Abstract
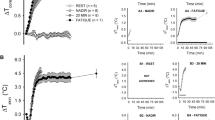
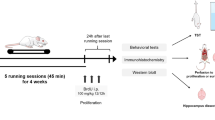
Acute and long-term exercise differentially affect brain functions. It has been suggested that neuronal activation is one of the mechanisms for exercise-induced enhancement of brain functions. However, the differential effects of acute and long-term exercise on the spatial and temporal profiles of neuronal activation in the brain have been scarcely explored. In this study, we profiled the expression of c-Fos, a marker of neuronal activation, in selected 26 brain regions of 2-month-old male C57/B6 mice that received either a single bout of treadmill running (acute exercise) or a 4-week treadmill training (long-term exercise) at the same duration (1 h/day) and intensity (10 m/min). The c-Fos expression was determined before, immediately after, and 2 h after the run. The results showed that acute exercise increased the densities of c-Fos+ cells in the ventral hippocampal CA1 region, followed by (in a high to low order) the primary somatosensory cortex, other hippocampal subregions, and striatum immediately after the run; significant changes remained evident in the hippocampal subregions after a 2-h rest. Long-term exercise increased the densities of c-Fos+ cells in the striatum, followed by the primary somatosensory, primary and secondary motor cortices, hippocampal subregions, hypothalamic nuclei, and lateral periaqueductal gray; significant changes remained evident in the striatum, hippocampal subregions, hypothalamic nuclei, and lateral periaqueductal gray after a 2-h rest. Interestingly, the densities of c-Fos+ cells in the substantia nigra and ventral tegmental area only increased after a 2-h rest after the run in the long-term exercise group. The densities of c-Fos+ cells were positively correlated with the expression of brain-derived neurotrophic factor in the selected brain regions. In conclusion, both acute and long-term treadmill running at mild intensity induce c-Fos expression in the limbic system and movement-associated cortical and subcortical regions, with long-term exercise involving more brain regions (i.e., hypothalamus and periaqueductal gray) and longer lasting effects.





Similar content being viewed by others
References
Adkins DL, Boychuk J, Remple MS (1985) Kleim JA (2006) Motor training induces experience-specific patterns of plasticity across motor cortex and spinal cord. J Appl Physiol 101(6):1776–1782. https://doi.org/10.1152/japplphysiol.00515.2006
Ahlskog JE, Geda YE, Graff-Radford NR, Petersen RC (2011) Physical exercise as a preventive or disease-modifying treatment of dementia and brain aging. Mayo Clin Proc 86(9):876–884. https://doi.org/10.4065/mcp.2011.0252
Black JE, Isaacs KR, Anderson BJ, Alcantara AA, Greenough WT (1990) Learning causes synaptogenesis, whereas motor activity causes angiogenesis, in cerebellar cortex of adult rats. Proc Natl Acad Sci U S A 87(14):5568–5572
Bolam JP, Hanley JJ, Booth PA, Bevan MD (2000) Synaptic organisation of the basal ganglia. J Anat 196(Pt 4):527–542
Budde H, Wegner M, Soya H, Voelcker-Rehage C, McMorris T (2016) Neuroscience of Exercise: neuroplasticity and Its Behavioral Consequences. Neural Plast 2016:3643879. https://doi.org/10.1155/2016/3643879
Byun K, Hyodo K, Suwabe K, Ochi G, Sakairi Y, Kato M, Dan I, Soya H (2014) Positive effect of acute mild exercise on executive function via arousal-related prefrontal activations: an fNIRS study. Neuroimage 98:336–345. https://doi.org/10.1016/j.neuroimage.2014.04.067
