Abstract
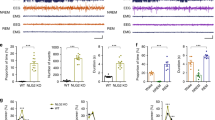
Childhood absence epilepsy (CAE) is an epilepsy syndrome with seizures occurring in the early childhood, highlighting that seizures susceptibility in CAE is dependent on brain development. The Notch 1 signalling pathway is important in brain development, yet the role of the Notch1 signalling pathway in CAE remains elusive. We here explored Notch1 and its modulator notchless homologue 1 (NLE1) expression in WAG/Rij and control rats using immunohistochemistry. Functional Notch 1 effects were assessed in WAG/Rij rats in vivo. WAG/Rij rats lack the developmental increase in cortical Notch1 and NLE 1 mRNA expression seen in controls, and Notch 1 and NLE1 mRNA and protein expression were lower in somatosensory cortices of WAG/Rij rats when compared to controls. This coincided with an overall decreased cortical GFAP expression in the early development in WAG/Rij rats. These effects were region-specific as they were not observed in thalamic tissues. Neuron-to-glia ratio as a marker of the impact of Notch signalling on differentiation was higher in layer 4 of somatosensory cortex of WAG/Rij rats. Acute application of Notch 1 agonist Jagged 1 suppressed, whereas DAPT, a Notch antagonist, facilitated spike and wave discharges (SWDs) in WAG/Rij rats. These findings point to Notch1 as an important signalling pathway in CAE which likely shapes architectural organization of the somatosensory cortex, a region critically involved in developmental epileptogenesis in CAE. More immediate effects of Notch 1 signalling are seen on in vivo SWDs in CAE, pointing to the Notch 1 pathway as a possible treatment target in CAE.









Similar content being viewed by others
References
Ables JL, Breunig JJ, Eisch AJ, Rakic P (2011) Not(ch) just development: notch signalling in the adult brain. Nat Rev Neurosci 12:269–283. doi:10.1038/nrn3024
Artavanis-Tsakonas S, Rand MD, Lake RJ (1999) Notch signaling: cell fate control and signal integration in development. Science 284:770–776. doi:10.1126/science.284.5415.770
Bastany ZJ, Askari S, Dumont GA, Speckmann EJ, Gorji A (2016) Non-invasive monitoring of spreading depression. Neuroscience 333:1–12. doi:10.1016/j.neuroscience.2016.06.056
Berezovska O, Xia MQ, Hyman BT (1998) Notch is expressed in adult brain, is coexpressed with presenilin-1, and is altered in Alzheimer disease. J Neuropathol Exp Neurol 57:738–745. doi:10.1097/00005072-199808000-00003
Breunig JJ, Silbereis J, Vaccarino FM, Sestan N, Rakic P (2007) Notch regulates cell fate and dendrite morphology of newborn neurons in the postnatal dentate gyrus. Proc Natl Acad Sci USA 104:20558–20563. doi:10.1073/pnas.0710156104
Carlén M, Meletis K, Göritz C, Darsalia V, Evergren E, Tanigaki K et al (2009) Forebrain ependymal cells are Notch-dependent and generate neuroblasts and astrocytes after stroke. Nat Neurosci 12:259–267. doi:10.1038/nn.2268
Chantha S-C, Tebbji F, Matton DP (2007) From the notch signaling pathway to ribosome biogenesis. Plant Signal Behav 2:168–170. doi:10.1007/s00425-006-0420-z
Coenen AML, Van Luijtelaar ELJM (2003) Genetic animal models for absence epilepsy: a review of the WAG/Rij strain of rats. Behav Genet 33:635–655. doi:10.1023/A:1026179013847
Conlon RA, Reaume AG, Rossant J (1995) Notch1 is required for the coordinate segmentation of somites. Development 121:1533–1545
Costa RM, Honjo T, Silva AJ (2003) Learning and memory deficits in Notch mutant mice. Curr Biol 13:1348–1354. 10.1016/S0960-9822(03)00492-5
Durner M, Keddache MA, Tomasini L, Shinnar S, Resor SR, Cohen J et al (2001) Genome scan of idiopathic generalized epilepsy: evidence for major susceptibility gene and modifying genes influencing the seizure type. Ann Neurol 49:328–335. doi:10.1002/ana.69
