Abstract
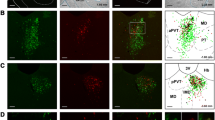
The parasubthalamic nucleus (PSTN) and the ventrally adjacent calbindin nucleus (CbN) form a nuclear complex in the posterior lateral hypothalamic area (LHA), recently characterized as connected with the central nucleus of the amygdala (CEA). The aim of the present work is to analyze in detail the projections from the amygdala into the PSTN/CbN, also focusing on pathways into the LHA. After fluorogold injections into the PSTN/CbN, the medial part of the CEA (CEAm) appears to be the main supplier of projections from the CEA. Other amygdalar nuclei contribute to the innervation of the PSTN/CbN complex, including the anterior part of the basomedial nucleus (BMAa). Injections of the anterograde tracer, Phaseolus vulgaris leucoagglutinin (PHAL), into the CEAm and BMAa revealed that projections from the CEAm follow two pathways into the LHA: a dorsal pathway formed by axons that also innervate the paraventricular hypothalamic nucleus, the anterior perifornical LHA and the PSTN, and a ventral pathway that runs laterally adjacent to the ventrolateral hypothalamic tract (vlt) and ends in the CbN. By contrast, the BMAa and other telencephalic structures, such as the fundus striatum project to the CbN via the ventral pathway. Confirming the microscopic observation, a semi-quantitative analysis of the density of these projections showed that the PSTN and the CbN are the major hypothalamic targets for the projections from the CEAm and the BMAa, respectively. PSTN and CbN receive these projections through distinct dorsal and ventral routes in the LHA. The ventral pathway forms a differentiated tract, named here the ventrolateral amygdalo-hypothalamic tract (vlah), that is distinct from, but runs adjacent to, the vlt. Both the vlt and the vlah had been previously described as forming an olfactory path into the LHA. These results help to better characterize the CbN within the PSTN/CbN complex and are discussed in terms of the functional organization of the network involving the PSTN and the CbN as well as the CEA and the BMAa.





















Similar content being viewed by others
Abbreviations
- AAA:
-
Anterior amygdalar area
- Act:
-
Anterior commissure, temporal limb
- AHN:
-
Anterior hypothalamic nucleus
- AMB:
-
Nucleus ambiguus
- ARH:
-
Arcuate hypothalamic nucleus
- B:
-
Barrington’s nucleus
- BA:
-
Bed nucleus accessory olfactory tract
- BLA:
-
Basolateral amygdalar nucleus
- BLAa:
-
Basolateral amygdalar nucleus, anterior part
- BLAp:
-
Basolateral amygdalar nucleus, posterior part
- BMAa:
-
Basomedial amygdalar nucleus, anterior part
- BMAp:
-
Basomedial amygdalar nucleus, posterior part
- BST:
-
Bed nuclei stria terminalis
- BSTad:
-
Bed nuclei stria terminalis, anterior division, anterodorsal area
- CA:
-
Ammon’s horn
- Cb:
-
Calbindin
- CbN:
-
Calbindin nucleus
- CEA:
-
Central amygdalar nucleus
- CEAc:
-
Central amygdalar nucleus, capsular part
- CEAl:
-
Central amygdalar nucleus, lateral part
- CEAm:
-
Central amygdalar nucleus, medial part
- CGRP:
-
Calcitonin gene-related peptide
- CM:
-
Central medial thalamic nucleus
- COAa:
-
Cortical amygdalar nucleus, anterior part
- COApl:
-
Cortical amygdalar nucleus, posterior part, lateral zone
- COApm:
-
Cortical amygdalar nucleus, posterior part, medial zone
- CP:
-
Caudoputamen
- cpd:
-
Cerebral peduncle
- ddp:
-
Dorsal descending pathway
- DMH:
-
Dorsomedial hypothalamic nucleus
- ENTl:
-
Entorhinal area, lateral part
- ENTm:
-
Entorhinal area, medial part, dorsal zone
- EPd:
-
Endopiriform nucleus, dorsal part
- EPv:
-
Endopiriform nucleus, ventral part
- FG:
-
Fluorogold
- FS:
-
Fundus striatum
- fx:
-
Columns of the fornix
- GABA:
-
Gamma-aminobutyric acid
- GAD:
-
Glutamic acid decarboxylase
- GP:
-
Globus pallidus
- GRN:
-
Gigantocellular reticular nucleus
- IA:
-
