Abstract
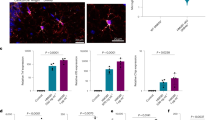
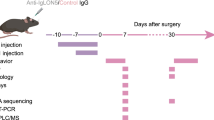
The antiphospholipid syndrome (APS) is an autoimmune disease characterized by the presence of antiphospholipid antibodies, which may trigger vascular thrombosis with consecutive infarcts. However, cognitive dysfunctions representing one of the most commonest neuropsychiatric symptoms are frequently present despite the absence of any ischemic brain lesions. Data on the structural and functional basis of the neuropsychiatric symptoms are sparse. To examine the effect of APS on hippocampal neurogenesis and on white matter, we induced experimental APS (eAPS) in adult female Balb/C mice by immunization with β2-glycoprotein 1. To investigate cell proliferation in the dentate gyrus granular cell layer (DG GCL), eAPS and control mice (n = 5, each) were injected with 5-bromo-2′-deoxyuridine (BrdU) once a day for 10 subsequent days. Sixteen weeks after immunization, eAPS resulted in a significant reduction of BrdU-positive cells in the DG GCL compared to control animals. However, double staining with doublecortin and NeuN revealed a largely preserved neurogenesis. Ultrastructural analysis of corpus callosum (CC) axons in eAPS (n = 6) and control mice (n = 7) revealed no significant changes in CC axon diameter or g-ratio. In conclusion, decreased cellular proliferation in the hippocampus of eAPS mice indicates a limited regenerative potential and may represent one neuropathological substrate of cognitive changes in APS while evidence for alterations of white matter integrity is lacking.



Similar content being viewed by others
Abbreviations
- aCL:
-
Anti-cardiolipin antibodies
- aPL:
-
Antiphospholipid antibodies
- APS:
-
Antiphospholipid syndrome
- BBB:
-
Blood–brain barrier
- BrdU:
-
5-Bromo-2′-deoxyuridine
- BSA:
-
Bovine serum albumin
- CA:
-
Cornu ammonis
- CNS:
-
Central nervous system
- CC:
-
Corpus callosum
- CFA:
-
Complete Freund’s adjuvant
- DCX:
-
Doublecortin
- DG GCL:
-
Dentate gyrus granular cell layer
- EAE:
-
Experimental autoimmune encephalomyelitis
- eAPS:
-
Experimental antiphospholipid syndrome
- LPS:
-
Lipopolysaccharide
- NGS:
-
Normal goat serum
- OD:
-
Optical density
- PBS:
-
Phosphate buffered saline
- PFA:
-
Paraformaldehyde
- RT:
-
Room temperature
- SEM:
-
Standard error of mean
- SLE:
-
Systemic lupus erythematosus
- SSC:
-
Saline-sodium citrate
- TBS:
-
Tris-buffered saline
- TBS-T:
-
TBS with Triton X-100
- WM:
-
White matter
References
Alenina N, Klempin F (2015) The role of serotonin in adult hippocampal neurogenesis. Behav Brain Res 277:49–57
Bakimer R, Fishman P, Blank M, Sredni B, Djaldetti M, Shoenfeld Y (1992) Induction of primary antiphospholipid syndrome in mice by immunization with a human monoclonal anticardiolipin antibody (H-3). J Clin Invest 89:1558–1563
Banasr M, Hery M, Printemps R, Daszuta A (2004) Serotonin-induced increases in adult cell proliferation and neurogenesis are mediated through different and common 5-HT receptor subtypes in the DG and the subventricular zone. Neuropsychopharmacology 29:450–460
Bastos GN, Moriya T, Inui F, Katura T, Nakahata N (2008) Involvement of cyclooxygenase-2 in lipopolysaccharide-induced impairment of the newborn cell survival in the adult mouse dentate gyrus. Neuroscience 155:454–462