Carro E, Nunez A, Busiguina S, Torres-Aleman I (2000) Circulating insulin-like growth factor I mediates effects of exercise on the brain. J Neurosci 20(8):2926–2933
Choi SH, Bylykbashi E, Chatila ZK, Lee SW, Pulli B, Clemenson GD, Kim E, Rompala A, Oram MK, Asselin C, Aronson J, Zhang C, Miller SJ, Lesinski A, Chen JW, Kim DY, van Praag H, Spiegelman BM, Gage FH, Tanzi RE (2018) Combined adult neurogenesis and BDNF mimic exercise effects on cognition in an Alzheimer’s mouse model. Science. https://doi.org/10.1126/science.aan8821
Clark PJ, Bhattacharya TK, Miller DS, Rhodes JS (2011) Induction of c-Fos, Zif268, and Arc from acute bouts of voluntary wheel running in new and pre-existing adult mouse hippocampal granule neurons. Neuroscience 184:16–27. https://doi.org/10.1016/j.neuroscience.2011.03.072
Collier TJ, Kanaan NM, Kordower JH (2011) Ageing as a primary risk factor for Parkinson’s disease: evidence from studies of non-human primates. Nat Rev Neurosci 12(6):359–366. https://doi.org/10.1038/nrn3039
Cotman CW, Berchtold NC (2002) Exercise: a behavioral intervention to enhance brain health and plasticity. Trends Neurosci 25(6):295–301
Cotman CW, Engesser-Cesar C (2002) Exercise enhances and protects brain function. Exerc Sport Sci Rev 30(2):75–79
Cotman CW, Berchtold NC, Christie LA (2007) Exercise builds brain health: key roles of growth factor cascades and inflammation. Trends Neurosci 30(9):464–472. https://doi.org/10.1016/j.tins.2007.06.011
Cui L, Hofer T, Rani A, Leeuwenburgh C, Foster TC (2009) Comparison of lifelong and late life exercise on oxidative stress in the cerebellum. Neurobiol Aging 30(6):903–909. https://doi.org/10.1016/j.neurobiolaging.2007.09.005
D’Angelo E (2018) Physiology of the cerebellum. Handb Clin Neurol 154:85–108. https://doi.org/10.1016/B978-0-444-63956-1.00006-0
Duman CH, Schlesinger L, Russell DS, Duman RS (2008) Voluntary exercise produces antidepressant and anxiolytic behavioral effects in mice. Brain Res 1199:148–158. https://doi.org/10.1016/j.brainres.2007.12.047
Ernst C, Olson AK, Pinel JP, Lam RW, Christie BR (2006) Antidepressant effects of exercise: evidence for an adult-neurogenesis hypothesis? J Psychiatry Neurosci 31(2):84–92
Flavell SW, Greenberg ME (2008) Signaling mechanisms linking neuronal activity to gene expression and plasticity of the nervous system. Annu Rev Neurosci 31:563–590. https://doi.org/10.1146/annurev.neuro.31.060407.125631
Gao HM, Jiang J, Wilson B, Zhang W, Hong JS, Liu B (2002) Microglial activation-mediated delayed and progressive degeneration of rat nigral dopaminergic neurons: relevance to Parkinson’s disease. J Neurochem 81(6):1285–1297
Gomez-Pinilla F, Ying Z, Roy RR, Molteni R, Edgerton VR (2002) Voluntary exercise induces a BDNF-mediated mechanism that promotes neuroplasticity. J Neurophysiol 88(5):2187–2195. https://doi.org/10.1152/jn.00152.2002
Grande I, Fries GR, Kunz M, Kapczinski F (2010) The role of BDNF as a mediator of neuroplasticity in bipolar disorder. Psychiatry Investig 7(4):243–250. https://doi.org/10.4306/pi.2010.7.4.243
Groenewegen HJ (2003) The basal ganglia and motor control. Neural Plast 10(1–2):107–120. https://doi.org/10.1155/NP.2003.107
Herrero MT, Barcia C, Navarro JM (2002) Functional anatomy of thalamus and basal ganglia. Childs Nerv Syst 18(8):386–404. https://doi.org/10.1007/s00381-002-0604-1
Holschneider DP, Yang J, Guo Y, Maarek JM (2007) Reorganization of functional brain maps after exercise training: importance of cerebellar-thalamic-cortical pathway. Brain Res 1184:96–107. https://doi.org/10.1016/j.brainres.2007.09.081
