Abstract
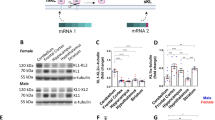
Brain-derived neurotrophic factor (BDNF) is an activity-dependent neurotrophin critical for neuronal plasticity in the hippocampus. BDNF is encoded by multiple transcripts with alternative 5′ untranslated regions (5′UTRS) that display activity-induced targeting to distinct subcellular compartments. While individual Bdnf 5′UTR transcripts influence dendrite morphology in cultured hippocampal neurons, it is unknown whether Bdnf splice variants impact dendrite arborization in functional classes of neurons in the intact hippocampus. Moreover, the contribution of Bdnf 5′UTR splice variants to dendritic spine density and shape has not been explored. We analyzed the structure of CA1 and CA3 dendrite arbors in transgenic mice lacking BDNF production from exon (Ex) 1, 2, 4, or 6 splice variants (Bdnf-e1, -e2, -e4, and -e6−/− mice) and found that loss of BDNF from individual Bdnf mRNA variants differentially impacts the complexity of apical and basal arbors in vivo. Consistent with the subcellular localization studies, Bdnf Ex2 and Ex6 transcripts significantly contributed to dendrite morphology in both CA1 and CA3 neurons. While Bdnf-e2−/− mice showed increased branching proximal to the soma in CA1 and CA3 apical arbors, Bdnf-e6−/− mice showed decreased apical and basal dendrite complexity. Analysis of spine morphology on Bdnf-e6−/− CA1 dendrites revealed changes in the percentage of differently sized spines on apical, but not basal, branches. These results provide further evidence that Bdnf splice variants generate a spatial code that mediates the local actions of BDNF in distinct dendritic compartments on structural and functional plasticity.






Similar content being viewed by others
References
Aid T, Kazantseva A, Piirsoo M, Palm K, Timmusk T (2007) Mouse and rat BDNF gene structure and expression revisited. J Neurosci Res 85:525–535 doi:10.1002/jnr.21139
Alonso M, Medina JH, Pozzo-Miller L (2004) ERK1/2 activation is necessary for BDNF to increase dendritic spine density in hippocampal CA1 pyramidal neurons. Learn Mem 11:172–178. doi:10.1101/lm.67804
An JJ et al (2008) Distinct role of long 3′ UTR BDNF mRNA in spine morphology and synaptic plasticity in hippocampal neurons. Cell 134:175–187. doi:10.1016/j.cell.2008.05.045
Baj G, Leone E, Chao MV, Tongiorgi E (2011) Spatial segregation of BDNF transcripts enables BDNF to differentially shape distinct dendritic compartments. Proc Natl Acad Sci USA 108:16813–16818. doi:10.1073/pnas.1014168108
Baj G et al (2012) Physical exercise and antidepressants enhance BDNF targeting in hippocampal CA3 dendrites: further evidence of a spatial code for BDNF splice variants Neuropsychopharmacology 37:1600–1611. doi:10.1038/npp.2012.5
Belichenko PV, Wright EE, Belichenko NP, Masliah E, Li HH, Mobley WC, Francke U (2009) Widespread changes in dendritic and axonal morphology in Mecp2-mutant mouse models of Rett syndrome: evidence for disruption of neuronal networks. J Comp Neurol 514:240–258 doi:10.1002/cne.22009
Chakravarthy S et al (2006) Postsynaptic TrkB signaling has distinct roles in spine maintenance in adult visual cortex and hippocampus. Proc Natl Acad Sci USA 103:1071–1076. doi:10.1073/pnas.0506305103
Chiaruttini C, Sonego M, Baj G, Simonato M, Tongiorgi E (2008) BDNF mRNA splice variants display activity-dependent targeting to distinct hippocampal laminae. Mol Cell Neurosci 37:11–19. doi:10.1016/j.mcn.2007.08.011
