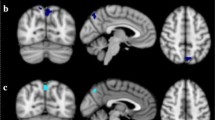
Abstract
Previous research has shown associations between brain structure and resting state high-frequency heart rate variability (HF HRV). Age affects both brain structure and HF HRV. Therefore, we sought to examine the relationship between brain structure and HF HRV as a function of age. Data from two independent studies were used for the present analysis. Study 1 included 19 older adults (10 males, age range 62–78 years) and 19 younger adults (12 males, age range 19–37). Study 2 included 23 older adults (12 males; age range 55–75) and 27 younger adults (17 males; age range 18–34). The root-mean-square of successive R–R-interval differences (RMSSD) from ECG recordings was used as time-domain measure of HF HRV. MRI scans were performed on a 3.0-T Siemens Magnetom Trio scanner. Cortical reconstruction and volumetric segmentation were performed with the Freesurfer image analysis suite, including 12 regions as regions of interests (ROI). Zero-order and partial correlations were used to assess the correlation of RMSSD with cortical thickness in selected ROIs. Lateral orbitofrontal cortex (OFC) cortical thickness was significantly associated with RMSSD. Further, both studies, in line with previous research, showed correlations between RMSSD and anterior cingulate cortex (ACC) cortical thickness. Meta-analysis on adjusted correlation coefficients from individual studies confirmed an association of RMSSD with the left rostral ACC and the left lateral OFC. Future longitudinal studies are necessary to trace individual trajectories in the association of HRV and brain structure across aging.


Similar content being viewed by others
References
Allen B, Jennings JR, Gianaros PJ, Thayer JF, Manuck SB (2015) Resting high-frequency heart rate variability is related to resting brain perfusion. Psychophysiology 52(2):277–287. doi:10.1111/psyp.12321
Alosco ML, Gunstad J, Xu X, Clark US, Labbe DR, Riskin-Jones HH, Terrero G, Schwarz NF, Walsh EG, Poppas A, Cohen RA, Sweet LH (2014) The impact of hypertension on cerebral perfusion and cortical thickness in older adults. J Am Soc Hypertens 8(8):561–570. doi:10.1016/j.jash.2014.04.002
Alvares GA, Quintana DS, Kemp AH, Van Zwieten A, Balleine BW, Hickie IB, Guastella AJ (2013) Reduced heart rate variability in social anxiety disorder: associations with gender and symptom severity. PLoS One 8:e70468
Antelmi I, de Paula RS, Shinzato AR, Peres CA, Mansur AJ, Grupi CJ (2004) Influence of age, gender, body mass index, and functional capacity on heart rate variability in a cohort of subjects without heart disease. Am J Cardiol 93:381–385
Beauchaine TP, Thayer JF (2015) Heart rate variability as a transdiagnostic biomarker of psychopathology. Int J Psychophysiol Off J Int Organ Psychophysiol 98:338–350
Beissner F, Meissner K, Bär KJ, Napadow V (2013) The autonomic brain: an activation likelihood estimation meta-analysis for central processing of autonomic function. J Neurosci 33(25):10503–10511. doi:10.1523/JNEUROSCI.1103-13.2013
Bertsch K, Hagemann D, Naumann E, Schächinger H, Schulz A (2012) Stability of heart rate variability indices reflecting parasympathetic activity. Psychophysiology 49:672–682
Clamor A, Lincoln TM, Thayer JF, Koenig J (2016) Resting vagal activity in schizophrenia: meta-analysis of heart rate variability as a potential endophenotype. Br J Psychiatry J Ment Sci 208:9–16
Cooper H, Hedges LV, Valentine JC (eds) (2009) The handbook of research synthesis and meta-analysis. Russell Sage Foundation, New York
Dale AM, Sereno MI (1993) Improved localization of cortical activity by combining EEG and MEG with MRI cortical surface reconstruction: a linear approach. J Cogn Neurosci 5:162–176
Dale AM, Fischl B, Sereno MI (1999) Cortical surface-based analysis. I. Segmentation and surface reconstruction. NeuroImage 9:179–194
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7(3):177–188
Development Core Team R (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Fischl B, Dale AM (2000) Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc Natl Acad Sci USA 97:11050–11055
