Abstract
Because the cerebellum emerged at the agnathan-gnathostome transition and cartilaginous fishes are at the base of the gnathostome lineage, this group is crucial to determine the basic developmental pattern of the cerebellum and to gain insights into its origin. We have systematically analyzed key events in the development of cerebellum and cerebellum-related structures of the shark Scyliorhinus canicula. Three developmental periods are distinguished based on anatomical observations combined with molecular analysis. We present neurochemical and genoarchitectonic evidence on the onset of cerebellar development, the rostral and caudal cerebellar boundaries, the compartmentalization of the cerebellum, and correspondence of cerebellar domains to rhombomeric segmentation of the rostral hindbrain. Our observations, mainly based on the expression pattern of ScHoxA2, support the origin of both the upper and lower auricular leaves from r1 and exclude any cerebellar origin from r2. Correlation between subrhombomeres r1a/r1b and cerebellar domains is proposed based on the ScEn2 expression. The ScEn2 and ScOtx2 expression patterns revealed an antero-posterior cerebellar compartmentalization similar to that of mammals, and supported certain fissures (commonly used to define cerebellar domains) as reliable anatomical landmarks. At difference from mammals, the expression of ScEn2 along the cerebellar median-lateral axis does not reveal a multiple-banded pattern. The present study provides an atlas of cerebellar development in one of the most basal extant gnathostome lineages and emphasizes the importance of combining classic descriptive with modern molecular studies to gain knowledge on the ancestral condition of cerebellar developmental processes and the origins and evolution of the cerebellum.











Similar content being viewed by others
Abbreviations
- AMV:
-
Anterior medullary velum
- Aur:
-
Auricles
- c:
-
Sulcus c
- Cb:
-
Cerebellum
- Cbcl:
-
Caudal lobe of cerebellum
- CbCr:
-
Cerebellar crest
- Cbdp:
-
Dorsal part of the cerebellar body
- Cbp:
-
Cerebellar plate
- Cbrl:
-
Rostral lobe of cerebellum
- Cbvp:
-
Ventral part of the cerebellar body
- ChP:
-
Choroid plexus
- CP:
-
Cerebellar peduncle
- CPp:
-
Cerebellar peduncle primordium
- Di:
-
Diencephalon
- DON:
-
Dorsal octaval nucleus
- e2:
-
Sulcus e2
- e2′:
-
Subcerebellar sulcus e2′
- FL:
-
Fibrous layer
- fmi:
-
Inferior median fissure
- GE:
-
Granular eminence
- GL:
-
Cerebellar granular layer
- IIIn:
-
Oculomotor nerve
- IS:
-
Isthmus
- Isfo:
-
Isthmic fovea
- IVv:
-
Fourth ventricle
- IVn:
-
Trochlear nerve
- IXn:
-
Glossopharyngeal nerve
- iz:
-
Intermediate zone
- LAL:
-
Lower auricular leaf
- Lf:
-
Longitudinal fissure
- LPost:
-
Lobulus posticus
- LR:
-
Lateral recess
- LRL:
-
Lower rhombic lip
- MedE:
-
Median eminence
- Mes:
-
Mesencephalon
- MHB:
-
Midbrain-hindbrain boundary
- MOL:
-
Cerebellar molecular layer
- MON:
-
Medial octaval nucleus
- Mrf:
-
Meso-rhombencephalic fissure
- Nlla:
-
Anterior lateral line nerve
- OLA:
-
Octavolateral area
- OT:
-
Optic tectum
- PL:
-
Purkinje cell layer
- plf:
-
Posterolateral fissure
- prf:
-
Primary transverse fissure
- Pros:
-
Prosencephalon
- r0-2:
-
Rhombomeres 0–2
- Ra:
-
Raphe
- Rh:
-
Rombencephalon
- sid:
-
Intermediate dorsal sulcus
- slH:
-
His sulcus or sulcus limitans
- sms:
-
Median superior sulcus
- UAL:
-
Upper auricular leaf
- UALp:
-
Upper auricular leaf primordium
- URL:
-
Upper rhombic lip
- vAP:
-
Ventral area of the alar plate