Ferri R, Iliceto G, Carlucci V (1995) Topographic EEG mapping of 3/s spike-and-wave complexes during absence seizures. Ital J Neurol Sci 16:541–547. doi:10.1007/BF02282912
Gaiano N, Nye JS, Fishell G (2000) Radial glial identity is promoted by Notch1 signaling in the murine forebrain. Neuron 26:395–404. 10.1016/S0896-6273(00)81172-1
Genton P (2000) When antiepileptic drugs aggravate epilepsy. Brain Dev 22:75–80. 10.1016/S0387-7604(99)00113-8
Gorji A, Mittag C, Shahabi P, Seidenbecher T, Pape H-C (2011) Seizure-related activity of intralaminar thalamic neurons in a genetic model of absence epilepsy. Neurobiol Dis 43:266–274. doi:10.1016/j.nbd.2011.03.019
Greenberg DA, Durner M, Delgado-Escueta AV (1992) Evidence for multiple gene loci in the expression of the common generalized epilepsies. Neurology 42:56–62. 10.1086/302371
Hatakeyama J, Kageyama R (2006) Notch1 expression is spatiotemporally correlated with neurogenesis and negatively regulated by Notch1-independent Hes genes in the developing nervous system. Cereb Cortex 16(Suppl 1):i132–i137. doi:10.1093/cercor/bhj166
Jafarian M, Karimzadeh F, Alipour F, Attari F, Lotfinia AA, Speckmann E-J et al (2015) Cognitive impairments and neuronal injury in different brain regions of a genetic rat model of absence epilepsy. Neuroscience 298:161–170. doi:10.1016/j.neuroscience.2015.04.033
Khodaie B, Lotfinia AA, Ahmadi M, Lotfinia M, Jafarian M, Karimzadeh F et al (2015) Structural and functional effects of social isolation on the hippocampus of rats with traumatic brain injury. Behav Brain Res 278:55–65. doi:10.1016/j.bbr.2014.09.034
Liu X, Yang Z, Yin Y, Deng X (2014) Increased expression of Notch1 in temporal lobe epilepsy: animal models and clinical evidence. Neural Regen Res 9: 526–533. doi:10.4103/1673-5374.130083
Loiseau P, Panayiotopoulos CP (2005) Childhood absence epilepsy. Available at http://www.ilae-epilepsy.org/Visitors/Centre/ctf. Ref Type: Internet Communication. Accessed 10 Sept 2016
Meeren HKM, Pijn JPM, Van Luijtelaar ELJM, Coenen AML, Lopes da Silva FH (2002) Cortical focus drives widespread corticothalamic networks during spontaneous absence seizures in rats. J Neurosci 22:1480–1495
Niedermeyer E (1996) Primary (idiopathic) generalized epilepsy and underlying mechanisms. Clin Electroencephalogr 27:1–21. doi:10.1177/155005949602700103
Nyfeler Y, Kirch RD, Mantei N, Leone DP, Radtke F, Suter U et al (2005) Jagged1 signals in the postnatal subventricular zone are required for neural stem cell self-renewal. EMBO J 24:3504–3515. doi:10.1038/sj.emboj.7600816
Patten BA, Peyrin JM, Weinmaster G, Corfas G (2003) Sequential signaling through Notch1 and erbB receptors mediates radial glia differentiation. J Neurosci 23:6132–6140
Patten BA, Sardi SP, Koirala S, Nakafuku M, Corfas G (2006) Notch1 signaling regulates radial glia differentiation through multiple transcriptional mechanisms. J Neurosci 26:3102–3108. doi:10.1523/JNEUROSCI.4829-05.2006
Paxinos G, Watson C (1997) The rat brain in stereotaxic coordinates, Compact 3rd edn. Academic Press, San Diego
Peinnequin A, Mouret C, Birot O, Alonso A, Mathieu J, Clarençon D et al (2004) Rat pro-inflammatory cytokine and cytokine related mRNA quantification by real-time polymerase chain reaction using SYBR green. BMC Immunol 5:3. doi:10.1186/1471-2172-5-3
Polack P-O, Guillemain I, Hu E, Deransart C, Depaulis A, Charpier S (2007) Deep layer somatosensory cortical neurons initiate spike-and-wave discharges in a genetic model of absence seizures. J Neurosci 27:6590–6599. doi:10.1523/JNEUROSCI.0753-07.2007
