Abstract
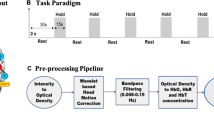
Cerebrovascular reactivity (CVR) is of great significance for the treatment and prevention of cerebrovascular diseases. CVR can be mapped using the blood oxygenation level-dependent (BOLD) signal of fMRI. Breath holding (BH) is a reliable method to produce the desired increase in arterial CO2, while its application in clinical research is limited due to subject’s compliance and variability. BH task with variable respiratory rates could allow more flexibility in clinical populations. In this study, 50 healthy volunteers were scanned for end-inspiration BH tasks with three different respiration rates. For the three respiratory rates BH tasks, the CVR was estimated based on the BOLD signal and general linear model (GLM) separately. Specifically, the extra time delay was considered for the hemodynamic response function, and the optimal delay was estimated for each voxel. To measure CVR in grey matter, BOLD signals of end-inspiration BH were used as regressors in general linear models to quantify their impact on CVR. This was performed for regions and voxels. Systematic differences were observed between the three end-inspiratory breathing rates. The greatest increase in activation intensity was found in fast breathing followed by self-paced and slow breathing. We conclude that the BH task of variable respiratory rates allows for CVR measurement, making breath-holding challenges more flexible and appropriate for routine practice.





Similar content being viewed by others
References
Abbott DF, Opdam HI, Briellmann RS, Jackson GD (2005) Brief breath holding may confound functional magnetic resonance imaging studies. Hum Brain Mapp 24(4):284–290. https://doi.org/10.1002/hbm.20086
Birn RM, Diamond JB, Smith MA, Bandettini PA (2006) Separating respiratory-variation-related fluctuations from neuronal-activity-related fluctuations in fMRI. Neuroimage 31(4):1536–1548. https://doi.org/10.1016/j.neuroimage.2006.02.048
Birn RM, Smith MA, Jones TB, Bandettini PA (2008) The respiration response function: the temporal dynamics of fMRI signal fluctuations related to changes in respiration. Neuroimage 40(2):644–654. https://doi.org/10.1016/j.neuroimage.2007.11.059
Bright MG, Murphy K (2013) Reliable quantification of BOLD fMRI cerebrovascular reactivity despite poor breath-hold performance. Neuroimage 83:559–568. https://doi.org/10.1016/j.neuroimage.2013.07.007
Buunk G, van der Hoeven JG, Meinders AE (1997) Cerebrovascular reactivity in comatose patients resuscitated from a cardiac arrest. Stroke 28(8):1569–1573. https://doi.org/10.1161/01.str.28.8.1569
Calhoun VD, Wager TD, Krishnan A, Rosch KS, Seymour KE, Nebel MB, Kiehl K (2017) The impact of T1 versus EPI spatial normalization templates for fMRI data analyses. Hum Brain Mapp 38(11):5331–5342. https://doi.org/10.1002/hbm.23737
Chang C, Thomason ME, Glover GH (2008) Mapping and correction of vascular hemodynamic latency in the BOLD signal. Neuroimage 43(1):90–102. https://doi.org/10.1016/j.neuroimage.2008.06.030
Chang TY, Liu HL, Lee TH, Kuan WC, Chang CH, Wu HC, Chang YJ (2009) Change in cerebral perfusion after carotid angioplasty with stenting is related to cerebral vasoreactivity: a study using dynamic susceptibility-weighted contrast-enhanced MR imaging and functional MR imaging with a breath-holding paradigm. AJNR Am J Neuroradiol 30(7):1330–1336. https://doi.org/10.3174/ajnr.A1589
Cohen ER, Ugurbil K, Kim SG (2002) Effect of basal conditions on the magnitude and dynamics of the blood oxygenation level-dependent fMRI response. J Cereb Blood Flow Metab 22(9):1042–1053. https://doi.org/10.1097/00004647-200209000-00002
Derdeyn CP, Videen TO, Yundt KD, Fritsch SM, Carpenter DA, Grubb RL, Powers WJ (2002) Variability of cerebral blood volume and oxygen extraction: stages of cerebral haemodynamic impairment revisited. Brain 125:595–607. https://doi.org/10.1093/brain/awf047
Di X, Kannurpatti SS, Rypma B, Biswal BB (2013) Calibrating BOLD fMRI activations with neurovascular and anatomical constraints. Cereb Cortex 23(2):255–263. https://doi.org/10.1093/cercor/bhs001
Friedman L, Turner JA, Stern H, Mathalon DH, Trondsen LC, Potkin SG (2008) Chronic smoking and the BOLD response to a visual activation task and a breath hold task in patients with schizophrenia and healthy controls. Neuroimage 40(3):1181–1194. https://doi.org/10.1016/j.neuroimage.2007.12.040
Glodzik L, Randall C, Rusinek H, de Leon MJ (2013) Cerebrovascular reactivity to carbon dioxide in Alzheimer’s disease. J Alzheimers Dis 35(3):427–440. https://doi.org/10.3233/JAD-122011
Hou X, Liu P, Li Y, Jiang D, De Vis JB, Lin Z, Lu H (2020) The association between BOLD-based cerebrovascular reactivity (CVR) and end-tidal CO2 in healthy subjects. Neuroimage 207:116365. https://doi.org/10.1016/j.neuroimage.2019.116365
Iadecola C, Davisson RL (2008) Hypertension and cerebrovascular dysfunction. Cell Metab 7(6):476–484. https://doi.org/10.1016/j.cmet.2008.03.010
Kannurpatti SS, Biswal BB, Hudetz AG (2002) Differential fMRI-BOLD signal response to apnea in humans and anesthetized rats. Magn Reson Med 47(5):864–870. https://doi.org/10.1002/mrm.10131