Fischl B, Sereno MI, Dale AM (1999a) Cortical surface-based analysis. II: inflation, flattening, and a surface-based coordinate system. NeuroImage 9:195–207
Fischl B, Sereno MI, Tootell RB, Dale AM (1999b) High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum Brain Mapp 8:272–284
Fischl B, Liu A, Dale AM (2001) Automated manifold surgery: constructing geometrically accurate and topologically correct models of the human cerebral cortex. IEEE Trans Med Imaging 20:70–80
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, van der Kouwe A, Killiany R, Kennedy D, Klaveness S, Montillo A, Makris N, Rosen B, Dale AM (2002) Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33:341–355
Fischl B, Salat DH, van der Kouwe AJW, Makris N, Ségonne F, Quinn BT, Dale AM (2004a) Sequence-independent segmentation of magnetic resonance images. NeuroImage 23(Suppl 1):S69–S84
Fischl B, van der Kouwe A, Destrieux C, Halgren E, Ségonne F, Salat DH, Busa E, Seidman LJ, Goldstein J, Kennedy D, Caviness V, Makris N, Rosen B, Dale AM (2004b) Automatically parcellating the human cerebral cortex. Cereb Cortex N Y N 14:11–22
Fjell AM, Walhovd KB (2010) Structural brain changes in aging: courses, causes and cognitive consequences. Rev Neurosci 21:187–221
Fjell AM, Westlye LT, Amlien I, Espeseth T, Reinvang I, Raz N, Agartz I, Salat DH, Greve DN, Fischl B, Dale AM, Walhovd KB (2009) High consistency of regional cortical thinning in aging across multiple samples. Cereb Cortex N Y N 19:2001–2012
FS Wiki (2016) Website. http://www.freesurfer.net/fswiki/eTIV. Last access 31 May 2017
Gatz M, Reynolds C, Nikolic J, Lowe B, Karel M, Pedersen N (1995) An empirical test of telephone screening to identify potential dementia cases. Int Psychogeriatr 7:429–438
Gianaros PJ, Wager TD (2015) Brain-body pathways linking psychological stress and physical health. Curr Dir Psychol Sci 24(4):313–321
Han X, Jovicich J, Salat D, van der Kouwe A, Quinn B, Czanner S, Busa E, Pacheco J, Albert M, Killiany R, Maguire P, Rosas D, Makris N, Dale A, Dickerson B, Fischl B (2006) Reliability of MRI-derived measurements of human cerebral cortical thickness: the effects of field strength, scanner upgrade and manufacturer. NeuroImage 32:180–194
Harms MP, Wang L, Campanella C, Aldridge K, Moffitt AJ, Kuelper J, Ratnanather JT, Miller MI, Barch DM, Csernansky JG (2010) Correspondence. Br J Psychiatry 196(5):414–415
Jarczok MN, Kleber ME, Koenig J, Loerbroks A, Herr RM, Hoffmann K, Fischer JE, Benyamini Y, Thayer JF (2015) Investigating the associations of self-rated health: heart rate variability is more strongly associated than inflammatory and other frequently used biomarkers in a cross sectional occupational sample. PLoS One 10:e0117196
Jovicich J, Czanner S, Greve D, Haley E, van der Kouwe A, Gollub R, Kennedy D, Schmitt F, Brown G, Macfall J, Fischl B, Dale A (2006) Reliability in multi-site structural MRI studies: effects of gradient non-linearity correction on phantom and human data. NeuroImage 30:436–443
Kemp AH, Quintana DS, Gray MA, Felmingham KL, Brown K, Gatt JM (2010) Impact of depression and antidepressant treatment on heart rate variability: a review and meta-analysis. Biol Psychiatry 67:1067–1074
Koenig J, Kemp AH, Feeling NR, Thayer JF, Kaess M (2015) Resting state vagal tone in borderline personality disorder: a meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry 64:18–26
Koenig J, Kemp AH, Beauchaine TP, Thayer JF, Kaess M (2016) Depression and resting state heart rate variability in children and adolescents—a systematic review and meta-analysis. Clin Psychol Rev 46:136–150
Kuo TBJ, Lai CJ, Huang Y-T, Yang CCH (2005) Regression analysis between heart rate variability and baroreflex-related vagus nerve activity in rats. J Cardiovasc Electrophysiol 16:864–869
Levy MN (1997) Neural control of cardiac function. Baillières Clin Neurol 6:227–244
Mather M (2016) The Affective Neuroscience of Aging. Annu Rev Psychol 67:213–238
O’Brien IA, O’Hare P, Corrall RJ (1986) Heart rate variability in healthy subjects: effect of age and the derivation of normal ranges for tests of autonomic function. Br Heart J 55:348–354
