Abstract
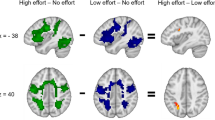
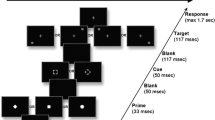
The default mode network (DMN) efficient deactivation and suppressed functional connectivity (FC) during goal-directed tasks, which require attentional resources, have been considered essential to healthy brain cognition. However, recent studies have shown that DMN regions do not always show the expected behavior. Then, we aimed to investigate the functional activation and connectivity of DMN nodes in young, healthy controls during a goal-directed task. We used an adaptation of the symbol digit modalities test (SDMT) to evaluate the information processing speed (IPS). Twenty-four subjects (10 women, age: 29 ± 7 years) underwent two functional Magnetic Resonance Imaging experiments: one during resting-state and one during a block-designed SDMT paradigm. We superimposed the templates of the DMN on the group activation map and observed the reorganization of the network. For the posterior cingulate cortex (PCC) node of the DMN, which is spatially extensive, comprising the precuneus (dorsal portion) and the posterior cingulate gyrus (PCG, ventral portion), the extent of each region was different between conditions, suggesting different functional roles for them. Therefore, for the functional connectivity (FC) analysis, we split the DMN-PCC region into two regions: left precuneus (BA 7) and PCG. The left precuneus (BA 7) was positively correlated with the left lingual gyrus (BA 17), a task-positive region, and negatively associated with the DMN nodes when comparing task performance with the resting-state condition. The other DMN regions presented the classical antagonistic role during the attentional task. In conclusion, we found that the activation and functional connectivity of the DMN is, in general, suppressed during the information processing. However, the left precuneus BA 7 presented a context-dependent modulatory behavior, working as a transient in-between hub connecting the DMN to task-positive areas. Such findings support studies that show increased activation and excitatory functional connectivity of DMN portions during goal-directed tasks. Moreover, our results may contribute to defining more precise functional correlates of IPS deficits in a wide range of clinical and neurological diseases.



Similar content being viewed by others
Code availability
The software and routines used in the present study are all available in their respective references.
Data availability
Data is not available due to patient confidentiality.
References
Anticevic A, Cole MW, Murray JD et al (2012) The role of default network deactivation in cognition and disease. Trends Cogn Sci 16:584–592
Ashburner J, Friston KJ (2005) Unified segmentation. Neuroimage. https://doi.org/10.1016/j.neuroimage.2005.02.018
Bae S, Hong JS, Kim SM, Han DH (2018) Bupropion shows different effects on brain functional connectivity in patients with internet-based gambling disorder and internet gaming disorder. Front Psychiatry. https://doi.org/10.3389/fpsyt.2018.00130
Bartova L, Meyer BM, Diers K et al (2015) Reduced default mode network suppression during a working memory task in remitted major depression. J Psychiatr Res. https://doi.org/10.1016/j.jpsychires.2015.02.025
Benedict RHB, Deluca J, Phillips G et al (2017) Validity of the symbol digit modalities test as a cognition performance outcome measure for multiple sclerosis. Mult Scler 23(5):721–733
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Castellanos FX, Margulies DS, Kelly C et al (2008) Cingulate-precuneus interactions: a new locus of dysfunction in adult attention-deficit/hyperactivity disorder. Biol Psychiatry. https://doi.org/10.1016/j.biopsych.2007.06.025
Cattaneo Z, Silvanto J, Pascual-Leone A, Battelli L (2009) The role of the angular gyrus in the modulation of visuospatial attention by the mental number line. Neuroimage. https://doi.org/10.1016/j.neuroimage.2008.09.003
Cavanna AE (2007) The precuneus and consciousness. CNS Spectr 12(7):545–552
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129(3):564–583
Chen G, Cox RW, Glen DR et al (2019) A tail of two sides: artificially doubled false positive rates in neuroimaging due to the sidedness choice with t-tests. Hum Brain Mapp. https://doi.org/10.1002/hbm.24399
Chen L, Fan X, Li H et al (2018) Topological reorganization of the default mode network in severe male obstructive sleep apnea. Front Neurol. https://doi.org/10.3389/fneur.2018.00363
