Abstract
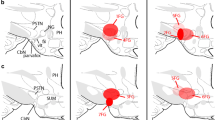
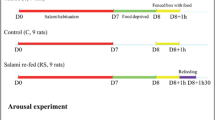
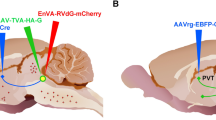
Following fasting, satiety is accompanied by neuronal activation in brain areas including the central amygdalar nucleus (CEA). Since CEA is known to inhibit food intake, we hypothesized that CEA contributes to the termination of meal during refeeding. To better understand the organization of this satiety-related circuit, the interconnections of the CEA with refeeding-activated neuronal groups were elucidated using retrograde (cholera toxin-β subunit, CTB) and anterograde (phaseolus vulgaris leucoagglutinin, PHA-L) tracers in male rats. C-Fos-immunoreactivity was used as marker of neuronal activation. The refeeding-activated input of the CEA primarily originated from the paraventricular thalamic, parasubthalamic and parabrachial nuclei. Few CTB-c-Fos double-labeled neurons were detected in the prefrontal cortex, lateral hypothalamic area, nucleus of the solitary tract (NTS) and the bed nuclei of the stria terminalis (BNST). Only few refeeding-activated proopiomelanocortin-producing neurons of the arcuate nucleus projected to the CEA. Anterograde tract tracing revealed a high density of PHAL-labeled axons contacted with refeeding-activated neurons in the BNST, lateral hypothalamic area, parasubthalamic, paraventricular thalamic and parabrachial nuclei and NTS; a low density of labeled axons was found in the paraventricular hypothalamic nucleus. Chemogenetic activation of the medial CEA (CEAm) inhibited food intake during the first hour of refeeding, while activation of lateral CEA had no effect. These data demonstrate the existence of reciprocal connections between the CEA and distinct refeeding-activated hypothalamic, thalamic and brainstem nuclei, suggesting the importance of short feedback loops in the regulation of satiety and importance of the CEAm in the regulation of food intake during refeeding.













Similar content being viewed by others
References
Alexander GM, Rogan SC, Abbas AI, Armbruster BN, Pei Y, Allen JA, Nonneman RJ, Hartmann J, Moy SS, Nicolelis MA, McNamara JO, Roth BL (2009) Remote control of neuronal activity in transgenic mice expressing evolved G protein-coupled receptors. Neuron 63(1):27–39. doi:10.1016/j.neuron.2009.06.014
Baldwin AE, Holahan MR, Sadeghian K, Kelley AE (2000) N-methyl-d-aspartate receptor-dependent plasticity within a distributed corticostriatal network mediates appetitive instrumental learning. Behav Neurosci 114(1):84–98
Baldwin AE, Sadeghian K, Kelley AE (2002) Appetitive instrumental learning requires coincident activation of NMDA and dopamine D1 receptors within the medial prefrontal cortex. J Neurosci Off J Soc Neurosci 22(3):1063–1071
Balfour RH, Trapp S (2007) Ionic currents underlying the response of rat dorsal vagal neurones to hypoglycaemia and chemical anoxia. J Physiol 579(Pt 3):691–702. doi:10.1113/jphysiol.2006.126094
Balleine BW, Dickinson A (1998) Goal-directed instrumental action: contingency and incentive learning and their cortical substrates. Neuropharmacology 37(4–5):407–419
Balthasar N, Dalgaard LT, Lee CE, Yu J, Funahashi H, Williams T, Ferreira M, Tang V, McGovern RA, Kenny CD, Christiansen LM, Edelstein E, Choi B, Boss O, Aschkenasi C, Zhang CY, Mountjoy K, Kishi T, Elmquist JK, Lowell BB (2005) Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 123(3):493–505. doi:10.1016/j.cell.2005.08.035