Intercaleted amygdalar nuclei
- ic:
-
Internal capsule
- IC:
-
Insular cortex
- LA:
-
Lateral amygdalar nucleus
- LDT:
-
Laterodorsal tegmental nucleus
- LHA:
-
Lateral hypothalamic area
- LHApfx:
-
Lateral hypothalamic area, perifonical region
- LHAvl:
-
Lateral hypothalamic area, ventral region, lateral zone
- LRN:
-
Lateral reticular nucleus
- LS:
-
Lateral septal nucleus
- MA:
-
Magnocellular preoptic nucleus
- MCH:
-
Melanin-concentrating hormone
- MD:
-
Mediodorsal thalamic nucleus
- MDRN:
-
Medullary reticular nucleus
- MEA:
-
Medial amygdalar nucleus
- MEApv:
-
Medial amygdalar nucleus, posteroventral part
- mfb:
-
Medial forebrain bundle
- MG:
-
Medial geniculate complex
- MRN:
-
Mesencephalic reticular nucleus
- MS:
-
Medial septal nucleus
- mtt:
-
Mammillothalamic tract
- NDB:
-
Diagonal band nucleus
- NG:
-
Nucleus gemini
- NLOT:
-
Nucleus of the lateral olfactory tract
- NTS:
-
Nucleus of the solitary tract
- opt:
-
Optic tract
- OT:
-
Olfactory tubercle
- PA:
-
Posterior amygdalar nucleus
- PAG:
-
Periaqueductal gray
- PARN:
-
Parvicellular reticular nucleus
- PB:
-
Parabrachial nucleus
- PH:
-
Posterior hypothalamic nucleus
- PHAL:
-
Phaseolus vulgaris leucoagglutinin
- PIR:
-
Piriform area
- PMv:
-
Ventral premammillary nucleus
- POR:
-
Periolivary region
- PRN:
-
Pontine reticular nucleus
- PV:
-
Parvalbumin
- PV1/parvafox:
-
Parvalbumin-positive (PV1) nucleus/parvafox nucleus
- PVH:
-
Paraventricular hypothalamic nucleus
- PVHlp-f:
-
Paraventricular hypothalamic nucleus, lateral parvicellular part–forniceal part
- PVT:
-
Paraventricular thalamic nucleus
- PSTN:
-
Parasubthalamic nucleus
- PSV:
-
Principal sensory nucleus of the trigeminal
- RE:
-
Nucleus reuniens
- RH:
-
Rhomboid nucleus
- RM:
-
Nucleus raphe magnus
- SC:
-
Superior colliculus
- SI:
-
Substantia innominate
- sm:
-
Stria medullaris
- SN:
-
Substantia nigra
- SO:
-
Supraoptic nucleus
- SPVI:
-
Spinal nucleus of the trigeminal, interpolar part
- st:
-
Stria terminalis
- STN:
-
Subthalamic nucleus
- SUM:
-
Supramammillary nucleus
- Sup:
-
Supraoptic commissures
- TU:
-
Tuberal nucleus
- V:
-
Motor nucleus of the trigeminal nerve
- VII:
-
Facial nucleus
- vlah:
-
Ventrolateral amygdalo-hypothalamic pathway
- vlt:
-
Ventrolateral hypothalamic tract
- VMH:
-
Ventromedial hypothalamic nucleus
- VP:
-
Ventral pallidum
- VTA:
-
Ventral tegmental area
- ZI:
-
Zona incerta
References
Allen GV, Cechetto DF (1995) Neurotensin in the lateral hypothalamic area: origin and function. Neuroscience 69:533–544
Alvarez-Bolado G, Celio MR (2016) The ventrolateral hypothalamic area and the parvafox nucleus: role in the expression of (positive) emotions? J Comp Neurol 524:1616–1623. doi:10.1002/cne.23853
Bernard J-F, Alden M, Besson J-M (1993) The organization of the efferent projections from the pontine parabrachial area to the amygdaloid complex: a Phaseolus vulgaris leucoagglutinin (PHA-L) study in the rat. J Comp Neurol 329:201–229. doi:10.1002/cne.903290205
Bester H, Besson JM, Bernard JF (1997) Organization of efferent projections from the parabrachial area to the hypothalamus: a Phaseolus vulgaris-leucoagglutinin study in the rat. J Comp Neurol 383:245–281
Bienkowski MS, Rinaman L (2013) Common and distinct neural inputs to the medial central nucleus of the amygdala and anterior ventrolateral bed nucleus of stria terminalis in rats. Brain Struct Funct 218:187–208. doi:10.1007/s00429-012-0393-6
Bilella A, Alvarez-Bolado G, Celio MR (2014) Coaxiality of Foxb1- and parvalbumin-expressing neurons in the lateral hypothalamic PV1-nucleus. Neurosci Lett 566:111–114. doi:10.1016/j.neulet.2014.02.028
Bilella A, Alvarez-Bolado G, Celio MR (2016a) Birthdate of parvalbumin-neurons in the Parvafox-nucleus of the lateral hypothalamus. Brain Res 1633:111–114. doi:10.1016/j.brainres.2015.12.054