- VbCb:
-
Vestibulocerebellum
- VIIIm:
-
Magnocellular octaval nucleus
- VIIIn:
-
Octaval nerve
- VIIn:
-
Facial nerve
- Vn:
-
Trigeminal nerve
References
Alexander T, Nolte C, Krumlauf R (2009) Hox genes and segmentation of the hindbrain and axial skeleton. Annu Rev Cell Dev Biol 25:431–456
Altman J, Bayer SA (1985) Embryonic development of the rat cerebellum. I. Delineation of the cerebellar primordium and early cell movements. J Comp Neurol 231:1–26
Álvarez R, Anadón R (1987) The cerebellum of the dogfish, Scyliorhinus canicula: a quantitative study. J Hirnforsch 28:133–137
Álvarez-Otero R, Anadón R (1992) GOLGI cells of the cerebellum of the dogfish, Scyliorhinus canicula (elasmobranchs): a GOLGI and ultrastructural study. J Hirnforsch 33:321–327
Álvarez-Otero R, Regueira SD, Anadón R (1993) New structural aspects of the synaptic contacts on Purkinje cells in an elasmobranch cerebellum. J Anat 182:13–21
Álvarez-Otero R, Pérez SE, Rodríguez MA, Adrio F, Anadón R (1995) GABAergic neuronal circuits in the cerebellum of the dogfish Scyliorhinus canicula (Elasmobranchs): an immunocytochemical study. Neurosci Lett 187:87–90
Anadón R, Molist P, Rodríguez-Moldes I, López JM, Quintela I, Cerviño MC, Barja P, González A (2000) Distribution of choline acetyltransferase immunoreactivity in the brain of an elasmobranch, the lesser spotted dogfish (Scyliorhinus canicula). J Comp Neurol 420:139–170
Anadón R, Ferreiro-Galve S, Sueiro C, Graña P, Carrera I, Yáñez J, Rodríguez-Moldes I (2009) Calretinin-immunoreactive systems in the cerebellum and cerebellum-related lateral-line medullary nuclei of an elasmobranch, Scyliorhinus canicula. J Chem Neuroanat 37:46–54
Aroca P, Puelles L (2005) Postulated boundaries and differential fate in the developing rostral hindbrain. Brain Res Rev 49:179–190
Aruga J, Inoue T, Hoshino J, Mikoshiba K (2002) Zic2 controls cerebellar development in cooperation with Zic1. J Neurosci 22:218–225
Ashwell KW (2012) Development of the cerebellum in the platypus (Ornithorhynchus anatinus) and short-beaked echidna (Tachyglossus aculeatus). Brain Behav Evol 79:237–251
Ballard WW, Mellinger J, Lechenault H (1993) A series of normal stages for development of Scyliorhinus canicula, the lesser spotted dogfish (Chondrichthyes: Scyliorhinidae). J Exp Zool 267:318–336
Bell CC (2002) Evolution of cerebellum-like structures. Brain Behav Evol 59:312–326
Bell CC, Han V, Sawtell NB (2008) Cerebellum-like structures and their implications for cerebellar function. Annu Rev Neurosci 31:1–24
Berry M, Ibrahim M, Carlile J, Ruge F, Duncan A, Butt AM (1995) Axon-glial relationships in the anterior medullary velum of the adult rat. J Neurocytol 24:965–983
Bovolenta P (2005) Morphogen signaling at the vertebrate growth cone: a few cases or a general strategy? J Neurobiol 64:405–416
Buffo A, Rossi F (2013) Origin, lineage and function of cerebellar glia. Prog Neurobiol 109:42–63
Butler A, Hodos W (2005) Comparative vertebrate neuroanatomy. Evolution and adaption, 2nd edn. Wiley-Liss, Hoboken, NJ, pp 180–197
Cambronero F, Puelles L (2000) Rostrocaudal nuclear relationships in the avian medula oblongata: a fate map with quail chick chimeras. J Comp Neurol 427:522–545
Candal E, Anadón R, Bourrat F, Rodríguez-Moldes I (2005) Cell proliferation in the developing and adult hindbrain and midbrain of trout and medaka (teleosts): a segmental approach. Brain Res 160:157–175
Carrera (2008) Desarrollo de los sistemas gabaérgico y aminérgicos en el sistema nervioso central de peces cartilaginosos. Dissertation, University of Santiago de Compostela
