Abstract
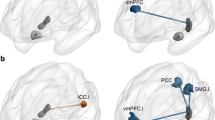
Recent research suggests the anterior and posterior hippocampus form part of two distinct functional neural networks. Here we investigate the structural underpinnings of this functional connectivity difference using diffusion-weighted imaging-based parcellation. Using this technique, we substantiated that the hippocampus can be parcellated into distinct anterior and posterior segments. These structurally defined segments did indeed show different patterns of resting state functional connectivity, in that the anterior segment showed greater connectivity with temporal and orbitofrontal cortex, whereas the posterior segment was more highly connected to medial and lateral parietal cortex. Furthermore, we showed that the posterior hippocampal connectivity to memory processing regions, including the dorsolateral prefrontal cortex, parahippocampal, inferior temporal and fusiform gyri and the precuneus, predicted interindividual relational memory performance. These findings provide important support for the integration of structural and functional connectivity in understanding the brain networks underlying episodic memory.




Similar content being viewed by others
References
Aggleton JP (2012) Multiple anatomical systems embedded within the primate medial temporal lobe: implications for hippocampal function. Neurosci Biobehav R 36:1579–1596. doi:10.1016/j.neubiorev.2011.09.005
Aggleton JP, Wright NF, Vann SD, Saunders RC (2012) Medial temporal lobe projections to the retrosplenial cortex of the macaque monkey. Hippocampus 22:1883–1900. doi:10.1002/hipo.22024
Andrews-Hanna JR, Reidler JS, Sepulcre J, Poulin R, Buckner RL (2010) Functional-anatomic fractionation of the brain’s default network. Neuron 65:550–562. doi:10.1016/j.neuron.2010.02.005
Bannerman DM, Rawlins JNP, McHugh SB, Deacon RMJ, Yee BK, Bast T, Zhang WN, Pothuizen HHJ, Feldon J (2004) Regional dissociations within the hippocampus—memory and anxiety. Neurosci Biobehav R 28:273–283. doi:10.1016/j.neubiorev.2004.03.004
Beckmann CF, DeLuca M, Devlin JT, Smith SM (2005) Investigations into resting-state connectivity using independent component analysis. Philos T Roy Soc B 360:1001–1013. doi:10.1098/rstb.2005.1634
Beckmann M, Johansen-Berg H, Rushworth MF (2009) Connectivity-based parcellation of human cingulate cortex and its relation to functional specialization. J Neurosci 29:1175–1190. doi:10.1523/JNEUROSCI.3328-08.2009
Behrens TEJ, Berg HJ, Jbabdi S, Rushworth MFS, Woolrich MW (2007) Probabilistic diffusion tractography with multiple fibre orientations: what can we gain? Neuroimage 34:144–155. doi:10.1016/j.neuroimage.2006.09.018
Behzadi Y, Restom K, Liau J, Liu TT (2007) A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. Neuroimage 37:90–101. doi:10.1016/j.neuroimage.2007.04.042
Burwell RD, Amaral DG (1998) Perirhinal and postrhinal cortices of the rat: interconnectivity and connections with the entorhinal cortex. J Comp Neurol 391:293–321
Catenoix H, Magnin M, Mauguiere F, Ryvlin P (2011) Evoked potential study of hippocampal efferent projections in the human brain. Clin Neurophysiol 122:2488–2497. doi:10.1016/j.clinph.2011.05.007
Clower DM, West RA, Lynch JC, Strick PL (2001) The inferior parietal lobule is the target of output from the superior colliculus, hippocampus, and cerebellum. J Neurosci 21:6283–6291
Cohn M, McAndrews MP, Moscovitch M (2009a) Associative reinstatement: a novel approach to assessing associative memory in patients with unilateral temporal lobe excisions. Neuropsychologia 47:2989–2994. doi:10.1016/j.neuropsychologia.2009.06.029