Bilella A, Alvarez-Bolado G, Celio MR (2016b) The Foxb1-expressing neurons of the ventrolateral hypothalamic parvafox nucleus project to defensive circuits: Foxb1-expressing neurons of the VL hypothalamic Parvafox nucleus. J Comp Neurol 524:2955–2981. doi:10.1002/cne.24057
Bourgeais L, Gauriau C, Bernard JF (2001) Projections from the nociceptive area of the central nucleus of the amygdala to the forebrain: a PHA-L study in the rat. Eur J Neurosci 14:229–255
Cai H, Haubensak W, Anthony TE, Anderson DJ (2014) Central amygdala PKC-δ+ neurons mediate the influence of multiple anorexigenic signals. Nat Neurosci 17:1240–1248. doi:10.1038/nn.3767
Campos CA, Bowen AJ, Schwartz MW, Palmiter RD (2016) Parabrachial CGRP neurons control meal termination. Cell Metab 23:811–820. doi:10.1016/j.cmet.2016.04.006
Canteras NS, Simerly RB, Swanson LW (1992) Connections of the posterior nucleus of the amygdala. J Comp Neurol 324:143–179. doi:10.1002/cne.903240203
Canteras NS, Simerly RB, Swanson LW (1995) Organization of projections from the medial nucleus of the amygdala: a PHAL study in the rat. J Comp Neurol 360:213–245. doi:10.1002/cne.903600203
Carter ME, Soden ME, Zweifel LS, Palmiter RD (2013) Genetic identification of a neural circuit that suppresses appetite. Nature 503:111–114. doi:10.1038/nature12596
Carter ME, Han S, Palmiter RD (2015) Parabrachial calcitonin gene-related peptide neurons mediate conditioned taste aversion. J Neurosci 35:4582–4586. doi:10.1523/JNEUROSCI.3729-14.2015
Carvalho MC, Santos JM, Brandão ML (2015) Dorsal periaqueductal gray post-stimulation freezing is counteracted by neurokinin-1 receptor antagonism in the central nucleus of the amygdala in rats. Neurobiol Learn Mem 121:52–58. doi:10.1016/j.nlm.2015.04.001
Cassell MD, Gray TS, Kiss JZ (1986) Neuronal architecture in the rat central nucleus of the amygdala: a cytological, hodological, and immunocytochemical study. J Comp Neurol 246:478–499. doi:10.1002/cne.902460406
Celio MR (1990) Calbindin D-28k and parvalbumin in the rat nervous system. Neuroscience 35:375–475
Celio MR, Babalian A, Ha QH et al (2013) Efferent connections of the parvalbumin-positive (PV1) nucleus in the lateral hypothalamus of rodents. J Comp Neurol 521:3133–3153. doi:10.1002/cne.23344
Chometton S, Pedron S, Peterschmitt Y, et al (2016) A premammillary lateral hypothalamic nuclear complex responds to hedonic but not aversive tastes in the male rat. Brain Struct Funct 221:2183–2208. doi:10.1007/s00429-015-1038-3
Croizier S, Cardot J, Brischoux F et al (2013) The vertebrate diencephalic MCH system: a versatile neuronal population in an evolving brain. Front Neuroendocrinol 34:65–87. doi:10.1016/j.yfrne.2012.10.001
D’Hanis W, Linke R, Yilmazer-Hanke DM (2007) Topography of thalamic and parabrachial calcitonin gene-related peptide (CGRP) immunoreactive neurons projecting to subnuclei of the amygdala and extended amygdala. J Comp Neurol 505:268–291. doi:10.1002/cne.21495
Dobolyi A, Irwin S, Makara G et al (2005) Calcitonin gene-related peptide-containing pathways in the rat forebrain. J Comp Neurol 489:92–119. doi:10.1002/cne.20618
Goforth PB, Leinninger GM, Patterson CM et al (2014) leptin acts via lateral hypothalamic area neurotensin neurons to inhibit orexin neurons by multiple GABA-independent mechanisms. J Neurosci 34:11405–11415. doi:10.1523/JNEUROSCI.5167-13.2014
Gonzales C, Chesselet MF (1990) Amygdalonigral pathway: an anterograde study in the rat with Phaseolus vulgaris leucoagglutinin (PHA-L). J Comp Neurol 297:182–200. doi:10.1002/cne.902970203
Goto M, Swanson LW (2004) Axonal projections from the parasubthalamic nucleus. J Comp Neurol 469:581–607. doi:10.1002/cne.11036
Gray TS, Carney ME, Magnuson DJ (1989) Direct projections from the central amygdaloid nucleus to the hypothalamic paraventricular nucleus: possible role in stress-induced adrenocorticotropin release. Neuroendocrinology 50:433–446