Hopkins ME, Davis FC, Vantieghem MR, Whalen PJ, Bucci DJ (2012) Differential effects of acute and regular physical exercise on cognition and affect. Neuroscience 215:59–68. https://doi.org/10.1016/j.neuroscience.2012.04.056
Horn EM, Kramer JM, Waldrop TG (2000) Development of hypoxia-induced Fos expression in rat caudal hypothalamic neurons. Neuroscience 99(4):711–720
Hsu YC, Chen HI, Kuo YM, Yu L, Huang TY, Chen SJ, Chuang JI, Wu FS, Jen CJ (2011) Chronic treadmill running in normotensive rats resets the resting blood pressure to lower levels by upregulating the hypothalamic GABAergic system. J Hypertens 29(12):2339–2348. https://doi.org/10.1097/HJH.0b013e32834c628f
Huang TY, Lin LS, Cho KC, Chen SJ, Kuo YM, Yu L, Wu FS, Chuang JI, Chen HI (1985) Jen CJ (2012) Chronic treadmill exercise in rats delicately alters the Purkinje cell structure to improve motor performance and toxin resistance in the cerebellum. J Appl Physiol 113(6):889–895. https://doi.org/10.1152/japplphysiol.01363.2011
Ichiyama RM, Gilbert AB, Waldrop TG, Iwamoto GA (2002) Changes in the exercise activation of diencephalic and brainstem cardiorespiratory areas after training. Brain Res 947(2):225–233
Isaacs KR, Anderson BJ, Alcantara AA, Black JE, Greenough WT (1992) Exercise and the brain: angiogenesis in the adult rat cerebellum after vigorous physical activity and motor skill learning. J Cereb Blood Flow Metab 12(1):110–119. https://doi.org/10.1038/jcbfm.1992.14
Jee YS, Ko IG, Sung YH, Lee JW, Kim YS, Kim SE, Kim BK, Seo JH, Shin MS, Lee HH, Cho HJ, Kim CJ (2008) Effects of treadmill exercise on memory and c-Fos expression in the hippocampus of the rats with intracerebroventricular injection of streptozotocin. Neurosci Lett 443(3):188–192. https://doi.org/10.1016/j.neulet.2008.07.078
Jenner P (2001) Parkinson’s disease, pesticides and mitochondrial dysfunction. Trends Neurosci 24(5):245–247
Kramer JM, Plowey ED, Beatty JA, Little HR, Waldrop TG (2000) Hypothalamus, hypertension, and exercise. Brain Res Bull 53(1):77–85
Legrand F, Heuze JP (2007) Antidepressant effects associated with different exercise conditions in participants with depression: a pilot study. J Sport Exerc Psychol 29(3):348–364
Lin TW, Chen SJ, Huang TY, Chang CY, Chuang JI, Wu FS, Kuo YM, Jen CJ (2012) Different types of exercise induce differential effects on neuronal adaptations and memory performance. Neurobiol Learn Mem 97(1):140–147. https://doi.org/10.1016/j.nlm.2011.10.006
Lin TW, Shih YH, Chen SJ, Lien CH, Chang CY, Huang TY, Chen SH, Jen CJ, Kuo YM (2015) Running exercise delays neurodegeneration in amygdala and hippocampus of Alzheimer’s disease (APP/PS1) transgenic mice. Neurobiol Learn Mem 118:189–197. https://doi.org/10.1016/j.nlm.2014.12.005
Liu PZ, Nusslock R (2018) Exercise-mediated neurogenesis in the hippocampus via BDNF. Front Neurosci 12:52. https://doi.org/10.3389/fnins.2018.00052
Liu YF, Chen HI, Wu CL, Kuo YM, Yu L, Huang AM, Wu FS, Chuang JI, Jen CJ (2009) Differential effects of treadmill running and wheel running on spatial or aversive learning and memory: roles of amygdalar brain-derived neurotrophic factor and synaptotagmin I. J Physiol 587(Pt 13):3221–3231. https://doi.org/10.1113/jphysiol.2009.173088
Marques-Aleixo I, Santos-Alves E, Balca MM, Rizo-Roca D, Moreira PI, Oliveira PJ, Magalhaes J, Ascensao A (2015) Physical exercise improves brain cortex and cerebellum mitochondrial bioenergetics and alters apoptotic, dynamic and auto(mito)phagy markers. Neuroscience 301:480–495. https://doi.org/10.1016/j.neuroscience.2015.06.027