Barrachina MD, Martinez V, Wang L, Wei JY, Tache Y (1997) Synergistic interaction between leptin and cholecystokinin to reduce short-term food intake in lean mice. Proc Natl Acad Sci USA 94(19):10455–10460
Bhatnagar S, Dallman MF (1999) The paraventricular nucleus of the thalamus alters rhythms in core temperature and energy balance in a state-dependent manner. Brain Res 851(1–2):66–75
Bienkowski MS, Rinaman L (2013) Common and distinct neural inputs to the medial central nucleus of the amygdala and anterior ventrolateral bed nucleus of stria terminalis in rats. Brain Struct Funct 218(1):187–208. doi:10.1007/s00429-012-0393-6
Blouet C, Schwartz GJ (2012) Brainstem nutrient sensing in the nucleus of the solitary tract inhibits feeding. Cell Metab 16(5):579–587. doi:10.1016/j.cmet.2012.10.003
Carter ME, Soden ME, Zweifel LS, Palmiter RD (2013) Genetic identification of a neural circuit that suppresses appetite. Nature 503(7474):111–114. doi:10.1038/nature12596
Chen S, Su HS (1990) Afferent connections of the thalamic paraventricular and parataenial nuclei in the rat—a retrograde tracing study with iontophoretic application of Fluoro-Gold. Brain Res 522(1):1–6
Chometton S, Pedron S, Peterschmitt Y, Van Waes V, Fellmann D, Risold PY (2016) A premammillary lateral hypothalamic nuclear complex responds to hedonic but not aversive tastes in the male rat. Brain Struct Funct 221(4):2183–2208. doi:10.1007/s00429-015-1038-3
Cornwall J, Phillipson OT (1988) Afferent projections to the dorsal thalamus of the rat as shown by retrograde lectin transport. II. The midline nuclei. Brain Res Bull 21(2):147–161
DeCoteau WE, Kesner RP, Williams JM (1997) Short-term memory for food reward magnitude: the role of the prefrontal cortex. Behav Brain Res 88(2):239–249
Delgado JM, Anand BK (1953) Increase of food intake induced by electrical stimulation of the lateral hypothalamus. Am J Physiol 172(1):162–168
Elmquist JK, Scammell TE, Jacobson CD, Saper CB (1996) Distribution of Fos-like immunoreactivity in the rat brain following intravenous lipopolysaccharide administration. J Comp Neurol 371(1):85–103. doi:10.1002/(SICI)1096-9861(19960715)371:1<85:AID-CNE5>3.0.CO;2-H
Elmquist JK, Elias CF, Saper CB (1999) From lesions to leptin: hypothalamic control of food intake and body weight. Neuron 22(2):221–232
Fekete C, Zseli G, Singru PS, Kadar A, Wittmann G, Fuzesi T, El-Bermani W, Lechan RM (2012) Activation of anorexigenic pro-opiomelanocortin neurones during refeeding is independent of vagal and brainstem inputs. J Neuroendocrinol 24(11):1423–1431. doi:10.1111/j.1365-2826.2012.02354.x
French SJ, Murray B, Rumsey RD, Sepple CP, Read NW (1993) Preliminary studies on the gastrointestinal responses to fatty meals in obese people. Int J Obes Relat Metab Disord J Int Assoc Study Obes 17(5):295–300
Geliebter A, Westreich S, Gage D (1988) Gastric distention by balloon and test-meal intake in obese and lean subjects. Am J Clin Nutr 48(3):592–594
Gibson CD, Carnell S, Ochner CN, Geliebter A (2010) Neuroimaging, gut peptides and obesity: novel studies of the neurobiology of appetite. J Neuroendocrinol 22(8):833–845. doi:10.1111/j.1365-2826.2010.02025.x
Goto M, Swanson LW (2004) Axonal projections from the parasubthalamic nucleus. J Comp Neurol 469(4):581–607. doi:10.1002/cne.11036
Grill HJ, Hayes MR (2009) The nucleus tractus solitarius: a portal for visceral afferent signal processing, energy status assessment and integration of their combined effects on food intake. Int J Obes 33(Suppl 1):S11–S15. doi:10.1038/ijo.2009.10