Groenewegen HJ, Berendse HW, Haber SN (1993) Organization of the output of the ventral striatopallidal system in the rat: ventral pallidal efferents. Neuroscience 57:113–142
Grove EA (1988a) Efferent connections of the substantia innominata in the rat. J Comp Neurol 277:347–364. doi:10.1002/cne.902770303
Grove EA (1988b) Neural associations of the substantia innominata in the rat: afferent connections. J Comp Neurol 277:315–346. doi:10.1002/cne.902770302
Hahn JD, Swanson LW (2012) Connections of the lateral hypothalamic area juxtadorsomedial region in the male rat. J Comp Neurol 520:1831–1890. doi:10.1002/cne.23064
Han S, Soleiman MT, Soden ME et al (2015) Elucidating an affective pain circuit that creates a threat memory. Cell 162:363–374. doi:10.1016/j.cell.2015.05.057
Heimer L, Zahm DS, Schmued LC (1990) The basal forebrain projection to the region of the nuclei gemini in the rat; a combined light and electron microscopic study employing horseradish peroxidase, fluorescent tracers and Phaseolus vulgaris-leucoagglutinin. Neuroscience 34:707–731
Heimer L, Harlan RE, Alheid GF et al (1997) Substantia innominata: a notion which impedes clinical-anatomical correlations in neuropsychiatric disorders. Neuroscience 76:957–1006
Krettek JE, Price JL (1978) Amygdaloid projections to subcortical structures within the basal forebrain and brainstem in the rat and cat. J Comp Neurol 178:225–253. doi:10.1002/cne.901780204
Lantos TA, Görcs TJ, Palkovits M (1995) Immunohistochemical mapping of neuropeptides in the premamillary region of the hypothalamus in rats. Brain Res Brain Res Rev 20:209–249
Larsen PJ (1992) Distribution of substance P-immunoreactive elements in the preoptic area and the hypothalamus of the rat. J Comp Neurol 316:287–313. doi:10.1002/cne.903160304
Leinninger GM, Jo Y-H, Leshan RL et al (2009) Leptin acts via leptin receptor-expressing lateral hypothalamic neurons to modulate the mesolimbic dopamine system and suppress feeding. Cell Metab 10:89–98. doi:10.1016/j.cmet.2009.06.011
Leinninger GM, Opland DM, Jo Y-H et al (2011) Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab 14:313–323. doi:10.1016/j.cmet.2011.06.016
Leonard CM, Scott JW (1971) Origin and distribution of the amygdalofugal pathways in the rat: an experimental neuronatomical study. J Comp Neurol 141:313–329. doi:10.1002/cne.901410304
Liang S-H, Yin J-B, Sun Y et al (2016) Collateral projections from the lateral parabrachial nucleus to the paraventricular thalamic nucleus and the central amygdaloid nucleus in the rat. Neurosci Lett 629:245–250. doi:10.1016/j.neulet.2016.07.017
McDonald AJ (1982) Cytoarchitecture of the central amygdaloid nucleus of the rat. J Comp Neurol 208:401–418. doi:10.1002/cne.902080409
Mészár Z, Girard F, Saper CB, Celio MR (2012) The lateral hypothalamic parvalbumin-immunoreactive (PV1) nucleus in rodents. J Comp Neurol 520:798–815. doi:10.1002/cne.22789
Millhouse OE (1969) A Golgi study of the desending medial forebrain bundle. Brain Res 15:341–363
Millhouse OE, Uemura-Sumi M (1985) The structure of the nucleus of the lateral olfactory tract. J Comp Neurol 233:517–552. doi:10.1002/cne.902330411
Nakamura S, Tsumori T, Yokota S et al (2009) Amygdaloid axons innervate melanin-concentrating hormone- and orexin-containing neurons in the mouse lateral hypothalamus. Brain Res 1278:66–74. doi:10.1016/j.brainres.2009.04.049
Niu J-G, Yokota S, Tsumori T et al (2012) Projections from the anterior basomedial and anterior cortical amygdaloid nuclei to melanin-concentrating hormone-containing neurons in the lateral hypothalamus of the rat. Brain Res 1479:31–43. doi:10.1016/j.brainres.2012.08.011
Norgren R (1976) Taste pathways to hypothalamus and amygdala. J Comp Neurol 166:17–30. doi:10.1002/cne.901660103