Carrera I, Anadón R, Rodríguez-Moldes I (2012) Development of tyrosine hydroxylase-immunoreactive cell populations and fiber pathways in the brain of the dogfish Scyliorhinus canicula: New perspectives on the evolution of the vertebrate catecholaminergic system. J Comp Neurol 520:3574–3603
Cerri S, Piccolini VM, Bernocchi G (2010) Postnatal development of the central nervous system: anomalies in the formation of cerebellum fissures. Anat Rec (Hoboken) 293:492–501
Chaplin N, Tendeng C, Wingate RJ (2010) Absence of an external germinal layer in zebrafish and shark reveals a distinct, anamniote ground plan of cerebellum development. J Neurosci 30:3048–3057
Cheng Y, Sudarov A, Szulc KU, Sgaier SK, Stephen D, Turnbull DH, Joyner AL (2010) The Engrailed homeobox genes determine the different foliation patterns in the vermis and hemispheres of the mammalian cerebellum. Development 137:519–529
Compagnucci C, Debiais-Thibaud M, Coolen M, Fish J, Griffin JN, Bertocchini F, Minoux M, Rijli FM, Borday-Birraux V, Casane D, Mazan S, Depew MJ (2013) Pattern and polarity in the development and evolution of the gnathostome jaw: both conservation and heterotopy in the branchial arches of the shark, Scyliorhinus canicula. Dev Biol 377:428–448
Conley RA, Bodznick D (1994) The cerebellar dorsal granular ridge in an elasmobranch has proprioceptive and electroreceptive representations and projects homotopically to the medullary electrosensory nucleus. J Comp Physiol A 174:707–721
Coolen M, Sauka-Spengler T, Nicolle D, Le-Mentec C, Lallemand Y, Da Silva C, Plouhinec J-L, Robert B, Wincker P, Shi D-L, Mazan S (2007) Evolution of axis specification mechanisms in jawed vertebrates: insights from a chondrichthyan. PLoS One 2(4):e374. doi:10.1371/journal.pone.0000374
Coolen M, Menuet A, Chassoux D, Compagnucci C, Henry S, Lévèque L, Da Silva C, Gavory F, Samain S, Wincker P, Thermes C, D’Aubenton-Carafa Y, Rodriguez-Moldes I, Naylor G, Depew M, Sourdaine P, Mazan S (2009) The dogfish Scyliorhinus canicula, a reference in jawed vertebrates. In: Behringer RR, Johnson AD, Krumlauf RE (eds) Emerging model organisms. A laboratory manual, vol 1. Cold Spring Harbor, CSHL Press, NY, pp 431–446
Davis CA, Joyner A (1988) Expression patterns of the homeo box-containing genes En-1 and En-2 and the proto-oncogene int-1 diverge during mouse development. Genes Dev 2:1736–1744
Davis CA, Noble-Topham SE, Rossant J, Joyner AL (1988) Expression of the homeo box-containing gene En-2 delineates a specific region of the developing mouse brain. Genes Dev 2:361–371
Devor A (2000) Is the cerebellum like cerebellar-like structures? Brain Res Rev 34:149–156
Dun XP (2012) Origin of climbing fiber neurons and the definition of rhombic lip. Int J Dev Neurosci 30:391–395
Ferreiro-Galve S (2010) Brain and retina regionalization in sharks: study based on the spatiotemporal expression pattern of Pax6 and other neurochemical markers. Dissertation, University of Santiago de Compostela
Ferreiro-Galve S, Candal E, Rodríguez-Moldes I (2012) Dynamic expression of Pax6 in the shark olfactory system: evidence for the presence of Pax6 cells along the olfactory nerve pathway. J Exp Zool B Mol Dev Evol 318:79–90
Fiebig E (1988) Connections of the corpus cerebelli in the Thornback guitarfish, Platyrhinoidis triseriata (Elasmobranchii): a study with WGA-HRP and extracellular granule cell recording. J Comp Neurol 268:567–583