Cervera R, Khamashta MA, Shoenfeld Y, Camps MT, Jacobsen S, Kiss E et al (2009) Morbidity and mortality in the antiphospholipid syndrome during a 5-year period: a multicentre prospective study of 1000 patients. Ann Rheum Dis 68:1428–1432
Cervera R, Serrano R, Pons-Estel GJ, Ceberio-Hualde L, Shoenfeld Y, de Ramón E et al (2015) Morbidity and mortality in the antiphospholipid syndrome during a 10-year period: a multicentre prospective study of 1000 patients. Ann Rheum Dis 74:1011–1018
Cohen J (1988) Statistical power analysis for the behavioral sciences 2. Lawrence Erlbaum Associates, Hillsdale
Conti F, Alessandri C, Perricone C, Scrivo R, Rezai S, Ceccarelli F et al (2012) Neurocognitive dysfunction in systemic lupus erythematosus: association with antiphospholipid antibodies, disease activity and chronic damage. PLoS One 7:e33824
Denenberg VH, Sherman GF, Rosen GD, Morrison L, Behan PO, Galaburda AM (1992) A behavior profile of the MRL/Mp lpr/lpr mouse and its association with hydrocephalus. Brain Behav Immun 6:40–49
Eisch AJ, Cameron HA, Encinas JM, Meltzer LA, Ming GL, Overstreet-Wadiche LS (2008) Adult neurogenesis, mental health, and mental illness: hope or hype? J Neurosci 28:11785–11791
Filley CM (2010) White matter: organization and functional relevance. Neuropsychol Rev 20:158–173
Frauenknecht K, Katzav A, Grimm C, Chapman J, Sommer CJ (2013) Neurological impairment in experimental antiphospholipid syndrome is associated with increased ligand binding to hippocampal and cortical serotonergic 5-HT1A receptors. Immunobiology 218:517–526
Frauenknecht K, Katzav A, Grimm C, Chapman J, Sommer CJ (2014) Altered receptor binding densities in experimental antiphospholipid syndrome despite only moderately enhanced autoantibody levels and absence of behavioral features. Immunobiology 219:341–349
Frauenknecht K, Katzav A, Weiss Lavi R, Sabag A, Otten S, Chapman J et al (2015) Mice with experimental antiphospholipid syndrome display hippocampal dysfunction and a reduction of dendritic complexity in hippocampal CA1 neurones. Neuropathol Appl Neurobiol 41:657–671
Fujioka H, Akema T (2010) Lipopolysaccharide acutely inhibits proliferation of neural precursor cells in the dentate gyrus in adult rats. Brain Res 1352:35–42
Gould E, Beylin A, Tanapat P, Reeves A, Shors TJ (1999) Learning enhances adult neurogenesis in the hippocampal formation. Nat Neurosci 2:260–265
Huehnchen P, Prozorovski T, Klaissle P, Lesemann A, Ingwersen J, Wolf SA et al (2011) Modulation of adult hippocampal neurogenesis during myelin-directed autoimmune neuroinflammation. Glia 59:132–142
Jacobson MW, Rapport LJ, Keenan PA, Coleman RD, Tietjen GE (1999) Neuropsychological deficits associated with antiphospholipid antibodies. J Clin Exp Neuropsychol 21:251–264
Kaichi Y, Kakeda S, Moriya J, Ohnari N, Saito K, Tanaka Y et al (2014) Brain MR findings in patients with systemic lupus erythematosus with and without antiphospholipid antibody syndrome. AJNR 35:100–105
Kapadia M, Stanojcic M, Earls AM, Pulapaka S, Lee J, Sakic B (2012) Altered olfactory function in the MRL model of CNS lupus. Behav Brain Res 234:303–311
Katzav A, Menachem A, Maggio N, Pollak L, Pick CG, Chapman J (2014) IgG accumulates in inhibitory hippocampal neurons of experimental antiphospholipid syndrome. J Autoimmun 55:86–93
Kempermann G, Song H, Gage FH (2015) Neurogenesis in the adult Hippocampus. Cold Spring Harb Perspect Biol 7:a018812
