Abstract
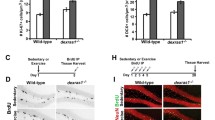
Adult neurogenesis occurs throughout life in the dentate gyrus (DG) and the subventricular zone (SVZ), where glia-like stem cells generate new neurons. Voluntary running is a powerful neurogenic stimulus triggering the proliferation of progenitor cells in the DG but, apparently, not in the SVZ. The antiproliferative gene Btg1 maintains the quiescence of DG and SVZ stem cells. Its ablation causes intense proliferation of DG and SVZ stem/progenitor cells in young mice, followed, during adulthood, by progressive decrease of the proliferative capacity. We have previously observed that running can rescue the deficit of DG Btg1-null neurogenesis. Here, we show that in adult Btg1-null SVZ stem and neuroblast cells, the reduction of proliferation is associated with a longer cell cycle and a more frequent entry into quiescence. Notably, running increases proliferation in Btg1-null SVZ stem cells highly above the levels of sedentary wild-type mice and restores normal values of cell cycle length and quiescence in stem and neuroblast cells, without affecting wild-type cells. Btg1-null SVZ neuroblasts show also increased migration throughout the rostral migratory stream and a deficiency of differentiated neurons in the olfactory bulb, possibly a consequence of premature exit from the cycle; running, however, normalizes migration and differentiation, increasing newborn neurons recruited to the olfactory circuitry. Furthermore, running increases the self-renewal of Btg1-null SVZ-derived neurospheres and, remarkably, in aged Btg1-null mice almost doubles the proliferating SVZ stem cells. Altogether, this reveals that SVZ stem cells are endowed with a hidden supply of self-renewal capacity, coupled to cell cycle acceleration and emerging after ablation of the quiescence-maintaining Btg1 gene and following exercise.







Similar content being viewed by others
References
Alvarez-Buylla A, Kohwi M, Nguyen TM, Merkle FT (2008) The heterogeneity of adult neural stem cells and the emerging complexity of their niche. Cold Spring Harb Symp Quant Biol 73:357–365
American College of Sports Medicine; Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA, Minson CT, Nigg CR, Salem GJ, Skinner JS American College of Sports Medicine position stand (2009) Exercise and physical activity for older adults. Med Sci Sports Exerc 41(7):1510–1530
Bednarczyk MR, Aumont A, Décary S, Bergeron R, Fernandes KJ (2009) Prolonged voluntary wheel-running stimulates neural precursors in the hippocampus and forebrain of adult CD1 mice. Hippocampus 19(10):913–927
Berchtold NC, Chinn G, Chou M, Kesslak JP, Cotman CW (2005) Exercise primes a molecular memory for brain-derived neurotrophic factor protein induction in the rat hippocampus. Neuroscience 133(3):853–861
Beukelaers P, Vanderbosch R, Caron N, Nguyen L, Belachew S, Moonen G, Kiyokawa H, Barbacid M, Santamaria D, Malgrange B (2011) Cdk6-dependent regulation of G1 length controls adult neurogenesis. Stem Cells 29:713–724
Blackmore DG, Reynolds BA, Golmohammadi MG, Large B, Aguilar RM, Haro L, Waters MJ, Rietze RL (2012) Growth hormone responsive neural precursor cells reside within the adult mammalian brain. Sci Rep 2:250
Bolijn S, Lucassen PJ (2015) How the body talks to the brain; peripheral mediators of physical activity-induced proliferation in the adult hippocampus. Brain Plastic 1(1):15–27
Brandt MD, Hǘbner M, Storch A (2012) Brief report: adult hippocampal precursor cells shorten S-phase and total cell cycle length during neuronal differentiation. Stem Cells 30(12):2843–2847
Brown J, Cooper-Kuhn CM, Kempermann G, Van Praag H, Winkler J, Gage FH, Kuhn HG (2003) Enriched environment and physical activity stimulate hippocampal but not olfactory bulb neurogenesis. Eur J Neurosci 17(10):2042–2046
Cai L, Hayes NL, Nowakowski RS (1997) Local homogeneity of cell cycle length in developing mouse cortex. J Neurosci 17(6):2079–2087
Carleton A, Petreanu LT, Lansford R, Alvarez-Buylla A, Lledo PM (2003) Becoming a new neuron in the adult olfactory bulb. Nat Neurosci 6(5):507–518