Frantz GD, Weimann JM, Levin ME, McConnell SK (1994) Otx1 and Otx2 define layers and regions in developing cerebral cortex and cerebellum. J Neurosci 14:5725–5740
Fritzsch B, Sonntag R (1987) The trochlear nerve of amphibians and its relation to proprioceptive fibers: a qualitative and quantitative HRP study. Anat Embryol (Berl) 177:105–114
Fujita H, Sugihara I (2013) Branching patterns of olivocerebellar axons in relation to the compartmental organization of the cerebellum. Front Neural Circuits 7:3
Gardner CA, Barald KF (1992) Expression patterns of engrailed-like proteins in the chick embryo. Dev Dyn 193:370–388
Germot A, Lecointre G, Plouhinec JL, Le Mentec C, Girardot F, Mazan S (2001) Structural evolution of Otx genes in craniates. Mol Biol Evol 18:1668–1678
Gómez A, Durán E, Ocaña FM, Jiménez-Moya F, Broglio C, Domezain A, Salas C, Rodríguez F (2004) Observation on the brain development of the sturgeon Acipenser naccarii. In: Carmona R, Domezain A, García-Gallego M, Hernando JA, Rodríguez F, Ruiz-Rejón M (eds) Biology, conservation and sustainable development of sturgeons. Springer, Berlin, pp 155–174
Herrup K, Kuemerle B (1997) The compartmentalization of the cerebellum. Annu Rev Neurosci 20:61–90
Hidalgo-Sánchez M, Backer S, Puelles L, Bloch-Gallego E (2012) Origin and plasticity of the inferior olivary complex. Dev Biol 371:215–226
Ibrahim M, Menoud PA, Celio MR (2000) Neurones in the adult rat anterior medullary velum. J Comp Neurol 419:122–134
Irving C, Malhas A, Guthrie S, Mason I (2002) Establishing the trochlear motor axon trajectory: role of the isthmic organizer and Fgf8. Development 129:5389–5398
Joven A, Morona R, González A, Moreno N (2013) Spatiotemporal patterns of Pax3, Pax6 and Pax7 expression in the developing brain of a urodele amphibian, Pleurodeles waltl. J Comp Neurol 521:3913–3953
Kaslin J, Ganz J, Geffarth M, Grandel H, Hans S, Brand M (2009) Stem cells in the adult zebrafish cerebellum: initiation and maintenance of a novel stem cell niche. J Neurosci 29:6142–6153
Landsberg RL, Awatramani RB, Hunter NL, Farago AF, DiPietrantonio HJ, Rodríguez CI, Dymecki SM (2005) Hindbrain rhombic lip is comprised of discrete progenitor cell populations allocated by Pax6. Neuron 48:933–947
Larsell O (1925) The development of the cerebellum in the frog (Hyla regilla) in relation to the vestibular and lateral-line systems. J Comp Neurol 39:249–289
Larsell O (1934) Morphogenesis and evolution of the cerebellum. Arch Neurol 31:373–395
Larsell O (1967) The comparative anatomy and histology of the cerebellum from myxinoids through birds. University of Minnesota Press, Minneapolis
Leclerc N, Doré L, Parent A, Hawkes R (1990) The compartmentalization of the monkey and rat cerebellar cortex: zebrin I and cytochrome oxidase. Brain Res 506:70–78
Ma S, Kwon HJ, Huang Z (2012) Ric-8a, a guanine nucleotide exchange factor for heterotrimeric G proteins, regulates Bergmann glia-basement membrane adhesion during cerebellar foliation. J Neurosci 32:14979–14993
Margotta V (2007) PCNA immunoreactivity revealing normal proliferative activity in the brain of an adult Elasmobranch, Torpedo marmorata. Ital J Anat Embryol 112:145–155
Marín F, Puelles L (1995) Morphological fate of rhombomeres in quail/chick chimeras: a segmental analysis of hindbrain nuclei. Eur J Neurosci 7:1714–1738
Martínez S, Andreu A, Mecklenburg N, Echevarría D (2013) Cellular and molecular basis of cerebellar development. Front Neuroanat 7:18