Chowdhury TG, Barbarich-Marsteller NC, Chan TE, Aoki C (2014) Activity-based anorexia has differential effects on apical dendritic branching in dorsal and ventral hippocampal CA1. Brain Struct Funct 219:1935–1945 doi:10.1007/s00429-013-0612-9
Cohen-Cory S, Kidane AH, Shirkey NJ, Marshak S (2010) Brain-derived neurotrophic factor and the development of structural neuronal connectivity. Dev Neurobiol 70:271–288. doi:10.1002/dneu.20774
Feng G et al (2000) Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28:41–51
Gao X, Smith GM, Chen J (2009) Impaired dendritic development and synaptic formation of postnatal-born dentate gyrus granular neurons in the absence of brain-derived neurotrophic factor signaling. Exp Neurol 215:178–190. doi:10.1016/j.expneurol.2008.10.009
Gourley SL, Swanson AM, Koleske AJ (2013) Corticosteroid-induced neural remodeling predicts behavioral vulnerability and resilience. J Neurosci 33:3107–3112. doi:10.1523/JNEUROSCI.2138-12.2013
Harward SC et al (2016) Autocrine BDNF-TrkB signalling within a single dendritic spine. Nature 538:99–103. doi:10.1038/nature19766
Hedrick NG, Harward SC, Hall CE, Murakoshi H, McNamara JO, Yasuda R (2016) Rho GTPase complementation underlies BDNF-dependent homo- and heterosynaptic plasticity. Nature 538:104–108. doi:10.1038/nature19784
Hill JL et al (2016) Loss of promoter IV-driven BDNF expression impacts oscillatory activity during sleep, sensory information processing and fear regulation. Transl Psychiatry 6:e873. doi:10.1038/tp.2016.153
Horch HW, Katz LC (2002) BDNF release from single cells elicits local dendritic growth in nearby neurons. Nat Neurosci 5:1177–1184 doi:10.1038/nn927
Horch HW, Kruttgen A, Portbury SD, Katz LC (1999) Destabilization of cortical dendrites and spines by BDNF. Neuron 23:353–364
Huang EJ, Reichardt LF (2001) Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci 24:677–736. doi:10.1146/annurev.neuro.24.1.677
Ji Y, Pang PT, Feng L, Lu B (2005) Cyclic AMP controls BDNF-induced TrkB phosphorylation and dendritic spine formation in mature hippocampal neurons Nature neuroscience 8:164–172 doi:10.1038/nn1381
Liu QR, Lu L, Zhu XG, Gong JP, Shaham Y, Uhl GR (2006) Rodent BDNF genes, novel promoters, novel splice variants, and regulation by cocaine. Brain Res 1067:1–12. doi:10.1016/j.brainres.2005.10.004
Lom B, Cohen-Cory S (1999) Brain-derived neurotrophic factor differentially regulates retinal ganglion cell dendritic and axonal arborization in vivo. J Neurosci 19:9928–9938
Matsutani S, Yamamoto N (2004) Brain-derived neurotrophic factor induces rapid morphological changes in dendritic spines of olfactory bulb granule cells in cultured slices through the modulation of glutamatergic signaling. Neuroscience 123:695–702
Maynard KR, Stein E (2012) DSCAM contributes to dendrite arborization and spine formation in the developing cerebral cortex. J Neurosci 32:16637–16650. doi:10.1523/JNEUROSCI.2811-12.2012
Maynard KR et al (2016) Functional role of BDNF production from unique promoters in aggression and serotonin signaling. Neuropsychopharmacology 41:1943–1955. doi:10.1038/npp.2015.349
McAllister AK, Lo DC, Katz LC (1995) Neurotrophins regulate dendritic growth in developing visual cortex. Neuron 15:791–803
McAllister AK, Katz LC, Lo DC (1997) Opposing roles for endogenous BDNF and NT-3 in regulating cortical dendritic growth. Neuron 18:767–778
McAllister AK, Katz LC, Lo DC (1999) Neurotrophins and synaptic plasticity. Annu Rev Neurosci 22:295–318. doi:10.1146/annurev.neuro.22.1.295