Ceccarelli M, Micheli L, D’Andrea G, De Bardi M, Scheijen B, Ciotti M, Leonardi L, Luvisetto S, Tirone F (2015) Altered cerebellum development and impaired motor coordination in mice lacking the Btg1 gene: involvement of cyclin D1. Dev Biol 408(1):109–125
Christie KJ, Turnley AM (2013) Regulation of endogenous neural stem/progenitor cells for neural repair-factors that promote neurogenesis and gliogenesis in the normal and damaged brain. Front Cell Neurosci 6:70
Colak D, Mori T, Brill MS, Pfeifer A, Falk S, Deng C, Monteiro R, Mummery C, Sommer L, Götz M (2008) Adult neurogenesis requires Smad4-mediated bone morphogenic protein signaling in stem cells. J Neurosci 28(2):434–446
Danzer SC (2012) Depression, stress, epilepsy and adult neurogenesis. Exp Neurol 233(1):22–32
Dietrich MO, Andrews ZB, Horvath TL (2008) Exercise-induced synaptogenesis in the hippocampus is dependent on UCP2-regulated mitochondrial adaptation. J Neurosci 28(42):10766–10771
Dimitrov EL, Tsuda MC, Cameron HA, Usdin TB (2014) Anxiety- and depression-like behavior and impaired neurogenesis evoked by peripheral neuropathy persist following resolution of prolonged tactile hypersensitivity. J Neurosci 34(37):12304–12312
Doetsch F, Caillé I, Lim DA, García-Verdugo JM, Alvarez-Buylla A (1999) Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97(6):703–716
Fabel K, Kempermann G (2008) Physical activity and the regulation of neurogenesis in the adult and aging brain. Neuromol Med 10(2):59–66
Farioli-Vecchioli S, Tirone F (2015) Control of cell cycle in adult neurogenesis and its relation with physical exercise. Brain Plastic 1:41–54
Farioli-Vecchioli S, Micheli L, Saraulli D, Ceccarelli M, Cannas S, Scardigli R, Leonardi L, Cinà I, Costanzi M, Ciotti MT, Moreira P, Rouault JP, Cestari V, Tirone F (2012) Btg1 is required to maintain the pool of stem and progenitor cells of the dentate gyrus and subventricular zone. Front Neurosci 6:124
Farioli-Vecchioli S, Mattera A, Micheli L, Ceccarelli M, Leonardi L, Saraulli D, Costanzi M, Cestari V, Rouault JP, Tirone F (2014) Running rescues defective adult neurogenesis by shortening the length of the cell cycle of neural stem and progenitor cells. Stem Cells 32(7): 1968–1982
Fischer TJ, Walker TL, Overall RW, Brandt MD, Kempermann G (2014) Acute effects of wheel running on adult hippocampal precursor cells in mice are not caused by changes in cell cycle length or S phase length. Front Neurosci 8:314
Gomez-Pinilla F, Vaynman S, Ying Z (2008) Brain-derived neurotrophic factor functions as a metabotrophin to mediate the effects of exercise on cognition. Eur J Neurosci 28(11):2278–2287
Imura T, Kornblum HI, Sofroniew MV (2003) The predominant neural stem cell isolated from postnatal and adult forebrain but not early embryonic forebrain expresses. GFAP J Neurosci 23(7):2824–2832
Intlekofer KA, Berchtold NC, Malvaez M, Carlos AJ, McQuown SC, Cunningham MJ, Wood MA, Cotman CW (2013) Exercise and sodium butyrate transform a subthreshold learning event into long-term memory via a brain-derived neurotrophic factor-dependent mechanism. Neuropsychopharmacology 38(10):2027–2034
Itoh T, Imano M, Nishida S, Tsubaki M, Hashimoto S, Ito A, Satou T (2011) Exercise increases neural stem cell proliferation surrounding the area of damage following rat traumatic brain injury. J Neural Transm (Vienna) 118(2):193–202
Kippin TE, Martens DJ, van der Kooy D (2005) p21 loss compromises the relative quiescence of forebrain stem cell proliferation leading to exhaustion of their proliferation capacity. Genes Dev 19(6):756–767
Kohwi M, Osumi N, Rubenstein JL, Alvarez-Buylla A (2005) Pax6 is required for making specific subpopulations of granule and periglomerular neurons in the olfactory bulb. J Neurosci 25(30):6997–7003
Kosaka K, Aika Y, Toida K, Heizmann CW, Hunziker W, Jacobowitz DM, Nagatsu I, Streit P, Visser TJ, Kosaka T (1995) Chemically defined neuron groups and their subpopulations in the glomerular layer of the rat main olfactory bulb. Neurosci Res 23(1):73–88
Kronenberg G, Bick-Sander A, Bunk E, Wolf C, Ehninger D, Kempermann G (2006) Physical exercise prevents age-related decline in precursor cell activity in the mouse dentate gyrus. Neurobiol Aging 27(10):1505–1513