Cohn M, Moscovitch M, Lahat A, McAndrews MP (2009b) Recollection versus strength as the primary determinant of hippocampal engagement at retrieval. Proc Natl Acad Sci USA 106:22451–22455. doi:10.1073/pnas.0908651106
Cohn M, St-Laurent M, Barnett A, McAndrews MP (2014) Social inference deficits in temporal lobe epilepsy and lobectomy: risk factors and neural substrates. Soc Cogn Affect Neur. doi: 10.1093/scan/nsu101. Accessed on 25 July 2014
Czerniawski J, Yoon T, Otto T (2009) Dissociating space and trace in dorsal and ventral hippocampus. Hippocampus 19:20–32. doi:10.1002/hipo.20469
Davachi L, Mitchell JP, Wagner AD (2003) Multiple routes to memory: distinct medial temporal lobe processes build item and source memories. Proc Natl Acad Sci USA 100:2157–2162. doi:10.1073/pnas.0337195100
Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D et al (2006) An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31:968–980
Diana RA, Yonelinas AP, Ranganath C (2010) Medial temporal lobe activity during source retrieval reflects information type, not memory strength. J Cognitive Neurosci 22:1808–1818. doi:10.1162/jocn.2009.21335
Duarte A, Henson RN, Graham KS (2011) Stimulus content and the neural correlates of source memory. Brain Res 1373:110–123. doi:10.1016/j.brainres.2010.11.086
Duvernoy HM (2005) The human hippocampus: functional anatomy, vascularization and serial sections with MRI. Springer, New York
Eldridge LL, Knowlton BJ, Furmanski CS, Bookheimer SY, Engel SA (2000) Remembering episodes: a selective role for the hippocampus during retrieval. Nature Neurosci 3:1149–1152
Fanselow MS, Dong HW (2010) Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65:7–19. doi:10.1016/j.neuron.2009.11.031
Henson RN, Rugg MD, Shallice T, Josephs O, Dolan RJ (1999) Recollection and familiarity in recognition memory: an event-related functional magnetic resonance imaging study. J Neurosci 19:3962–3972
Honey CJ, Sporns O, Cammoun L, Gigandet X, Thiran JP, Meuli R, Hagmann P (2009) Predicting human resting-state functional connectivity from structural connectivity. Proc Natl Acad Sci USA 106:2035–2040. doi:10.1073/pnas.0811168106
Hörtnagl H, Berger ML, Sperk G, Pifl CH (1991) Regional heterogeneity in the distribution of neurotransmitter markers in the rat hippocampus. Neuroscience 45:261–272
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17:825–841
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM (2012) Fsl. Neuroimage 62:782–790
Johansen-Berg H, Behrens TEJ, Robson MD, Drobnjak I, Rushworth MFS, Brady JM, Smith SM, Higham DJ, Matthews PM (2004) Changes in connectivity profiles define functionally distinct regions in human medial frontal cortex. Proc Natl Acad Sci USA 101:13335–13340. doi:10.1073/pnas.0403743101
Johnson JD, Suzuki M, Rugg MD (2013) Recollection, familiarity, and content-sensitivity in lateral parietal cortex: a high-resolution fMRI study. Front Human Neurosci 7:219. doi:10.3389/fnhum.2013.00219
Kahn I, Andrews-Hanna JR, Vincent JL, Snyder AZ, Buckner RL (2008) Distinct cortical anatomy linked to subregions of the medial temporal lobe revealed by intrinsic functional connectivity. J Neurophysiol 100:129–139. doi:10.1152/jn.00077.2008
Kensinger EA, Schacter DL (2006) Amygdala activity is associated with the successful encoding of item, but not source, information for positive and negative stimuli. J Neurosci 26:2564–2570. doi:10.1523/JNEUROSCI.5241-05.2006
Kier EL, Staib LH, Davis LM, Bronen RA (2004) MR imaging of the temporal stem: anatomic dissection tractography of the uncinate fasciculus, inferior occipitofrontal fasciculus, and Meyer’s loop of the optic radiation. Am J Neuroradiol 25:677–691