Marzban H, Chung SH, Pezhouh MK, Feirabend H, Watanabe M, Voogd J, Hawkes R (2010) Antigenic compartmentation of the cerebellar cortex in the chicken (Gallus domesticus). J Comp Neurol 518:2221–2239
Mazan S, Jaillard D, Baratte B, Janvier P (2000) Otx1 gene-controlled morphogenesis of the horizontal semicircular canal and the origin of the gnathostome characteristics. Evol Dev 2:186–193
Meek J, Hafmans TG, Maler L, Hawkes R (1992) Distribution of zebrin II in the gigantocerebellum of the mormyrid fish Gnathonemus petersii compared with other teleosts. J Comp Neurol 316:17–31
Millen KJ, Wurst W, Herrup K, Joyner A (1994) Abnormal embryonic cerebellar development and patterning of postnatal foliation in two mouse Engrailed-2 mutants. Development 120:695–706
Millet S, Alvarado-Mallart RM (1995) Expression of the homeobox-containing gene En-2 during development of the chick central nervous system. Eur J Neurosci 7:777–791
Montgomery JC (1981) Origin of the Parallel fibers in the cerebellar crest overlying the intermediate nucleus of the elasmobranch hindbrain. J Comp Neurol 202:185–191
Montgomery JC, Bodznick D, Yopak KE (2012) The cerebellum and cerebellum-like structures of cartilaginous fishes. Brain Behav Evol 80:152–165
Moreno-Bravo JA, Perez-Balaguer A, Martinez-Lopez JE, Aroca P, Puelles L, Martinez S, Puelles E (2014) Role of Shh in the development of molecularly characterized tegmental nuclei in mouse rhombomere 1. Brain Struct Funct 219:777–792
Namba K, Sugihara I, Hashimoto M (2011) Close correlation between the birth date of Purkinje cells and the longitudinal compartmentalization of the mouse adult cerebellum. J Comp Neurol 519:2594–2614
New JG (2001) Comparative neurobiology of the elasmobranch cerebellum: theme and variations on a sensorimotor interface. Environ Biol Fish 60:93–108
Nieuwenhuys R (1967) Comparative anatomy of the cerebellum. Prog Brain Res 25:1–93
Northcutt RG (2002) Understanding vertebrate brain evolution. Integ Comp Biol 42:743–756
Orvis GD, Barraza LH, Wilson SL, Szulc KU, Turnbull DH, Joyner AL (2012) The engrailed homeobox genes are required in multiple cell lineages to coordinate sequential formation of fissures and growth of the cerebellum. Dev Biol 367:25–39
Oulion S, Debiais-Thibaud M, d’Aubenton-Carafa Y, Thermes C, Da Silva C, Bernard-Samain S, Gavory F, Wincker P, Mazan S, Casane D (2010) Evolution of Hox gene clusters in gnathostomes: insights from a survey of a shark (Scyliorhinus canicula) transcriptome. Mol Biol Evol 27:2829–2838
Pakan JMP, Iwaniuk AN, Wylie DRW, Hawkes R, Marzban H (2007) Purkinje cell compartmentation as revealed by zebrin II expression in the cerebellar cortex of pigeons (Columba livia). J Comp Neurol 501:619–630
Palgrem A (1921) Embryological and morphological studies on the midbrain and cerebellum of vertebrates. Acta Zool 2:1–94
Paul DH, Roberts BL (1975) Responses of neurones in the cerebellar corpus of the dogfish (Scyliorhinus canicula). J Physiol 244:47P–49P
Peña-Melián A, Puerta Fonolla J, Gil Loyzaga P (1986) The ontogeny of the cerebellar fissures in the chick embryo. Anat Embryol (Berl) 175:119–128
Plouhinec JL, Leconte L, Sauka-Spengler TS, Bovolenta P, Mazan S, Saule S (2005) Comparative analysis of gnathostome Otx gene expression patterns in the developing eye: implications for the functional evolution of the multigene family. Dev Biol 278:560–575
Pose-Méndez S (2013) Developmental study of the cerebellum in cartilaginous fishes: Towards the identification of primitive features of the cerebellar formation in gnathostomes. Dissertation, University of Santiago de Compostela