Murphy DD, Cole NB, Segal M (1998) Brain-derived neurotrophic factor mediates estradiol-induced dendritic spine formation in hippocampal neurons. Proc Natl Acad Sci USA 95:11412–11417
Pattabiraman PP, Tropea D, Chiaruttini C, Tongiorgi E, Cattaneo A, Domenici L (2005) Neuronal activity regulates the developmental expression and subcellular localization of cortical BDNF mRNA isoforms in vivo. Mol Cell Neurosci 28:556–570. doi:10.1016/j.mcn.2004.11.010
Pruunsild P, Kazantseva A, Aid T, Palm K, Timmusk T (2007) Dissecting the human BDNF locus: bidirectional transcription, complex splicing, and multiple promoters. Genomics 90:397–406. doi:10.1016/j.ygeno.2007.05.004
Rauskolb S et al (2010) Global deprivation of brain-derived neurotrophic factor in the CNS reveals an area-specific requirement for dendritic growth. J Neurosci 30:1739–1749. doi:10.1523/JNEUROSCI.5100-09.2010
Sathanoori M, Dias BG, Nair AR, Banerjee SB, Tole S, Vaidya VA (2004) Differential regulation of multiple brain-derived neurotrophic factor transcripts in the postnatal and adult rat hippocampus during development, and in response to kainate administration. Brain Res Mol Brain Res 130:170–177. doi:10.1016/j.molbrainres.2004.08.002
Shimada A, Mason CA, Morrison ME (1998) TrkB signaling modulates spine density and morphology independent of dendrite structure in cultured neonatal Purkinje cells. J Neurosci 18:8559–8570
Timmusk T, Palm K, Metsis M, Reintam T, Paalme V, Saarma M, Persson H (1993) Multiple promoters direct tissue-specific expression of the rat BDNF gene. Neuron 10:475–489
Tolwani RJ, Buckmaster PS, Varma S, Cosgaya JM, Wu Y, Suri C, Shooter EM (2002) BDNF overexpression increases dendrite complexity in hippocampal dentate gyrus. Neuroscience 114:795–805
Tongiorgi E (2008) Activity-dependent expression of brain-derived neurotrophic factor in dendrites: facts and open questions. Neurosci Res 61:335–346. doi:10.1016/j.neures.2008.04.013
Tongiorgi E, Righi M, Cattaneo A (1997) Activity-dependent dendritic targeting of BDNF and TrkB mRNAs in hippocampal neurons. J Neurosci 17:9492–9505
Tongiorgi E et al (2004) Brain-derived neurotrophic factor mRNA and protein are targeted to discrete dendritic laminas by events that trigger epileptogenesis. J Neurosci 24:6842–6852. doi:10.1523/JNEUROSCI.5471-03.2004
Vigers AJ, Amin DS, Talley-Farnham T, Gorski JA, Xu B, Jones KR (2012) Sustained expression of brain-derived neurotrophic factor is required for maintenance of dendritic spines and normal behavior. Neuroscience 212:1–18. doi:10.1016/j.neuroscience.2012.03.031
Wirth MJ, Brun A, Grabert J, Patz S, Wahle P (2003) Accelerated dendritic development of rat cortical pyramidal cells and interneurons after biolistic transfection with BDNF and NT4/5 Development 130:5827–5838 doi:10.1242/dev.00826
Acknowledgements
We thank Dr. Daniel Hoeppner for microscopy assistance and Dr. Daniel Weinberger for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Funding for these studies was provided by the Lieber Institute for Brain Development and the National Institute for Mental Health (T32MH01533037 to KRM and RO1MH105592 to KM).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maynard, K.R., Hobbs, J.W., Sukumar, M. et al. Bdnf mRNA splice variants differentially impact CA1 and CA3 dendrite complexity and spine morphology in the hippocampus. Brain Struct Funct 222, 3295–3307 (2017). https://doi.org/10.1007/s00429-017-1405-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1405-3