Klein JC, Behrens TE, Robson MD, Mackay CE, Higham DJ, Johansen-Berg H (2007) Connectivity-based parcellation of human cortex using diffusion MRI: establishing reproducibility, validity and observer independence in BA 44/45 and SMA/pre-SMA. Neuroimage 34:204–211. doi:10.1016/j.neuroimage.2006.08.022
Kobayashi Y, Amaral DG (2003) Macaque monkey retrosplenial cortex: II. Cortical afferents. J Comp Neurol 466:48–79. doi:10.1002/cne.10883
Köhler C, Eriksson LG, Davies S, Chan-Palay V (1987) Co-localization of neuropeptide tyrosine and somatostatin immunoreactivity in neurons of individual subfields of the rat hippocampal region. Neurosci Lett 78:1–6
Libby LA, Ekstrom AD, Ragland JD, Ranganath C (2012) Differential connectivity of perirhinal and parahippocampal cortices within human hippocampal subregions revealed by high-resolution functional imaging. J Neurosci 32:6550–6560. doi:10.1016/0304-3940(87)90551-9
Maguire EA, Gadian DG, Johnsrude IS, Good CD, Ashburner J, Frackowiak RS, Frith CD (2000) Navigation-related structural change in the hippocampi of taxi drivers. Proc Natl Acad Sci USA 97:4398–4403. doi:10.1073/pnas.070039597
Malykhin NV, Lebel RM, Coupland NJ, Wilman AH, Carter R (2010) In vivo quantification of hippocampal subfields using 4.7 T fast spin echo imaging. Neuroimage 49:1224–1230. doi:10.1016/j.neuroimage.2009.09.042
Mandler G (1980) Recognizing: the judgment of previous occurrence. Psychol Rev 87:252
Mars RB et al (2011) Diffusion-weighted imaging tractography-based parcellation of the human parietal cortex and comparison with human and macaque resting-state functional connectivity. J Neurosci 31:4087–4100. doi:10.1523/JNEUROSCI.5102-10.2011
Mars RB, Sallet J, Schüffelgen U, Jbabdi S, Toni I, Rushworth MF (2012) Connectivity-based subdivisions of the human right “temporoparietal junction area”: evidence for different areas participating in different cortical networks. Cereb Cortex 22:1894–1903. doi:10.1093/cercor/bhr268
McCormick C, Quraan M, Cohn M, Valiante TA, McAndrews MP (2013a) Default mode network connectivity indicates episodic memory capacity in mesial temporal lobe epilepsy. Epilepsia 54:809–818. doi:10.1111/epi.12098
McCormick C, St-Laurent M, Ty A, Valiante TA, McAndrews MP (2013b) Functional and effective Hippocampal–Neocortical connectivity during construction and elaboration of autobiographical memory retrieval. Cereb Cortex. doi:10.1093/cercor/bht324. Accessed on 24 November 2013
Moayedi M, Salomons TV, Dunlop KA, Downar J, Davis KD (2014) Connectivity-based parcellation of the human frontal polar cortex. Brain Struct Funct:1-14. doi:10.1007/s00429-014-0809-6
Mohedano-Moriano A et al (2007) Topographical and laminar distribution of cortical input to the monkey entorhinal cortex. J Anat 211:250–260. doi:10.1111/j.1469-7580.2007.00764.x
Mohedano-Moriano A et al (2008) Convergence of unimodal and polymodal sensory input to the entorhinal cortex in the fascicularis monkey. Neuroscience 151:255–271. doi:10.1016/j.neuroscience.2007.09.074
Montaldi D, Spencer TJ, Roberts N, Mayes AR (2006) The neural system that mediates familiarity memory. Hippocampus 16:504–520. doi:10.1002/hipo.20178
Neubert FX, Mars RB, Thomas AG, Sallet J, Rushworth MF (2014) Comparison of human ventral frontal cortex areas for cognitive control and language with areas in monkey frontal cortex. Neuron 81:700–713. doi:10.1016/j.neuron.2013.11.012