Chiaravalloti ND, Stojanovic-Radic J, Deluca J (2013) The role of speed versus working memory in predicting learning new information in multiple sclerosis. J Clin Exp Neuropsychol 35:180–191. https://doi.org/10.1080/13803395.2012.760537
Coget A, Deverdun J, Bonafé A et al (2018) Transient immediate postoperative homotopic functional disconnectivity in low-grade glioma patients. NeuroImage Clin. https://doi.org/10.1016/j.nicl.2018.02.023
Cole MW, Ito T, Schultz D et al (2019) Task activations produce spurious but systematic inflation of task functional connectivity estimates. Neuroimage. https://doi.org/10.1016/j.neuroimage.2018.12.054
Corbetta M, Shulman GL (2002) Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci. https://doi.org/10.1038/nrn755
Costigan AG, Umla-Runge K, Evans CJ et al (2019) Neurochemical correlates of scene processing in the precuneus/posterior cingulate cortex: a multimodal fMRI and 1H-MRS study. Hum Brain Mapp. https://doi.org/10.1002/hbm.24566
Di X, Biswal BB (2015) Characterizations of resting-state modulatory interactions in the human brain. J Neurophysiol. https://doi.org/10.1152/jn.00893.2014
Elton A, Gao W (2015) Task-positive functional connectivity of the default mode network transcends task domain. J Cogn Neurosci. https://doi.org/10.1162/jocn_a_00859
Felmingham KL, Baguley IJ, Green AM (2004) Effects of diffuse axonal injury on speed of information processing following severe traumatic brain injury. Neuropsychology. https://doi.org/10.1037/0894-4105.18.3.564
Feys P, Lamers I, Francis G et al (2017) The nine-hole peg test as a manual dexterity performance measure for multiple sclerosis. Mult Scler J 23:711–720
Forn C, Ripollés P, Cruz-Gómez AJ et al (2013) Task-load manipulation in the symbol digit modalities test: an alternative measure of information processing speed. Brain Cogn 82:152–160. https://doi.org/10.1016/j.bandc.2013.04.003
Fox MD, Snyder AZ, Vincent JL et al (2005) The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.0504136102
Fransson P, Marrelec G (2008) The precuneus/posterior cingulate cortex plays a pivotal role in the default mode network: evidence from a partial correlation network analysis. Neuroimage. https://doi.org/10.1016/j.neuroimage.2008.05.059
Friston KJ, Frith CD, Frackowiak RSJ, Turner R (1995) Characterizing dynamic brain responses with fMRI: a multivariate approach. Neuroimage. https://doi.org/10.1006/nimg.1995.1019
Gao W, Lin W, Grewen K, Gilmore JH (2017) Functional connectivity of the infant human brain: plastic and modifiable. Neuroscientist 23(2):169–184
Geday J, Gjedde A (2009) Attention, emotion, and deactivation of default activity in inferior medial prefrontal cortex. Brain Cogn. https://doi.org/10.1016/j.bandc.2008.08.009
Gordon EM, Laumann TO, Adeyemo B et al (2016) Generation and evaluation of a cortical area parcellation from resting-state correlations. Cereb Cortex. https://doi.org/10.1093/cercor/bhu239
Haneef Z, Lenartowicz A, Yeh HJ et al (2014) Network analysis of the default mode network using functional connectivity MRI in temporal lobe epilepsy. J Vis Exp. https://doi.org/10.3791/51442
Haworth J, Phillips M, Newson M et al (2016) Measuring information processing speed in mild cognitive impairment: clinical versus research dichotomy. J Alzheimer’s Dis. https://doi.org/10.3233/JAD-150791
Hayden BY, Nair AC, McCoy AN, Platt ML (2008) Posterior cingulate cortex mediates outcome-contingent allocation of behavior. Neuron 60:19–25. https://doi.org/10.1016/j.neuron.2008.09.012
Hirnstein M, Bayer U, Ellison A, Hausmann M (2011) TMS over the left angular gyrus impairs the ability to discriminate left from right. Neuropsychologia. https://doi.org/10.1016/j.neuropsychologia.2010.10.028
Kim HJ, Cha J, Lee JM et al (2016) Distinctive resting state network disruptions among Alzheimer’s disease, subcortical vascular dementia, and mixed dementia patients. J Alzheimer’s Dis. https://doi.org/10.3233/JAD-150637
Knauff M, Fangmeier T, Ruff CC, Johnson-Laird PN (2003) Reasoning, models, and images: behavioral measures and cortical activity. J Cogn Neurosci. https://doi.org/10.1162/089892903321662949
Korgaonkar MS, Goldstein-Piekarski AN, Fornito A, Williams LM (2020) Intrinsic connectomes are a predictive biomarker of remission in major depressive disorder. Mol Psychiatry. https://doi.org/10.1038/s41380-019-0574-2
Laureys S, Goldman S, Phillips C et al (1999) Impaired effective cortical connectivity in vegetative state: preliminary investigation using PET. Neuroimage. https://doi.org/10.1006/nimg.1998.0414
Lee D, Lee J, Namkoong K, Jung YC (2018) Subregions of the anterior cingulate cortex form distinct functional connectivity patterns in young males with internet gaming disorder with comorbid depression. Front Psychiatry. https://doi.org/10.3389/fpsyt.2018.00380