Huszar D, Lynch CA, Fairchild-Huntress V, Dunmore JH, Fang Q, Berkemeier LR, Gu W, Kesterson RA, Boston BA, Cone RD, Smith FJ, Campfield LA, Burn P, Lee F (1997) Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88(1):131–141
Ji H, Friedman MI (1999) Compensatory hyperphagia after fasting tracks recovery of liver energy status. Physiol Behav 68(1–2):181–186
Johnstone LE, Fong TM, Leng G (2006) Neuronal activation in the hypothalamus and brainstem during feeding in rats. Cell Metab 4(4):313–321. doi:10.1016/j.cmet.2006.08.003
Kelley AE, Baldo BA, Pratt WE, Will MJ (2005) Corticostriatal-hypothalamic circuitry and food motivation: integration of energy, action and reward. Physiol Behav 86(5):773–795. doi:10.1016/j.physbeh.2005.08.066
Kesner RP, Gilbert PE (2007) The role of the agranular insular cortex in anticipation of reward contrast. Neurobiol Learn Mem 88(1):82–86. doi:10.1016/j.nlm.2007.02.002
Kirouac GJ, Parsons MP, Li S (2005) Orexin (hypocretin) innervation of the paraventricular nucleus of the thalamus. Brain Res 1059(2):179–188. doi:10.1016/j.brainres.2005.08.035
Kirouac GJ, Parsons MP, Li S (2006) Innervation of the paraventricular nucleus of the thalamus from cocaine- and amphetamine-regulated transcript (CART) containing neurons of the hypothalamus. J Comp Neurol 497(2):155–165. doi:10.1002/cne.20971
Kishi T, Aschkenasi CJ, Lee CE, Mountjoy KG, Saper CB, Elmquist JK (2003) Expression of melanocortin 4 receptor mRNA in the central nervous system of the rat. J Comp Neurol 457(3):213–235. doi:10.1002/cne.10454
Lin Z, Gao N, Hu HZ, Liu S, Gao C, Kim G, Ren J, Xia Y, Peck OC, Wood JD (2002) Immunoreactivity of Hu proteins facilitates identification of myenteric neurones in guinea-pig small intestine. Neurogastroenterol Motility Off J Eur Gastrointest Motil Soc 14(2):197–204
Liposits Z, Setalo G, Flerko B (1984) Application of the silver-gold intensified 3,3′-diaminobenzidine chromogen to the light and electron microscopic detection of the luteinizing hormone-releasing hormone system of the rat brain. Neuroscience 13(2):513–525
Llewellyn-Smith IJ, Reimann F, Gribble FM, Trapp S (2011) Preproglucagon neurons project widely to autonomic control areas in the mouse brain. Neuroscience 180:111–121. doi:10.1016/j.neuroscience.2011.02.023
Marusich MF, Furneaux HM, Henion PD, Weston JA (1994) Hu neuronal proteins are expressed in proliferating neurogenic cells. J Neurobiol 25(2):143–155. doi:10.1002/neu.480250206
Moga MM, Weis RP, Moore RY (1995) Efferent projections of the paraventricular thalamic nucleus in the rat. J Comp Neurol 359(2):221–238. doi:10.1002/cne.903590204
Morton GJ, Cummings DE, Baskin DG, Barsh GS, Schwartz MW (2006) Central nervous system control of food intake and body weight. Nature 443(7109):289–295. doi:10.1038/nature05026
Murphy EM, Defontgalland D, Costa M, Brookes SJ, Wattchow DA (2007) Quantification of subclasses of human colonic myenteric neurons by immunoreactivity to Hu, choline acetyltransferase and nitric oxide synthase. Neurogastroenterol Motil Off J Eur Gastrointest Motil Soc 19(2):126–134. doi:10.1111/j.1365-2982.2006.00843.x
Otake K (2005) Cholecystokinin and substance P immunoreactive projections to the paraventricular thalamic nucleus in the rat. Neurosci Res 51(4):383–394. doi:10.1016/j.neures.2004.12.009
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates. Academic Press, San Diego