Morales M, Margolis EB (2017) Ventral tegmental area: cellular heterogeneity, connectivity and behaviour. Nat Rev Neurosci 18(2):73–85. https://doi.org/10.1038/nrn.2016.165
Nambu A (2004) A new dynamic model of the cortico-basal ganglia loop. Prog Brain Res 143:461–466. https://doi.org/10.1016/S0079-6123(03)43043-4
Nestler EJ, Carlezon WA Jr (2006) The mesolimbic dopamine reward circuit in depression. Biol Psychiatry 59(12):1151–1159. https://doi.org/10.1016/j.biopsych.2005.09.018
Nishijima T, Kawakami M, Kita I (2013) Long-term exercise is a potent trigger for DeltaFosB induction in the hippocampus along the dorso-ventral axis. PLoS One 8(11):e81245. https://doi.org/10.1371/journal.pone.0081245
Okamoto M, Hojo Y, Inoue K, Matsui T, Kawato S, McEwen BS, Soya H (2012) Mild exercise increases dihydrotestosterone in hippocampus providing evidence for androgenic mediation of neurogenesis. Proc Natl Acad Sci USA 109(32):13100–13105. https://doi.org/10.1073/pnas.1210023109
Okamoto M, Yamamura Y, Liu YF, Min-Chul L, Matsui T, Shima T, Soya M, Takahashi K, Soya S, McEwen BS, Soya H (2015) Hormetic effects by exercise on hippocampal neurogenesis with glucocorticoid signaling. Brain Plast 1(1):149–158. https://doi.org/10.3233/BPL-150012
Paxinos G, Franklin KB (2004) The mouse brain in stereotaxic coordinates. Gulf Professional Publishing, Houston
Porcari JP, Bryant CX, Comana F (2015) Exercise physiology. F.A. Davis Company, Philadelphia
Porro CA, Francescato MP, Cettolo V, Diamond ME, Baraldi P, Zuiani C, Bazzocchi M, di Prampero PE (1996) Primary motor and sensory cortex activation during motor performance and motor imagery: a functional magnetic resonance imaging study. J Neurosci 16(23):7688–7698
Rhodes JS, Garland T Jr, Gammie SC (2003) Patterns of brain activity associated with variation in voluntary wheel-running behavior. Behav Neurosci 117(6):1243–1256. https://doi.org/10.1037/0735-7044.117.6.1243
Russo-Neustadt A, Beard RC, Cotman CW (1999) Exercise, antidepressant medications, and enhanced brain derived neurotrophic factor expression. Neuropsychopharmacology 21(5):679–682. https://doi.org/10.1016/S0893-133X(99)00059-7
Sasaki K, Gemba H (1984) Compensatory motor function of the somatosensory cortex for the motor cortex temporarily impaired by cooling in the monkey. Exp Brain Res 55(1):60–68
Smith Y, Bevan MD, Shink E, Bolam JP (1998) Microcircuitry of the direct and indirect pathways of the basal ganglia. Neuroscience 86(2):353–387
Soya H, Nakamura T, Deocaris CC, Kimpara A, Iimura M, Fujikawa T, Chang H, McEwen BS, Nishijima T (2007) BDNF induction with mild exercise in the rat hippocampus. Biochem Biophys Res Commun 358(4):961–967. https://doi.org/10.1016/j.bbrc.2007.04.173
Strohle A (2009) Physical activity, exercise, depression and anxiety disorders. J Neural Transm (Vienna) 116(6):777–784. https://doi.org/10.1007/s00702-008-0092-x
Tsai SF, Ku NW, Wang TF, Yang YH, Shih YH, Wu SY, Lee CW, Yu M, Yang TT, Kuo YM (2018a) Long-Term Moderate Exercise Rescues Age-Related Decline in Hippocampal Neuronal Complexity and Memory. Gerontology 64(6):551–561. https://doi.org/10.1159/000488589
Tsai SF, Wu HT, Chen PC, Chen YW, Yu M, Tzeng SF, Wu PH, Chen PS, Kuo YM (2018b) Stress Aggravates High-Fat-Diet-Induced Insulin Resistance via a Mechanism That Involves the Amygdala and Is Associated with Changes in Neuroplasticity. Neuroendocrinology 107:147–157. https://doi.org/10.1159/000491018