Leech R, Kamourieh S, Beckmann CF, Sharp DJ (2011) Fractionating the default mode network: distinct contributions of the ventral and dorsal posterior cingulate cortex to cognitive control. J Neurosci. https://doi.org/10.1523/JNEUROSCI.5626-10.2011
Leech R, Sharp DJ (2014) The role of the posterior cingulate cortex in cognition and disease. Brain 137:12–32
Lemiere J, Decruyenaere M, Evers-Kiebooms G et al (2002) Longitudinal study evaluating neuropsychological changes in so-called asymptomatic carriers of the Huntington’s disease mutation after 1 year. Acta Neurol Scand. https://doi.org/10.1034/j.1600-0404.2002.01192.x
Liechti ME (2017) Modern clinical research on LSD. Neuropsychopharmacology 42:2114–2127
Lundstrom BN, Ingvar M, Petersson KM (2005) The role of precuneus and left inferior frontal cortex during source memory episodic retrieval. Neuroimage. https://doi.org/10.1016/j.neuroimage.2005.05.008
Luo Z, Zeng L-L, Qin J et al (2019) Functional parcellation of human brain precuneus using density-based clustering. Cereb Cortex 30:269–282. https://doi.org/10.1093/cercor/bhz086
Maddock RJ, Garrett AS, Buonocore MH (2001) Remembering familiar people: the posterior cingulate cortex and autobiographical memory retrieval. Neuroscience. https://doi.org/10.1016/S0306-4522(01)00108-7
Maddock RJ, Garrett AS, Buonocore MH (2003) Posterior cingulate cortex activation by emotional words: fMRI evidence from a valence decision task. Hum Brain Mapp. https://doi.org/10.1002/hbm.10075
Maldjian J (1994) WFU PickAtlas user manual v2. 4. Hum Brain Mapp 63:1–13
Margulies DS, Vincent JL, Kelly C et al (2009) Precuneus shares intrinsic functional architecture in humans and monkeys. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.0905314106
Mattiaccio LM, Coman IL, Thompson CA et al (2018) Frontal dysconnectivity in 22q11.2 deletion syndrome: an atlas-based functional connectivity analysis. Behav Brain Funct. https://doi.org/10.1186/s12993-018-0134-y
Müller F, Lenz C, Dolder P et al (2017) Increased thalamic resting-state connectivity as a core driver of LSD-induced hallucinations. Acta Psychiatr Scand. https://doi.org/10.1111/acps.12818
O’Brien CP (2008) The CAGE questionnaire for detection of alcoholism: a remarkably useful but simple tool. J Am Med Assoc 300:2054–2056
Pantoni L, Marzi C, Poggesi A et al (2019) Fractal dimension of cerebral white matter: a consistent feature for prediction of the cognitive performance in patients with small vessel disease and mild cognitive impairment. NeuroImage Clin. https://doi.org/10.1016/j.nicl.2019.101990
Pascoe M, Alamri Y, Dalrymple-Alford J et al (2018) The symbol-digit modalities test in mild cognitive impairment: evidence from Parkinson’s disease patients. Eur Neurol. https://doi.org/10.1159/000485669
Patel VP, Feinstein A (2019) The link between depression and performance on the symbol digit modalities test: mechanisms and clinical significance. Mult Scler J. https://doi.org/10.1177/1352458518770086
Peirce JW (2007) PsychoPy-psychophysics software in Python. J Neurosci Methods 162:8–13. https://doi.org/10.1016/j.jneumeth.2006.11.017
Power JD, Barnes KA, Snyder AZ et al (2012) Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage. https://doi.org/10.1016/j.neuroimage.2011.10.018
Power JD, Mitra A, Laumann TO et al (2014) Methods to detect, characterize, and remove motion artifact in resting state fMRI. Neuroimage. https://doi.org/10.1016/j.neuroimage.2013.08.048
Raichle ME, MacLeod AM, Snyder AZ et al (2001) A default mode of brain function. Proc Natl Acad Sci USA 98:676–682. https://doi.org/10.1073/pnas.98.2.676
Sheridan LK, Fitzgerald HE, Adams KM et al (2006) Normative symbol digit modalities test performance in a community-based sample. Arch Clin Neuropsychol 21:23–28. https://doi.org/10.1016/j.acn.2005.07.003
Shulman GL, Fiez JA, Corbetta M et al (1997) Common blood flow changes across visual tasks: II. Decreases in cerebral cortex. J Cogn Neurosci. https://doi.org/10.1162/jocn.1997.9.5.648
Silva PHR, Spedo CT, Baldassarini CR et al (2019) Brain functional and effective connectivity underlying the information processing speed assessed by the symbol digit modalities test. Neuroimage. https://doi.org/10.1016/j.neuroimage.2018.09.080
Silva PHR, Spedo CT, Barreira AA, Leoni RF (2018) Symbol digit modalities test adaptation for magnetic resonance imaging environment: a systematic review and meta-analysis. Mult Scler Relat Disord. https://doi.org/10.1016/j.msard.2018.01.014