Polack P-O, Mahon S, Chavez M, Charpier S (2009) Inactivation of the somatosensory cortex prevents paroxysmal oscillations in cortical and related thalamic neurons in a genetic model of absence epilepsy. Cereb Cortex 19:2078–2091. doi:10.1093/cercor/bhn237
Redmond L, Oh SR, Hicks C, Weinmaster G, Ghosh A (2000) Nuclear Notch1 signaling and the regulation of dendritic development. Nat Neurosci 3:30–40. doi:10.1038/71104
Royet J, Bouwmeester T, Cohen SM (1998) Notchless encodes a novel WD40-repeat-containing protein that modulates Notch signaling activity. EMBO J 17:7351–7360. doi:10.1093/emboj/17.24.7351
Sadeghian H, Jafarian M, Karimzadeh F, Kafami L, Kazemi H, Coulon P et al (2012) Neuronal death by repetitive cortical spreading depression in juvenile rat brain. Exp Neurol 233:438–446. doi:10.1016/j.expneurol.2011.11.017
Salama-Cohen P, Arévalo M-A, Grantyn R, Rodríguez-Tébar A (2006) Notch and NGF/p75NTR control dendrite morphology and the balance of excitatory/inhibitory synaptic input to hippocampal neurones through Neurogenin 3. J Neurochem 97:1269–1278. doi:10.1111/j.1471-4159.2006.03783.x
Sarkisova K, van Luijtelaar G (2011) The WAG/Rij strain: a genetic animal model of absence epilepsy with comorbidity of depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 35: 854–876. doi:10.1016/j.pnpbp.2010.11.010
Schroeter EH, Kisslinger JA, Kopan R (1998) Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain. Nature 393:382–386. doi:10.1038/30756
Sestan N, Artavanis-Tsakonas S, Rakic P (1999) Contact-dependent inhibition of cortical neurite growth mediated by notch signaling. Science 286:741–746. doi:10.1126/science.286.5440.741
Sha L, Wu X, Yao Y, Wen B, Feng J, Sha Z et al (2014) Notch signaling activation promotes seizure activity in temporal lobe epilepsy. Mol Neurobiol 49:633–644. doi:10.1007/s12035-013-8545-0
Shimada IS, Borders A, Aronshtam A, Spees JL (2011) Proliferating reactive astrocytes are regulated by Notch-1 in the peri-infarct area after stroke. Stroke 42:3231–3237. doi:10.1161/STROKEAHA
Sibbe M, Häussler U, Dieni S, Althof D, Haas CA, Frotscher M (2012) Experimental epilepsy affects Notch1 signalling and the stem cell pool in the dentate gyrus. Eur J Neurosci 36:3643–3652. doi:10.1111/j.1460-9568.2012.08279.x
Stump G, Durrer A, Klein A-L, Lütolf S, Suter U, Taylor V (2002) Notch1 and its ligands Delta-like and Jagged are expressed and active in distinct cell populations in the postnatal mouse brain. Mech Dev 114:153–159. 10.1016/S0925-4773(02)00043-6
Wang Y, Chan SL, Miele L, Yao PJ, Mackes J, Ingram DK et al (2004) Involvement of Notch signaling in hippocampal synaptic plasticity. Proc Natl Acad Sci USA 101:9458–9462. doi:10.1073/pnas.0308126101
Zhai X, Liang P, Li Y, Li L, Zhou Y, Wu X et al (2015) Astrocytes regulate angiogenesis through the jagged1-mediated Notch1 pathway after status epilepticus. Mol Neurobiol 53(9):5893–5901. doi:10.1007/s12035-015-9492-8
Zhang Y, He K, Wang F, Li X, Liu D (2015) Notch-1 signaling regulates astrocytic proliferation and activation after hypoxia exposure. Neurosci Lett 603:12–18. doi:10.1016/j.neulet.2015.07.009
Acknowledgements
This work was supported by the Shefa Neuroscience Research Center, Tehran, Iran, grant related to Dr-Thesis 33817, and Iran National Science Foundation, Tehran, Iran (INSF).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Karimzadeh, F., Modarres Mousavi, S.M., Alipour, F. et al. Developmental changes in Notch1 and NLE1 expression in a genetic model of absence epilepsy. Brain Struct Funct 222, 2773–2785 (2017). https://doi.org/10.1007/s00429-017-1371-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1371-9