O’Reilly JX et al (2013) Causal effect of disconnection lesions on interhemispheric functional connectivity in rhesus monkeys. Proc Natl Acad Sci 110:13982–13987. doi:10.1073/pnas.1305062110
Otten LJ (2007) Fragments of a larger whole: retrieval cues constrain observed neural correlates of memory encoding. Cereb Cortex 17:2030–2038. doi:10.1093/cercor/bhl111
Patel J, Schomburg EW, Berényi A, Fujisawa S, Buzsáki G (2013) Local Generation and Propagation of Ripples along the Septotemporal Axis of the Hippocampus. J Neurosci 33:17029–17041. doi:10.1523/JNEUROSCI.2036-13.2013
Pitkänen A, Pikkarainen M, Nurminen N, Ylinen A (2000) Reciprocal connections between the amygdala and the hippocampal formation, perirhinal cortex, and postrhinal cortex in rat: a review. Ann N Y Acad Sci 911:369–391. doi:10.1111/j.1749-6632.2000.tb06738.x
Poppenk J, Moscovitch M (2011) A hippocampal marker of recollection memory ability among healthy young adults: contributions of posterior and anterior segments. Neuron 72:931–937. doi:10.1016/j.neuron.2011.10.014
Poppenk J, Evensmoen HR, Moscovitch M, Nadel L (2013) Long-axis specialization of the human hippocampus. Trends Cogn Sci 17:230–240. doi:10.1016/j.tics.2013.03.005
Ranganath C (2010) A unified framework for the functional organization of the medial temporal lobes and the phenomenology of episodic memory. Hippocampus. 20:1263–1290. doi:10.1002/hipo.20852
Ranganath C, Ritchey M (2012) Two cortical systems for memory-guided behaviour. Nat Rev Neurosci 13:713–726. doi:10.1038/nrn3338
Ranganath C, Johnson MK, D’Esposito M (2003) Prefrontal activity associated with working memory and episodic long-term memory. Neuropsychologia 41:378–389. doi:10.1016/S0028-3932(02)00169-0
Ranganath C, Yonelinas AP, Cohen MX, Dy CJ, Tom SM, D’Esposito M (2004) Dissociable correlates of recollection and familiarity within the medial temporal lobes. Neuropsychologia 42:2–13. doi:10.1016/j.neuropsychologia.2003.07.006
Rockland KS, Van Hoesen GW (1999) Some temporal and parietal cortical connections converge in CA1 of the primate hippocampus. Cereb Cortex 9:232–237. doi:10.1093/cercor/9.3.232
Rogers TT, Hocking J, Noppeney U, Mechelli A, Gorno-Tempini ML, Patterson K, Price CJ (2006) Anterior temporal cortex and semantic memory: reconciling findings from neuropsychology and functional imaging. Cogn Affect Behav Neurosci 6:201–213. doi:10.3758/CABN.6.3.201
Royer S, Sirota A, Patel J, Buzsáki G (2010) Distinct representations and theta dynamics in dorsal and ventral hippocampus. J Neurosci 30:1777–1787. doi:10.1523/JNEUROSCI.4681-09.2010
Rugg MD, Vilberg KL (2013) Brain networks underlying episodic memory retrieval. Curr Opin Neurobiol 23:255–260. doi:10.1016/j.conb.2012.11.005
Segal M, Richter-Levin G, Maggio N (2010) Stress-induced dynamic routing of hippocampal connectivity: a hypothesis. Hippocampus 20:1332–1338. doi:10.1002/hipo.20751
Sloviter RS, Lømo T (2012) Updating the lamellar hypothesis of hippocampal organization. Front Neural Circuits 6:1–16. doi:10.3389/fncir.2012.00102
Small SA, Nava AS, Perera GM, DeLaPaz R, Mayeux R, Stern Y (2001) Circuit mechanisms underlying memory encoding and retrieval in the long axis of the hippocampal formation. Nat Neurosci 4:442–449. doi:10.1038/86115
Strange BA, Witter MP, Lein ES, Moser EI (2014) Functional organization of the hippocampal longitudinal axis. Nat Rev Neurosci 15:655–669. doi:10.1038/nrn3785
Summerfield JJ, Hassabis D, Maguire EA (2009) Cortical midline involvement in autobiographical memory. Neuroimage 44:1188–1200. doi:10.1016/j.neuroimage.2008.09.033