Tsou YH, Shih CT, Ching CH, Huang JY, Jen CJ, Yu L, Kuo YM, Wu FS, Chuang JI (2015) Treadmill exercise activates Nrf2 antioxidant system to protect the nigrostriatal dopaminergic neurons from MPP + toxicity. Exp Neurol 263:50–62. https://doi.org/10.1016/j.expneurol.2014.09.021
Uhlendorf TL, Van Kummer BH, Yaspelkis BB 3rd, Cohen RW (2011) Neuroprotective effects of moderate aerobic exercise on the spastic Han-Wistar rat, a model of ataxia. Brain Res 1369:216–222. https://doi.org/10.1016/j.brainres.2010.10.094
van Praag H (2009) Exercise and the brain: something to chew on. Trends Neurosci 32(5):283–290. https://doi.org/10.1016/j.tins.2008.12.007
van Praag H, Kempermann G, Gage FH (1999) Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci 2(3):266–270. https://doi.org/10.1038/6368
Whyte JJ, Laughlin MH (2010) The effects of acute and chronic exercise on the vasculature. Acta Physiol (Oxf) 199(4):441–450. https://doi.org/10.1111/j.1748-1716.2010.02127.x
Wrann CD (2015) FNDC5/irisin - their role in the nervous system and as a mediator for beneficial effects of exercise on the brain. Brain Plast 1(1):55–61. https://doi.org/10.3233/BPL-150019
Wu CW, Chang YT, Yu L, Chen HI, Jen CJ, Wu SY, Lo CP (1985) Kuo YM (2008) Exercise enhances the proliferation of neural stem cells and neurite growth and survival of neuronal progenitor cells in dentate gyrus of middle-aged mice. J Appl Physiol 105(5):1585–1594. https://doi.org/10.1152/japplphysiol.90775.2008
Wu SY, Wang TF, Yu L, Jen CJ, Chuang JI, Wu FS, Wu CW, Kuo YM (2011) Running exercise protects the substantia nigra dopaminergic neurons against inflammation-induced degeneration via the activation of BDNF signaling pathway. Brain Behav Immun 25(1):135–146. https://doi.org/10.1016/j.bbi.2010.09.006
Xu Q, Park Y, Huang X, Hollenbeck A, Blair A, Schatzkin A, Chen H (2010) Physical activities and future risk of Parkinson disease. Neurology 75(4):341–348. https://doi.org/10.1212/WNL.0b013e3181ea1597
Yanagisawa H, Dan I, Tsuzuki D, Kato M, Okamoto M, Kyutoku Y, Soya H (2010) Acute moderate exercise elicits increased dorsolateral prefrontal activation and improves cognitive performance with Stroop test. Neuroimage 50(4):1702–1710. https://doi.org/10.1016/j.neuroimage.2009.12.023
Yasumoto Y, Nakao R, Oishi K (2015) Free access to a running-wheel advances the phase of behavioral and physiological circadian rhythms and peripheral molecular clocks in mice. PLoS ONE 10(1):e0116476. https://doi.org/10.1371/journal.pone.0116476
Young J, Angevaren M, Rusted J, Tabet N (2015) Aerobic exercise to improve cognitive function in older people without known cognitive impairment. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd005381.pub4
Funding
This study was supported by the Ministry of Science and Technology, Taiwan (Grant 107-2320-B-006-054-MY3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict to interest
The authors declare that they have no conflict of interest.
Ethical approval
All animal experiments were done in accordance with the National Institute of Health Guideline for Animal Research (Guide for the Care and Use of Laboratory Animals) and approved by the National Cheng Kung University Institutional Animal Care and Use Committee (ethical approval reference number: 104272).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tsai, SF., Liu, YW. & Kuo, YM. Acute and long-term treadmill running differentially induce c-Fos expression in region- and time-dependent manners in mouse brain. Brain Struct Funct 224, 2677–2689 (2019). https://doi.org/10.1007/s00429-019-01926-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-019-01926-5