Kannurpatti SS, Rypma B, Biswal BB (2012) Prediction of task-related BOLD fMRI with amplitude signatures of resting-state fMRI. Front Syst Neurosci 6:7. https://doi.org/10.3389/fnsys.2012.00007
Kastrup A, Li TQ, Takahashi A, Glover GH, Moseley ME (1998) Functional magnetic resonance imaging of regional cerebral blood oxygenation changes during breath holding. Stroke 29(12):2641–2645. https://doi.org/10.1161/01.str.29.12.2641
Kastrup A, Kruger G, Neumann-Haefelin T, Moseley ME (2001) Assessment of cerebrovascular reactivity with functional magnetic resonance imaging: comparison of CO(2) and breath holding. Magn Reson Imaging 19(1):13–20. https://doi.org/10.1016/s0730-725x(01)00227-2
Leoni RF, Paiva FF, Henning EC, Nascimento GC, Tannus A, de Araujo DB, Silva AC (2011) Magnetic resonance imaging quantification of regional cerebral blood flow and cerebrovascular reactivity to carbon dioxide in normotensive and hypertensive rats. Neuroimage 58(1):75–81. https://doi.org/10.1016/j.neuroimage.2011.06.030
Liu P, De Vis JB, Lu H (2019) Cerebrovascular reactivity (CVR) MRI with CO2 challenge: a technical review. Neuroimage 187:104–115. https://doi.org/10.1016/j.neuroimage.2018.03.047
Magon S, Basso G, Farace P, Ricciardi GK, Beltramello A, Sbarbati A (2009) Reproducibility of BOLD signal change induced by breath holding. Neuroimage 45(3):702–712. https://doi.org/10.1016/j.neuroimage.2008.12.059
Markus H, Cullinane M (2001) Severely impaired cerebrovascular reactivity predicts stroke and TIA risk in patients with carotid artery stenosis and occlusion. Brain 124(Pt 3):457–467. https://doi.org/10.1093/brain/124.3.457
Scouten A, Schwarzbauer C (2008) Paced respiration with end-expiration technique offers superior BOLD signal repeatability for breath-hold studies. Neuroimage 43(2):250–257. https://doi.org/10.1016/j.neuroimage.2008.03.052
Silvestrini M, Troisi E, Matteis M, Cupini LM, Caltagirone C (1996) Transcranial Doppler assessment of cerebrovascular reactivity in symptomatic and asymptomatic severe carotid stenosis. Stroke 27(11):1970–1973. https://doi.org/10.1161/01.str.27.11.1970
Sousa I, Vilela P, Figueiredo P (2014) Reproducibility of hypocapnic cerebrovascular reactivity measurements using BOLD fMRI in combination with a paced deep breathing task. Neuroimage 98:31–41. https://doi.org/10.1016/j.neuroimage.2014.04.049
Tancredi FB, Hoge RD (2013) Comparison of cerebral vascular reactivity measures obtained using breath-holding and CO2 inhalation. J Cereb Blood Flow Metab 33(7):1066–1074. https://doi.org/10.1038/jcbfm.2013.48
Thomason ME, Glover GH (2008) Controlled inspiration depth reduces variance in breath-holding-induced BOLD signal. Neuroimage 39(1):206–214. https://doi.org/10.1016/j.neuroimage.2007.08.014
Thomason ME, Burrows BE, Gabrieli JD, Glover GH (2005) Breath holding reveals differences in fMRI BOLD signal in children and adults. Neuroimage 25(3):824–837. https://doi.org/10.1016/j.neuroimage.2004.12.026
Tong Y, Bergethon PR, Frederick BD (2011) An improved method for mapping cerebrovascular reserve using concurrent fMRI and near-infrared spectroscopy with Regressor Interpolation at Progressive Time Delays (RIPTiDe). Neuroimage 56(4):2047–2057. https://doi.org/10.1016/j.neuroimage.2011.03.071
Webster MW, Makaroun MS, Steed DL, Smith HA, Johnson DW, Yonas H (1995) Compromised cerebral blood flow reactivity is a predictor of stroke in patients with symptomatic carotid artery occlusive disease. J Vasc Surg 21(2):338–344. https://doi.org/10.1016/s0741-5214(95)70274-1
Ziyeh S, Rick J, Reinhard M, Hetzel A, Mader I, Speck O (2005) Blood oxygen level-dependent MRI of cerebral CO2 reactivity in severe carotid stenosis and occlusion. Stroke 36(4):751–756. https://doi.org/10.1161/01.STR.0000157593.03470.3d
Acknowledgements
We thank Tom Trifon, Katherine Ji and Donna Chen for comments on this manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC) grant numbers 61871420.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared that there are no conflicts of interest in relation to the subject of this study.
Research involving human and/or animal participants
50 healthy adult volunteers from the University of Electronic Science and Technology of China and the surrounding community participated in the study. All subjects gave informed written consent after the nature of the study and its possible consequences were explained to them. The study and protocols were in accordance with the latest Revision of the Declaration of Helsinki and received full ethical approval from the Institutional Review Board of the University of Electronic Science and Technology of China.
Informed consent
Informed consent was obtained in writing from all individual participants included in the study.
Data and code availability
The data that support the findings of this study will be made available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, K., Yang, H., Zhang, H. et al. Altered cerebrovascular reactivity due to respiratory rate and breath holding: a BOLD-fMRI study on healthy adults. Brain Struct Funct 226, 1229–1239 (2021). https://doi.org/10.1007/s00429-021-02236-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02236-5