Oka T, Tsumori T, Yokota S, Yasui Y (2008) Neuroanatomical and neurochemical organization of projections from the central amygdaloid nucleus to the nucleus retroambiguus via the periaqueductal gray in the rat. Neurosci Res 62:286–298. doi:10.1016/j.neures.2008.10.004
Ono T, Luiten PG, Nishijo H et al (1985) Topographic organization of projections from the amygdala to the hypothalamus of the rat. Neurosci Res 2:221–238
Ottersen OP (1980) Afferent connections to the amygdaloid complex of the rat and cat: II. Afferents from the hypothalamus and the basal telencephalon. J Comp Neurol 194:267–289. doi:10.1002/cne.901940113
Pape HC, Pare D (2010) Plastic synaptic networks of the amygdala for the acquisition, expression, and extinction of conditioned fear. Physiol Rev 90:419–463. doi:10.1152/physrev.00037.2009
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates†, 5th ed. Elsevier Academic Press, Amsterdam
Petrovich GD, Swanson LW (1997) Projections from the lateral part of the central amygdalar nucleus to the postulated fear conditioning circuit. Brain Res 763:247–254
Petrovich GD, Risold PY, Swanson LW (1996) Organization of projections from the basomedial nucleus of the amygdala: a PHAL study in the rat. J Comp Neurol 374:387–420. doi:10.1002/(SICI)1096-9861(19961021)374:3<387::AID-CNE6>3.0.CO;2-Y
Petrovich GD, Ross CA, Mody P et al (2009) Central, but not basolateral, amygdala is critical for control of feeding by aversive learned cues. J Neurosci 29:15205–15212. doi:10.1523/JNEUROSCI.3656-09.2009
Price JL (1985) Beyond the primary olfactory cortex: olfactory-related areas in the neocortex, thalamus and hypothalamus. Chem Senses 10:239–258. doi:10.1093/chemse/10.2.239
Price JL, Slotnick BM, Revial M-F (1991) Olfactory projections to the hypothalamus. J Comp Neurol 306:447–461. doi:10.1002/cne.903060309
Reppucci CJ, Petrovich GD (2016) Organization of connections between the amygdala, medial prefrontal cortex, and lateral hypothalamus: a single and double retrograde tracing study in rats. Brain Struct Funct 221:2937–2962. doi:10.1007/s00429-015-1081-0
Risold PY, Fellmann D, Rivier J et al (1992) Immunoreactivities for antisera to three putative neuropeptides of the rat melanin-concentrating hormone precursor are coexpressed in neurons of the rat lateral dorsal hypothalamus. Neurosci Lett 136:145–149
Risold PY, Thompson RH, Swanson LW (1997) The structural organization of connections between hypothalamus and cerebral cortex. Brain Res Rev 24:197–254. doi:10.1016/S0165-0173(97)00007-6
Santiago AC, Shammah-Lagnado SJ (2004) Efferent connections of the nucleus of the lateral olfactory tract in the rat. J Comp Neurol 471:314–332. doi:10.1002/cne.20028
Schwaber JS, Sternini C, Brecha NC et al (1988) Neurons containing calcitonin gene-related peptide in the parabrachial nucleus project to the central nucleus of the amygdala. J Comp Neurol 270(416–426):398–399
Scott JW, Chafin BR (1975) Origin of olfactory projections to lateral hypothalamus and nuclei gemini of the rat. Brain Res 88:64–68
Scott JW, Leonard CM (1971) The olfactory connections of the lateral hypothalamus in the rat, mouse and hamster. J Comp Neurol 141:331–344. doi:10.1002/cne.901410305
Scott JW, Pfaffmann C (1967) Olfactory input to the hypothalamus: electrophysiological evidence. Science 158:1592–1594
Shammah-Lagnado SJ, Alheid GF, Heimer L (2001) Striatal and central extended amygdala parts of the interstitial nucleus of the posterior limb of the anterior commissure: evidence from tract-tracing techniques in the rat. J Comp Neurol 439:104–126. doi:10.1002/cne.1999
Shimada S, Shiosaka S, Emson PC et al (1985) Calcitonin gene-related peptidergic projection from the parabrachial area to the forebrain and diencephalon in the rat: an immunohistochemical analysis. Neuroscience 16:607–616. doi:10.1016/0306-4522(85)90195-2