Silva S, De Pasquale F, Vuillaume C et al (2015) Disruption of posteromedial large-scale neural communication predicts recovery from coma. Neurology. https://doi.org/10.1212/WNL.0000000000002196
Smith A (1968) The symbol-digit modalities test: a neuropsychologic test for economic screening of learning and other cerebral disorders. Learn Disord 3:83–91
Studer B, Cen D, Walsh V (2014) The angular gyrus and visuospatial attention in decision-making under risk. Neuroimage. https://doi.org/10.1016/j.neuroimage.2014.09.003
Suchan B, Yágüez L, Wunderlich G et al (2002) Hemispheric dissociation of visual-pattern processing and visual rotation. Behav Brain Res. https://doi.org/10.1016/S0166-4328(02)00204-8
Tanaka S, Kirino E (2019) Increased functional connectivity of the angular gyrus during imagined music performance. Front Hum Neurosci. https://doi.org/10.3389/fnhum.2019.00092
Thomas Yeo BT, Krienen FM, Sepulcre J et al (2011) The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. https://doi.org/10.1152/jn.00338.2011
Tzourio-Mazoyer N, Landeau B, Papathanassiou D et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. https://doi.org/10.1006/nimg.2001.0978
Utevsky AV, Smith DV, Huettel SA (2014) Precuneus is a functional core of the default-mode network. J Neurosci. https://doi.org/10.1523/JNEUROSCI.4227-13.2014
Van Schependom J, D’hooghe MB, Cleynhens K, et al (2014) The symbol digit modalities test as sentinel test for cognitive impairment in multiple sclerosis. Eur J Neurol. https://doi.org/10.1111/ene.12463
Vanhaudenhuyse A, Noirhomme Q, Tshibanda LJF et al (2010) Default network connectivity reflects the level of consciousness in non-communicative brain-damaged patients. Brain. https://doi.org/10.1093/brain/awp313
Vatansever D, Menon DK, Stamatakis EA (2017) Default mode contributions to automated information processing. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1710521114
Vigneau M, Beaucousin V, Hervé PY et al (2006) Meta-analyzing left hemisphere language areas: Phonology, semantics, and sentence processing. Neuroimage 30(4):1414–1432
Wang Z, Fei L, Sun Y et al (2019) The role of the precuneus and posterior cingulate cortex in the neural routes to action. Comput Assist Surg. https://doi.org/10.1080/24699322.2018.1557903
Wang Z, Liang P, Jia X et al (2012) The baseline and longitudinal changes of PCC connectivity in mild cognitive impairment: a combined structure and resting-state fMRI study. PLoS ONE. https://doi.org/10.1371/journal.pone.0036838
Whitfield-Gabrieli S, Nieto-Castanon A (2012) Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. https://doi.org/10.1089/brain.2012.0073
Yasuno F, Kazui H, Yamamoto A et al (2015) Resting-state synchrony between the retrosplenial cortex and anterior medial cortical structures relates to memory complaints in subjective cognitive impairment. Neurobiol Aging. https://doi.org/10.1016/j.neurobiolaging.2015.03.006
Zanto TP, Pa J, Gazzaley A (2014) Reliability measures of functional magnetic resonance imaging in a longitudinal evaluation of mild cognitive impairment. Neuroimage 84:443–452. https://doi.org/10.1016/j.neuroimage.2013.08.063
Zhang JY, Feinstein A (2016) Screening for cognitive impairments after traumatic brain injury: a comparison of a brief computerized battery with the montreal cognitive assessment. J Neuropsychiatry Clin Neurosci. https://doi.org/10.1176/appi.neuropsych.16010005
Zhang S, Li C, shan R, (2012) Functional connectivity mapping of the human precuneus by resting state fMRI. Neuroimage. https://doi.org/10.1016/j.neuroimage.2011.11.023
Zhou Y, Zeidman P, Wu S et al (2018) Altered intrinsic and extrinsic connectivity in schizophrenia. NeuroImage Clin. https://doi.org/10.1016/j.nicl.2017.12.006
Acknowledgements
The authors would like to acknowledge Dr. Carina Tellaroli Spedo for conducting the cognitive evaluation.
Funding
This work was supported by the Fundação de Apoio à Pesquisa do Estado de São Paulo (FAPESP), process number 2017/22212-0.
Author information
Authors and Affiliations
Contributions
PHRS contributed to study conception and design, image processing, analysis, and manuscript writing. CR contributed to discussion, manuscript writing, and revision. RFL contributed to study conception and design, analysis, and manuscript revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva, P.H.R., Rondinoni, C. & Leoni, R.F. Non-classical behavior of the default mode network regions during an information processing task. Brain Struct Funct 225, 2553–2562 (2020). https://doi.org/10.1007/s00429-020-02143-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-020-02143-1