Pose-Méndez S, Candal E, Adrio F, Rodríguez-Moldes I (2014) Development of the cerebellar afferent system in the shark Scyliorhinus canicula: insights into the basal organization of precerebellar nuclei in gnathostomes. J Comp Neurol 522:131–168
Pose-Méndez S, Candal E, Mazan S, Rodríguez-Moldes I (2015) Genoarchitecture of the rostral hindbrain of a shark: basis for understanding the emergence of the cerebellum at the agnathan-gnathostome transition. Brain Struct Funct. doi:10.1007/s00429-014-0973-8
Pouwels E (1978) On the development of the cerebellum of the trout, Salmo gairdneri. I. Patterns of cell migration. Anat Embryol (Berl) 152:291–308
Prayer D, Kasprian G, Krampl E, Ulm B, Witzani L, Prayer L, Brugger PC (2006) MRI of normal fetal brain development. Eur J Radiol 57:199–216
Puelles L, Ferrán JL (2012) Concept of neural genoarchitecture and its genomic fundament. Front Neuroanat 6:47
Puelles L, Harrison M, Paxinos G, Watson C (2013) A developmental ontology for the mammalian brain based on the prosomeric model. Trends Neurosci 36:570–578
Puzdrowski RL (1997) Anti-Zebrin II immunopositivity in the cerebellum and octavolateral nuclei in two species of stingrays. Brain Behav Evol 50:358–368
Quintana-Urzainqui I, Rodríguez-Moldes I, Candal E (2014) Developmental, tract-tracing and immunohistochemical study of the peripheral olfactory system in a basal vertebrate: insights on Pax6 neurons migrating along the olfactory nerve. Brain Struct Funct 219:85–104
Rodríguez-Moldes I, Manso MJ, Becerra M, Molist P, Anadón R (1993) Distribution of substance P-like immunoreactivity in the brain of the elasmobranch Scyliorhinus canicula. J Comp Neurol 335:228–244
Rodríguez-Moldes I, Ferreiro-Galve S, Carrera I, Sueiro C, Candal E, Manzan S, Anadón R (2008) Development of the cerebellar body in sharks: spatiotemporal relations of Pax6-expression, cell proliferation and differentiation. Neurosci Lett 432:105–110
Rodríguez-Moldes I, Carrera I, Pose-Méndez S, Quintana-Urzainqui I, Candal E, Anadón R, Mazan S, Ferreiro-Galve S (2011) Regionalization of the shark hindbrain: a survey of an ancestral organization. Front Neuroanat 5:16
Rüdeberg S (1961) Morphogenetic studies on the cerebellar nuclei and their homologization in different vertebrates including man. Dissertation, Univ of Lund
Sgaier SK, Millet S, Villanueva MP, Berenshteyn F, Song C, Joyner AL (2005) Morphogenetic and cellular movements that shape the mouse cerebellum; insights from genetic fate mapping. Neuron 45:27–40
Sgaier SK, Lao Z, Villanueva MP, Berenshteyn F, Stephen D, Turnbull RK, Joyner AL (2007) Genetic subdivision of the tectum and cerebellum into functionally related regions based on differential sensitivity to engrailed proteins. Development 134:2325–2335
Sillitoe RV, Stephen D, Lao Z, Joyner A (2008) Engrailed homeobox genes determine the organization of Purkinje cell sagittal stripe gene expression in the adult cerebellum. J Neurosci 28:12150–12162
Sillitoe RV, Vogel MW, Joyner AL (2010) Engrailed homeobox genes regulate establishment of the cerebellar afferent circuit map. J Neurosci 30:10015–10024
Smeets WJAJ (1998) Cartilaginous fish. In: Nieuwenhuys R, Ten Donkelaar HJ, Nicholson C (eds) The central nervous system of vertebrates, vol 1. Springer, Berlin, pp 551–654
Smeets WJ, Nieuwenhuys R (1976) Topological analysis of the brain stem of the sharks Squalus acanthias and Scyliorhinus canicula. J Comp Neurol 165:333–368
Smeets WJAJ, Nieuwenhuys R, Roberts BL (1983) The central nervous system of cartilaginous fishes. Structure and functional correlations. Springer, Berlin