Kim A, Feng P, Ohkuri T, Sauers D, Cohn ZJ, Chai J et al (2012) Defects in the peripheral taste structure and function in the MRL/lpr mouse model of autoimmune disease. PLoS One 7:e35588
Klempin F, Babu H, de Pietri Tonelli D, Alarcon E, Fabel K, Kempermann G (2010) Oppositional effects of serotonin receptors 5-HT1a, 2, and 2c in the regulation of adult hippocampal neurogenesis. Front Mol Neurosci. https://doi.org/10.3389/fnmol.2010.00014
Kronenberg G, Reuter K, Steiner B, Brandt MD, Jessberger S, Yamaguchi M et al (2003) Subpopulations of proliferating cells of the adult hippocampus respond differently to physiologic neurogenic stimuli. J Comp Neurol 467:455–463
Ma X, Foster J, Sakic B (2006) Distribution and prevalence of leukocyte phenotypes in brains of lupus-prone mice. J Neuroimmunol 179:26–36
Mandyam CD, Harburg GC, Eisch AJ (2007) Determination of key aspects of precursor cell proliferation, cell cycle length and kinetics in the adult mouse subgranular zone. Neuroscience 146:108–122
Nakagawa E, Aimi Y, Yasuhara O, Tooyama I, Shimada M, McGeer PL et al (2000) Enhancement of progenitor cell division in the dentate gyrus triggered by initial limbic seizures in rat models of epilepsy. Epilepsia 41:10–18
Parent JM, Elliott RC, Pleasure SJ, Barbaro NM, Lowenstein DH (2006) Aberrant seizure-induced neurogenesis in experimental temporal lobe epilepsy. Ann Neurol 59:81–91
Penke L, Maniega SM, Bastin ME, Valdés Hernández MC, Murray C, Royle NA et al (2012) Brain white matter tract integrity as a neural foundation for general intelligence. Mol Psychiatry 17:1026–1030
Pereira FR, Macri F, Jackowski MP, Kostis WJ, Gris JC, Beregi JP et al (2016) Diffusion tensor imaging in patients with obstetric antiphospholipid syndrome without neuropsychiatric symptoms. Eur Radiol 4:959–968
Pick CG, Peter Y, Terkel J, Gavish M, Weizman R (1996) Effect of the neuroactive steroid alpha-THDOC on staircase test behavior in mice. Psychopharmacology 128:61–66
Radley JJ, Jacobs BL (2002) 5-HT1A receptor antagonist administration decreases cell proliferation in the DG. Brain Res 955:264–267
Sakic B, Szechtman H, Denburg JA, Gorny G, Kolb B, Whishaw IQ (1998) Progressive atrophy of pyramidal neuron dendrites in autoimmune MRL-lpr mice. J Neuroimmunol 87:162–170
Sakic B, Kolb B, Whishaw IQ, Gorny G, Szechtman H, Denburg JA (2000) Immunosuppression prevents neuronal atrophy in lupus-prone mice: evidence for brain damage induced by autoimmune disease? J Neuroimmunol 111:93–101
Shrot S, Katzav A, Korczyn AD, Litvinju Y, Hershenson R, Pick CG et al (2002) Behavioral and cognitive deficits occur only after prolonged exposure of mice to antiphospholipid antibodies. Lupus 11:736–743
Simiand J, Keane PE, Morre M (1984) The staircase test in mice: a simple and efficient procedure for primary screening of anxiolytic agents. Psychopharmacology 84:48–53
Sled JG, Spring S, van Eede M, Lerch JP, Ullal S, Sakic B (2009) Time course and nature of brain atrophy in the MRL mouse model of central nervous system lupus. Arthritis Rheum 60:1764–1774
Sokol DK, O’Brien RS, Wagenknecht DR, Rao T, McIntyre JA (2007) Antiphospholipid antibodies in blood and cerebrospinal fluids of patients with psychosis. J Neuroimmunol 190:151–156
Stanojcic M, Burstyn-Cohen T, Nashi N, Lemke G, Sakic B (2009) Disturbed distribution of proliferative brain cells during lupus-like disease. Brain Behav Immun 23:1003–1013