Oppenheimer S, Cechetto D (2016) The insular cortex and the regulation of cardiac function. Compr Physiol 6(2):1081–1133. doi:10.1002/cphy.c140076
Peters R (2006) Ageing and the brain. Postgrad Med J 82:84–88
Sakaki M, Nga L, Mather M (2013) Amygdala functional connectivity with medial prefrontal cortex at rest predicts the positivity effect in older adults’ memory. J Cogn Neurosci 25:1206–1224
Sakaki M, Yoo HJ, Nga L, Lee T-H, Thayer JF, Mather M (2016) Heart rate variability is associated with amygdala functional connectivity with MPFC across younger and older adults. NeuroImage 139:44–52
Schwarzer G (2007) Meta: an R package for meta-analysis. R News 7(3):40–45
Ségonne F, Dale AM, Busa E, Glessner M, Salat D, Hahn HK, Fischl B (2004) A hybrid approach to the skull stripping problem in MRI. NeuroImage 22:1060–1075
Tarvainen MP, Niskanen J-P, Lipponen JA, Ranta-Aho PO, Karjalainen PA (2014) Kubios HRV—heart rate variability analysis software. Comput Methods Programs Biomed 113:210–220
Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology (1996) Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 93:1043–1065
Thayer JF, Lane RD (2000) A model of neurovisceral integration in emotion regulation and dysregulation. J Affect Disord 61:201–216
Thayer JF, Lane RD (2009) Claude Bernard and the heart-brain connection: further elaboration of a model of neurovisceral integration. Neurosci Biobehav Rev 33:81–88
Thayer JF, Hansen AL, Saus-Rose E, Johnsen BH (2009a) Heart rate variability, prefrontal neural function, and cognitive performance: the neurovisceral integration perspective on self-regulation, adaptation, and health. Ann Behav Med Publ Soc Behav Med 37:141–153
Thayer JF, Sollers JJ, Labiner DM, Weinand M, Herring AM, Lane RD, Ahern GL (2009b) Age-related differences in prefrontal control of heart rate in humans: a pharmacological blockade study. Int J Psychophysiol Off J Int Organ Psychophysiol 72:81–88
Thayer JF, Yamamoto SS, Brosschot JF (2010) The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int J Cardiol 141(2):122–131. doi:10.1016/j.ijcard.2009.09.543
Thayer JF, Ahs F, Fredrikson M, Sollers JJ, Wager TD (2012) A meta-analysis of heart rate variability and neuroimaging studies: implications for heart rate variability as a marker of stress and health. Neurosci Biobehav Rev 36:747–756
Virtanen R, Jula A, Kuusela T, Helenius H, Voipio-Pulkki LM (2003) Reduced heart rate variability in hypertension: associations with lifestyle factors and plasma renin activity. J Hum Hypertens 17(3):171–179
Winkelmann T, Thayer JF, Pohlack S, Nees F, Grimm O, Flor H (2017) Structural brain correlates of heart rate variability in a healthy young adult population. Brain Struct Funct 222:1061–1068
Woodward SH, Kaloupek DG, Schaer M, Martinez C, Eliez S (2008) Right anterior cingulate cortical volume covaries with respiratory sinus arrhythmia magnitude in combat veterans. J Rehabil Res Dev 45:451–463
Zhang J (2007) Effect of age and sex on heart rate variability in healthy subjects. J Manip Physiol Ther 30:374–379
Acknowledgements
JK is supported by Physician-Scientist-Fellowship provided by the Medical School, University of Heidelberg, Germany. JK acknowledges the financial support through a Post-Doctoral Scholarship provided by the Daimler and Benz Foundation (Ladenburg, Germany) and the Thrasher Research Fund Early Career Award provided by the Thrasher Research Fund (Salt Lake City, UT, USA). The research studies were funded by NIA R01AG025340. Michiko Sakaki received a fellowship by the European Commision PCIG13-GA-2013-618600, JSPS 16H05959.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Data are available online via the Open Science Framework (https://osf.io/pgmrx/).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoo, H.J., Thayer, J.F., Greening, S. et al. Brain structural concomitants of resting state heart rate variability in the young and old: evidence from two independent samples. Brain Struct Funct 223, 727–737 (2018). https://doi.org/10.1007/s00429-017-1519-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1519-7