Shin J-W, Geerling JC, Stein MK et al (2011) FoxP2 brainstem neurons project to sodium appetite regulatory sites. J Chem Neuroanat 42:1–23. doi:10.1016/j.jchemneu.2011.05.003
Shirasu M, Takahashi T, Yamamoto T et al (2011) Direct projections from the central amygdaloid nucleus to the mesencephalic trigeminal nucleus in rats. Brain Res 1400:19–30. doi:10.1016/j.brainres.2011.05.026
Spooren WP, Veening JG, Groenewegen HJ, Cools AR (1991) Efferent connections of the striatopallidal and amygdaloid components of the substantia innominata in the cat: projections to the nucleus accumbens and caudate nucleus. Neuroscience 44:431–447
Swanson LW (2000) Cerebral hemisphere regulation of motivated behavior. Brain Res 886:113–164
Swanson LW (2004) Brain maps: structure of the rat brain, 3 edn. Elsevier, San Diego
Swanson LW (2012) Brain architecture: understanding the basic plan. Oxford University Press
Swanson LW, Petrovich GD (1998) What is the amygdala? Trends Neurosci 21:323–331
Tsumori T, Yokota S, Qin Y et al (2006) A light and electron microscopic analysis of the convergent insular cortical and amygdaloid projections to the posterior lateral hypothalamus in the rat, with special reference to cardiovascular function. Neurosci Res 56:261–269. doi:10.1016/j.neures.2006.07.005
Veening JG, Swanson LW, Cowan WM et al (1982) The medial forebrain bundle of the rat. II. An autoradiographic study of the topography of the major descending and ascending components. J Comp Neurol 206:82–108. doi:10.1002/cne.902060107
Veening JG, Swanson LW, Sawchenko PE (1984) The organization of projections from the central nucleus of the amygdala to brainstem sites involved in central autonomic regulation: a combined retrograde transport-immunohistochemical study. Brain Res 303:337–357
Veening JG, Te Lie S, Posthuma P et al (1987) A topographical analysis of the origin of some efferent projections from the lateral hypothalamic area in the rat. Neuroscience 22:537–551
Wallace DM, Magnuson DJ, Gray TS (1992) Organization of amygdaloid projections to brainstem dopaminergic, noradrenergic, and adrenergic cell groups in the rat. Brain Res Bull 28:447–454
Wang PY, Zhang FC (1995) Outline and atlas of learning rat brain slices. Westnorth University Press, China
Yasoshima Y, Scott TR, Yamamoto T (2006) Memory-dependent c-Fos expression in the nucleus accumbens and extended amygdala following the expression of a conditioned taste aversive in the rat. Neuroscience 141:35–45. doi:10.1016/j.neuroscience.2006.03.019
Yu K, Garcia da Silva P, Albeanu DF, Li B (2016) Central amygdala somatostatin neurons gate passive and active defensive behaviors. J Neurosci 36:6488–6496. doi:10.1523/JNEUROSCI.4419-15.2016
Zahm DS, Williams E, Wohltmann C (1996) Ventral striatopallidothalamic projection: IV. Relative involvements of neurochemically distinct subterritories in the ventral pallidum and adjacent parts of the rostroventral forebrain. J Comp Neurol 364:340–362. doi:10.1002/(SICI)1096-9861(19960108)364:2<340::AID-CNE11>3.0.CO;2-T
Zséli G, Vida B, Martinez A et al (2016) Elucidation of the anatomy of a satiety network: focus on connectivity of the parabrachial nucleus in the adult rat: refeeding-activated contacts of the PB. J Comp Neurol 524:2803–2827. doi:10.1002/cne.23992
Acknowledgements
This work was supported by the Region Franche-Comté, France. Authors wish to thank C. Houdayer and G. Franchi for technical assistances.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Barbier, M., Chometton, S., Peterschmitt, Y. et al. Parasubthalamic and calbindin nuclei in the posterior lateral hypothalamus are the major hypothalamic targets for projections from the central and anterior basomedial nuclei of the amygdala. Brain Struct Funct 222, 2961–2991 (2017). https://doi.org/10.1007/s00429-017-1379-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1379-1