Stojanovich L, Kontic M, Smiljanic D, Djokovic A, Stamenkovic B, Marisavljevic D (2013) Association between non-thrombotic neurological and cardiac manifestations in patients with antiphospholipid syndrome. Clin Exp Rheumatol 31:756–760
Tektonidou MG, Varsou N, Kotoulas G, Antoniou A, Moutsopoulos HM (2006) Cognitive deficits in patients with antiphospholipid syndrome: association with clinical, laboratory, and brain magnetic resonance imaging finding. Arch Int Med 166:2278–2284
Tozuka Y, Fukuda S, Namba T, Seki T, Hisatsune T (2005) GABAergic excitation promotes neuronal differentiation in adult hippocampal progenitor cells. Neuron 47:803–815
Ueda S, Sakakibara S, Yoshimoto K (2005) Effect of long-lasting serotonin depletion on environmental enrichment-induced neurogenesis in adult rat hippocampus and spatial learning. Neuroscience 135:395–402
Ulrich-Lai YM, Herman JP (2009) Neural regulation of endocrine and autonomic stress responses. Nat Rev Neurosci 10:397–409
Valdés-Ferrer SI, Vega F, Cantú-Brito C, Ceballos-Ceballos J, Estañol B, García-Ramos G et al (2008) Cerebral changes in SLE with or without antiphospholipid syndrome: a case–control MRI study. J Neuroimaging 18:62–65
van Praag H, Kempermann G, Gage FH (1999) Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci 2:266–270
Wang LP, Kempermann G, Kettenmann H (2005) A subpopulation of precursor cells in the mouse DG receives synaptic GABAergic input. Mol Cell Neurosci 29:181–189
Yelnik CM, Kozora E, Appenzeller S (2016a) Cognitive disorders and antiphospholipid antibodies. Autoimmun Rev 15:1193–1198
Yelnik CM, Kozora E, Appenzeller S (2016b) Non-stroke central neurological manifestations in antiphospholipid syndrome. Curr Rheumatol Rep (2016) 18:11
Acknowledgements
The technical expertise of Magdeleine Herkt and Nicole Roder is kindly acknowledged. The authors are grateful to Professor Elisabeth Jane Rushing (University Hospital Zurich, Switzerland) for critically reading the manuscript.
Funding
The present work was supported by grants from the MAIFOR program (intramural funding, Medical Center of the Johannes Gutenberg University Mainz, Germany) from the Chief Scientist in the Israel Ministry of Health and from Israel Science Foundation. Katrin Frauenknecht is the recipient of a Career Development Award by the Stavros Niarchos Foundation (https://www.snf.org/22474).
Author information
Authors and Affiliations
Contributions
Experimental design: KF, AK, CJS; animal experiments: AK, RW; histology/volumetry/morphometric studies: KF, PL, HDP; figures: KF, PL, HDP; manuscript KF, PL, JC, CJS. All authors read and approved the final manuscript. The authors declare no competing interests.
Corresponding author
Ethics declarations
Ethical approval
Animal experiments have been approved Israeli Health Ministry (Ethical approval no. 775/12) and by the Chaim Sheba Medical Center Animal Welfare Committee. All experiments were in accordance with national and international guidelines and regulations. This article does not contain any studies with human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Frauenknecht, K., Leukel, P., Weiss, R. et al. Decreased hippocampal cell proliferation in mice with experimental antiphospholipid syndrome. Brain Struct Funct 223, 3463–3471 (2018). https://doi.org/10.1007/s00429-018-1699-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1699-9