic dendrites in the pars reticulata of the rat substantia nigra and their striatal input. Combined immunocytochemical localization of tyrosine hydroxylase and anterograde degeneration. Neuroscience 6:2125–2139. https://doi.org/10.1016/0306-4522(81)90003-8
Wilson CJ, Groves PM, Fifková E (1977) Monoaminergic synapses, including dendro-dendritic synapses in the rat substantia nigra. Exp Brain Res 30:161–174. https://doi.org/10.1007/BF00237248
Xin W, Schuebel KE, Jair K, Cimbro R, De Biase LM, Goldman D, Bonci A (2019) Ventral midbrain astrocytes display unique physiological features and sensitivity to dopamine D2 receptor signaling. Neuropsychopharmacology 44:344–355. https://doi.org/10.1038/s41386-018-0151-4
Yang S, Ben-Shalom R, Ahn M, Liptak AT, van Rijn RM, Whistler JL, Bender KJ (2016) β-Arrestin-dependent dopaminergic regulation of calcium channel activity in the axon initial segment. Cell Rep 16:1518–1526. https://doi.org/10.1016/j.celrep.2016.06.098
Yee AG, Forbes B, Cheung P-Y, Martini A, Burrell MH, Freestone PS, Lipski J (2019) Action potential and calcium dependence of tonic somatodendritic dopamine release in the Substantia Nigra pars compacta. J Neurochem 148:462–479. https://doi.org/10.1111/jnc.14587
Yuan A, Rao MV, Veeranna, Nixon RA (2012) Neurofilaments at a glance. J Cell Sci 125:3257–3263. https://doi.org/10.1242/jcs.104729
Yung KKL, Bolam JP, Smith AD, Hersch SM, Ciliax BJ, Levey AI (1995) Immunocytochemical localization of D1 and D2 dopamine receptors in the basal ganglia of the rat: light and electron microscopy. Neuroscience 65:709–730. https://doi.org/10.1016/0306-4522(94)00536-E
Acknowledgements
This work was supported by National Institutes of Health Grants DA004523 and DA007262 (Williams), and and P30 NS061800 (Aicher). The authors gratefully acknowledge the generous provision of beta IV-spectrin antibody by Dr. Matthew Rasband from the Baylor College of Medicine and the kind gift of ankyrin-G antibody from Paul Jenkins at the University of Michigan. We acknowledge expert technical assistance by staff in the Advanced Light Microscopy Core, supported by Grant P30NS061800.
Funding
This study was funded by National Institute on Drug Abuse grant nos. DA004523 and DA007262 (Williams), and National Institutes of Health grant no. P30 NS061800 (Aicher).
Author information
Authors and Affiliations
Contributions
JRB created the superecliptic pHluorin-tagged D2 receptor knockin mouse. JRB, SRS, and JTW designed the studies. JJL, JRB, BR, and SDLT performed and analyzed the fluorescent imaging experiments. MT, JJBP, SH, SA, and SRS performed and analyzed the electron microscopy experiments. The paper was written by JJL and MT with editorial contributions from JRB, SA, SRS, and JTW. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors report a conflict of interest. One author (SRS) acknowledges being a Co-Editor-In-Chief of this journal.
Ethics approval
All animal procedures and protocols were conducted in accordance with the National Institute of Health Guidelines for the Care and Use of Laboratory Animals. They also complied with the appropriate guidelines set forth by the Institutional Animal Care and Use Committees at Oregon Health & Science University and the University of Pittsburgh.
Consent to participate
Not applicable.
Consent to publication
All authors acknowledge participation in the study and development of the manuscript. All authors acknowledge reading the submitted form of this paper and consent to its submission for publication at this time.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lebowitz, J.J., Trinkle, M., Bunzow, J.R. et al. Subcellular localization of D2 receptors in the murine substantia nigra. Brain Struct Funct 227, 925–941 (2022). https://doi.org/10.1007/s00429-021-02432-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02432-3