Suzuki WA, Amaral DG (1994) Topographic organization of the reciprocal connections between the monkey entorhinal cortex and the perirhinal and parahippocampal cortices. J Neurosci 14:1856–1877
Thompson CL et al (2008) Genomic anatomy of the hippocampus. Neuron 60:1010–1021. doi:10.1016/j.neuron.2008.12.008
Tomassini V, Jbabdi S, Klein JC, Behrens TE, Pozzilli C, Matthews PM, Rushworth MFS, Johansen-Berg H (2007) Diffusion-weighted imaging tractography-based parcellation of the human lateral premotor cortex identifies dorsal and ventral subregions with anatomical and functional specializations. J Neurosci 27:10259–10269. doi:10.1523/JNEUROSCI.2144-07.2007
Uncapher MR, Rugg MD (2005) Encoding and the durability of episodic memory: a functional magnetic resonance imaging study. J Neurosci 25:7260–7267. doi:10.1523/JNEUROSCI.1641-05.2005
Vincent JL, Kahn I, Snyder AZ, Raichle ME, Buckner RL (2008) Evidence for a frontoparietal control system revealed by intrinsic functional connectivity. J Neurophysiol 100:3328–3342. doi:10.1152/jn.90355.2008
Voets NL, Beckmann CF, Cole DM, Hong S, Bernasconi A, Bernasconi N (2012) Structural substrates for resting network disruption in temporal lobe epilepsy. Brain 135:2350–2357. doi:10.1093/brain/aws137
Ward AM, Schultz AP, Huijbers W, Van Dijk KR, Hedden R, Sperling RA (2014) The parahippocampal gyrus links the default-mode cortical network with the medial temporal lobe memory system. Hum Brain Mapp 35:1061–1073. doi:10.1002/hbm.22234
Whitfield-Gabrieli S, Nieto-Castanon A (2012) Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Conn 2:125–141. doi:10.1089/brain.2012.0073
Witter MP, Van Hoesen GW, Amaral DG (1989) Topographical organization of the entorhinal projection to the dentate gyrus of the monkey. J Neurosci 9:216–228
Wong JX, de Chastelaine M, Rugg MD (2013) Comparison of the neural correlates of encoding item-item and item-context associations. Front Hum Neurosci 7:436. doi:10.3389/fnhum.2013.00436
Yang S et al (2014) Interlamellar CA1 network in the hippocampus. Proc Natl Acad Sci 111:12919–12924. doi:10.1073/pnas.1405468111
Yonelinas AP (2002) The nature of recollection and familiarity: a review of 30 years of research. J Mem Lang 46:441–517. doi:10.1006/jmla.2002.2864
Yonelinas AP, Otten LJ, Shaw KN, Rugg MD (2005) Separating the brain regions involved in recollection and familiarity in recognition memory. J Neurosci 25:3002–3008. doi:10.1523/JNEUROSCI.5295-04.2005
Yonelinas AP, Aly M, Wang WC, Koen JD (2010) Recollection and familiarity: examining controversial assumptions and new directions. Hippocampus 20:1178–1194. doi:10.1002/hipo.20864
Acknowledgments
The authors would like to thank Irene Giannoylis, Keith Ta and Eugene Hlasny for their help in fMRI data collection. The authors declare no competing financial interests. This work was supported by Grants from the Canadian Institute of Health Research (CIHR; Grant Number 97891 to M.P.M; CIHR fellowship to M.M) and the James S. McDonnell Foundation (JSMF # 22002055 to M.P.M).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no potential conflict of interest. The institutions ethics board approved the presented research and informed consent was received from all participants.
Additional information
A. Adnan, A. Barnett and M. Moayedi contributed equally to this work and share first authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Adnan, A., Barnett, A., Moayedi, M. et al. Distinct hippocampal functional networks revealed by tractography-based parcellation. Brain Struct Funct 221, 2999–3012 (2016). https://doi.org/10.1007/s00429-015-1084-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-015-1084-x