Peters JH, Ritter RC, Simasko SM (2006) Leptin and CCK selectively activate vagal afferent neurons innervating the stomach and duodenum. Am J Physiol Regul Integr Comp Physiol 290(6):R1544–R1549. doi:10.1152/ajpregu.00811.2005
Petrovich GD, Ross CA, Mody P, Holland PC, Gallagher M (2009) Central, but not basolateral, amygdala is critical for control of feeding by aversive learned cues. J Neurosci Off J Soc Neurosci 29(48):15205–15212. doi:10.1523/JNEUROSCI.3656-09.2009
Reznikov LR, Reagan LP, Fadel JR (2008) Activation of phenotypically distinct neuronal subpopulations in the anterior subdivision of the rat basolateral amygdala following acute and repeated stress. J Comp Neurol 508(3):458–472. doi:10.1002/cne.21687
Ritter S, Dinh TT, Zhang Y (2000) Localization of hindbrain glucoreceptive sites controlling food intake and blood glucose. Brain Res 856(1–2):37–47
Rizvi TA, Ennis M, Behbehani MM, Shipley MT (1991) Connections between the central nucleus of the amygdala and the midbrain periaqueductal gray: topography and reciprocity. J Comp Neurol 303(1):121–131. doi:10.1002/cne.903030111
Rollins BL, King BM (2000) Amygdala-lesion obesity: what is the role of the various amygdaloid nuclei? Am J Physiol Regul Integr Comp Physiol 279(4):R1348–R1356
Sah P, Faber ES, Lopez De Armentia M, Power J (2003) The amygdaloid complex: anatomy and physiology. Physiol Rev 83(3):803–834. doi:10.1152/physrev.00002.2003
Sato I, Arima H, Ozaki N, Watanabe M, Goto M, Hayashi M, Banno R, Nagasaki H, Oiso Y (2005) Insulin inhibits neuropeptide Y gene expression in the arcuate nucleus through GABAergic systems. J Neurosci Off J Soc Neurosci 25(38):8657–8664. doi:10.1523/JNEUROSCI.2739-05.2005
Schwartz GJ (2000) The role of gastrointestinal vagal afferents in the control of food intake: current prospects. Nutrition 16(10):866–873
Schwartz MW, Woods SC, Porte D Jr, Seeley RJ, Baskin DG (2000) Central nervous system control of food intake. Nature 404(6778):661–671. doi:10.1038/35007534
Serrats J, Sawchenko PE (2006) CNS activational responses to staphylococcal enterotoxin B: T-lymphocyte-dependent immune challenge effects on stress-related circuitry. J Comp Neurol 495(2):236–254. doi:10.1002/cne.20872
Singru PS, Sanchez E, Fekete C, Lechan RM (2007) Importance of melanocortin signaling in refeeding-induced neuronal activation and satiety. Endocrinology 148(2):638–646. doi:10.1210/en.2006-1233
Stachniak TJ, Ghosh A, Sternson SM (2014) Chemogenetic synaptic silencing of neural circuits localizes a hypothalamus → midbrain pathway for feeding behavior. Neuron 82(4):797–808. doi:10.1016/j.neuron.2014.04.008
Stanley BG, Urstadt KR, Charles JR, Kee T (2011) Glutamate and GABA in lateral hypothalamic mechanisms controlling food intake. Physiol Behav 104(1):40–46. doi:10.1016/j.physbeh.2011.04.046
Ste Marie L, Miura GI, Marsh DJ, Yagaloff K, Palmiter RD (2000) A metabolic defect promotes obesity in mice lacking melanocortin-4 receptors. Proc Natl Acad Sci USA 97(22):12339–12344. doi:10.1073/pnas.220409497
Stratford JM, Finger TE (2011) Central representation of postingestive chemosensory cues in mice that lack the ability to taste. J Neurosci Off J Soc Neurosci 31(25):9101–9110. doi:10.1523/jneurosci.0404-11.2011
Stratford TR, Wirtshafter D (2013) Injections of muscimol into the paraventricular thalamic nucleus, but not mediodorsal thalamic nuclei, induce feeding in rats. Brain Res 1490:128–133. doi:10.1016/j.brainres.2012.10.043