Laywell ED, Rakic P, Kukekov VG, Holland EC, Steindler DA (2009) Identification of a multipotent astrocytic stem cell in the immature and adult mouse brain. Proc Natl Acad Sci USA 97(25):13883–13888
Lee JC, Yau SY, Lee TM, Lau BW, So KF (2016) Voluntary wheel running reverses the decrease in subventricular zone neurogenesis caused by corticosterone. Cell Transpl. doi:10.3727/096368916X692195
Lim DA, Alvarez-Buylla A (2016) The adult ventricular-subventricular zone (V-SVZ) and olfactory bulb (OB) neurogenesis. Cold Spring Harb Perspect Biol 8(5)
Lin TW, Chen SJ, Huang TY, Chang CY, Chuang JI, Wu FS, Kuo YM, Jen CJ (2012) Different types of exercise induce differential effects on neuronal adaptations and memory performance. Neurobiol Learn Mem 97(1):140–147
Luo J, Daniels SB, Lennington JB, Notti RQ, Conover JC (2006) The aging neurogenic subventricular zone. Aging Cell 5(2):139–152
Molofsky AV, He S, Bydon M, Morrison SJ, Pardal R (2005) Bmi-1 promotes neural stem cell self-renewal and neural development but not mouse growth and survival by repressing the p16Ink4a and p19Arf senescence pathways. Genes Dev 19(12):1432–1437
Molofsky AV, Slutsky SG, Joseph NM, He S, Pardal R, Krishnamurthy J, Sharpless NE, Morrison SJ (2006) Increasing p16INK4a expression decreases forebrain progenitors and neurogenesis during ageing. Nature 443(7110):448–452
Niwa A, Nishibori M, Hamasaki S, Kobori T, Liu K, Wake H, Mori S, Yoshino T, Takahashi H (2016) Voluntary exercise induces neurogenesis in the hypothalamus and ependymal lining of the third ventricle. Brain Struct Funct 221(3):1653–1666
Olson AK, Eadie BD, Ernst C, Christie BR (2006) Environmental enrichment and voluntary exercise massively increase neurogenesis in the adult hippocampus via dissociable pathways. Hippocampus 16(3):250–260
Overall RW, Walker TL, Fischer TJ, Brandt MD, Kempermann G (2016) Different mechanisms must be considered to explain the increase in hippocampal neural precursor cell proliferation by physical activity. Front Neurosci 10:362
Paillard T, Rolland Y, de Souto Barreto P (2015) Protective effects of physical exercise in Alzheimer’s disease and Parkinson’s disease: a narrative review J Clin Neurol 11(3):212–219
Patten AR, Sickmann H, Hryciw BN, Kucharsky T, Parton R, Kernick A, Christie BR (2013) Long-term exercise is needed to enhance synaptic plasticity in the hippocampus. Learn Mem 20(11):642–647
Platel JC, Bordey A (2016) The multifaceted subventricular zone astrocyte: From a metabolic and pro-neurogenic role to acting as a neural stem cell. Neuroscience 323:20–28
Ruan L, Lau BW, Wang J, Huang L, Zhuge Q, Wang B, Jin K, So KF (2014) Neurogenesis in neurological and psychiatric diseases and brain injury: from bench to bedside. Prog Neurobiol 115:116–137
Ryan SM, Kelly ÁM (2016) Exercise as a pro-cognitive. Pro-neurogenic anti-inflammatory intervention in transgenic mouse models of Alzheimer’s disease. Age Res Rev 27:77–92
Stranahan AM, Zhou Y, Martin B, Maudsley S (2009) Pharmacomimetics of exercise: novel approaches for hippocampally-targeted neuroprotective agents. Curr Med Chem 16(35):4668–4678
van Praag H (2008) Neurogenesis and exercise: past and future directions. Neuromolecular Med 10(2):128–140
van Praag H, Christie BR, Sejnowski TJ, Gage FH (1999) Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc Natl Acad Sci USA 96(23):13427–13431
van Praag H, Shubert T, Zhao C, Gage FH (2005) Exercise enhances learning and hippocampal neurogenesis in aged mice. J Neurosci 25(38):8680–8685
Wichterle H, Garcia-Verdugo JM, Alvarez-Buylla A (1997) Direct evidence for homotypic, glia-independent neuronal migration. Neuron 18(5):779–791
Zhao C, Deng W, Gage FH (2008) Mechanisms and functional implications of adult neurogenesis. Cell 132(4):645–660
Zhu Y, Jin K, Mao XO, Greenberg DA (2003) Vascular endothelial growth factor promotes proliferation of cortical neuron precursors by regulating E2F expression. FASEB J 17(2):186–193
Acknowledgements
This work was supported by CNR projects DSB.AD004.093 to Felice Tirone.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors indicate no potential conflict of interests.
Additional information
Stefano Farioli-Vecchioli and Felice Tirone contributed equally.
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2017_1376_MOESM1_ESM.tif
Supplement figure 1. Volume measurement of the SVZ carried out in 2 month-old (A) and in 15 month-old mice (B) did not show any differences among the experimental conditions analyzed (TIF 238 KB)
429_2017_1376_MOESM2_ESM.tif
Supplement figure 2. Quantification of the number per SVZ area of the different markers demonstrated that 5 days of running enhanced SVZ neurogenesis in the KO RUN mice with respect to the KO NO RUN but not when compared with WT NO RUN mice. Graphs indicated a significant increment in the KO RUN mice with respect to the KO NO RUN mice of Ki67-dividing cells (genotype x exercise interaction F1,78= 32.76 p<0.001, followed by the analysis of simple effects, p<0.01 KO RUN vs. KO NO RUN), of total B stem cells (GFAP+, genotype x exercise interaction F1,56= 39.87 p<0.001, followed by the analysis of simple effects p<0.01) and of total A neuroblast cells (Dcx+, genotype x exercise interaction F1,56= 18.87 p<0.001, followed by the analysis of simple effects p<0.01). Moreover, we detected a significant increase of dividing B cells in the KO RUN mice with respect to the KO NO RUN (Ki67+/GFAP+, genotype x exercise interaction F1,23= 31.43 p<0.01, followed by analysis of simple effects p<0.01) and of A neuroblast cells (Ki67+/Dcx+, genotype x exercise interaction F1,23= 12.56 p<0.01, followed by analysis of simple effects, p<0.05). Cell numbers in the SVZ are means ± SEM of the analysis of at least three animals per group. *p<0.05, ***p<0.001 (TIF 334 KB)
429_2017_1376_MOESM3_ESM.tif
Supplement figure 3. Graphs illustrating that 5 days of physical activity induced in the KO RUN mice a significant shortening of S-phase in comparison with the KO NO RUN mice (genotype x exercise interaction F1,44= 13.98 p<0.01, followed by the analysis of simple effects, ***p>0.001) and a concomitant increase of the fraction of cells exiting from the S-phase (KO RUN vs KO NO RUN genotype x exercise interaction F1,44= 19.56 p<0.01, followed by the analysis of simple effects, ***p< 0.001). These events result in a shortening of the whole cell cycle length in the KO RUN with respect to the KO NO RUN mice (genotype x exercise interaction F1,44= 26.97, p<0.01, followed by the analysis of simple effects, *p<0.05). Cell cycle analysis was performed in the SVZ of at least three animals per group. Data obtained through the 12 day running protocol are those shown in Figure 2. * p<0.05, *** p<0.001 (TIF 273 KB)
Rights and permissions
About this article
Cite this article
Mastrorilli, V., Scopa, C., Saraulli, D. et al. Physical exercise rescues defective neural stem cells and neurogenesis in the adult subventricular zone of Btg1 knockout mice. Brain Struct Funct 222, 2855–2876 (2017). https://doi.org/10.1007/s00429-017-1376-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1376-4