Swanson LW (2000) Cerebral hemisphere regulation of motivated behavior. Brain Res 886(1–2):113–164
Timofeeva E, Picard F, Duclos M, Deshaies Y, Richard D (2002) Neuronal activation and corticotropin-releasing hormone expression in the brain of obese (fa/fa) and lean (fa/?) Zucker rats in response to refeeding. Eur J Neurosci 15(6):1013–1029
Turenius CI, Htut MM, Prodon DA, Ebersole PL, Ngo PT, Lara RN, Wilczynski JL, Stanley BG (2009) GABA(A) receptors in the lateral hypothalamus as mediators of satiety and body weight regulation. Brain Res 1262:16–24. doi:10.1016/j.brainres.2009.01.016
Wirth MM, Olszewski PK, Yu C, Levine AS, Giraudo SQ (2001) Paraventricular hypothalamic alpha-melanocyte-stimulating hormone and MTII reduce feeding without causing aversive effects. Peptides 22(1):129–134
Wise RA (1974) Lateral hypothalamic electrical stimulation: does it make animals ‘hungry’? Brain Res 67(2):187–209
Wittmann G, Hrabovszky E, Lechan RM (2013) Distinct glutamatergic and GABAergic subsets of hypothalamic pro-opiomelanocortin neurons revealed by in situ hybridization in male rats and mice. J Comp Neurol 521(14):3287–3302. doi:10.1002/cne.23350
Woods SC, D’Alessio DA (2008) Central control of body weight and appetite. J Clin Endocrinol Metab 93(11 Suppl 1):S37–S50. doi:10.1210/jc.2008-1630
Wu Q, Lemus MB, Stark R, Bayliss JA, Reichenbach A, Lockie SH, Andrews ZB (2014) The temporal pattern of cfos activation in hypothalamic, cortical, and brainstem nuclei in response to fasting and refeeding in male mice. Endocrinology 155(3):840–853. doi:10.1210/en.2013-1831
Zseli G, Vida B, Martinez A, Lechan RM, Khan AM, Fekete C (2016) Elucidation of the anatomy of a satiety network: focus on connectivity of the parabrachial nucleus in the adult rat. J Comp Neurol 524(14):2803–2827. doi:10.1002/cne.23992
Acknowledgements
The authors are grateful for Vera Maruzs and Ágnes Simon for the expert technical assistant. This work was supported by Grants from the Hungarian Science Foundation (OTKA K109710), Hungarian National Brain Research Program, Lendület Award of the Hungarian Academy of Sciences, the Dr. Gerald J. and Dorothy R. Friedman New York Foundation for Medical Research and the NIDDK HD087958 Grant.
Author information
Authors and Affiliations
Contributions
GZ: anterograde tract tracing, comparing c-Fos immunoreactivity in fasted and refed animals in order to choose the areas of interest, DREADD virus injection, c-Fos analysis of DREADD virus-injected animals, immunohistochemistry, image preparation and analysis. BV: Retrograde tract tracing, DREADD virus injection, immunohistochemistry, image analysis. AS-S: analyses of feeding behavior. MT: analyses of feeding behavior. RML: data interpretation, manuscript preparation. CF: experimental design, data acquisition, drafting, revising and interpretation of the work, takes primary responsibility.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were reviewed and approved by the Animal Welfare Committee of the Institute of Experimental Medicine, Hungarian Academy of Sciences, Budapest, Hungary.
Rights and permissions
About this article
Cite this article
Zséli, G., Vida, B., Szilvásy-Szabó, A. et al. Neuronal connections of the central amygdalar nucleus with refeeding-activated brain areas in rats. Brain Struct Funct 223, 391–414 (2018). https://doi.org/10.1007/s00429-017-1501-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1501-4