Sterzi G (1912) Il sistema nervoso centrale dei vertebrate Pesci, vol II. Draghi, Padova
Sudarov A, Joyner AL (2007) Cerebellum morphogenesis: the foliation pattern is orchestrated by multi-cellular anchoring centers. Neural Dev 2:26
ten Donkelaar HJ, Lammens M, Wesseling P, Thijssen HO, Renier WO (2003) Development and developmental disorders of the human cerebellum. J Neurol 250:1025–1036
Triulzi F, Parazzini C, Righini A (2005) MRI of fetal and neonatal cerebellar development. Semin Fetal Neonatal Med 10:411–420
Tubbs RS, Wellons JC 3rd, Salter G, Oakes WJ (2004) Fenestration of the superior medullary velum as treatment for a trapped fourth ventricle: a feasibility study. Clin Anat 17:82–87
Tümpel S, Cambronero F, Sims C, Krumlauf R, Wiedemann LM (2008) A regulatory module embedded in the coding region of Hoxa2 controls expression in rhombomere 2. Proc Natl Acad Sci USA 105:20077–20082
Van Essen DC (1997) A tension-based theory of morphogenesis and compact wiring in the central nervous system. Nature 385:313–318
Vecino E, Ekström P (1991) Expression of the homeobox engrailed gene during the embryonic development of the nervous system of the trout (Salmo fario L.). Neurosci Lett 129:311–314
Vogel-Höpker A, Rohrer H (2002) The specification of noradrenergic locus coeruleus (LC) neurones depends on bone morphogenetic proteins (BMPs). Development 129:983–991
von Kupffer C (1906) “Die Morphogenie des Zentral nerven systems”. In: O. Hertwig (Jena: Fischer) (ed) Handbuch der vergleichenden und experimentellen Entwicklungslehre der Wirbeltiere. Vol. 2, Part 3 pp 1–272
Watanabe Y, Toyoda R, Nakamura H (2004) Navigation of trochlear motor axons along the midbrain-hindbrain boundary by neuropilin 2. Development 131:681–692
White JJ, Reeber SL, Hawkes R, Sillitoe RV (2012) Wholemount immunohistochemistry for revealing complex brain topography. J Vis Exp 62:e4042
Wilson SL, Kalinovsky A, Orvis GD, Joyner A (2011) Spatially restricted and developmentally dynamic expression of engrailed genes in multiple cerebellar cell types. Cerebellum 10:356–372
Wingate RJ, Hatten ME (1999) The role of the rhombic lip in avian cerebellum development. Development 126:4395–4404
Zupanc GK, Hinsch K, Gage FH (2005) Proliferation, migration, neuronal differentiation, and long-term survival of new cells in the adult zebrafish brain. J Comp Neurol 488:290–319
Acknowledgments
We thank Prof. Dr. R. Anadón for the valuable comments made during the preparation of this paper and his critical reading of the manuscript. This work was supported by grants from the Spanish Dirección General de Investigación-FEDER (BFU2010- 15816), the Xunta de Galicia (10PXIB200051PR, CN 2012/237), and European Community-Research Infrastructure Action under the FP7 "Capacities" Specific Programme (ASSEMBLE 227799).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The manuscript does not contain clinical studies or patient data.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2015_998_MOESM1_ESM.tif
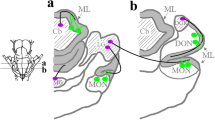
Supplementary material 1 Transverse sections of the caudal hindbrain in Scyliorhinus canicula at late embryonic and juvenile stages, showing specific neurochemical features of the intermediate zone (indicated with the red dotted line) in between the lower auricular leaf and dorsal octaval nucleus. In this area, it was observed: granule cells with the hematoxylin–eosin staining (a), expression of ScHoxA2 (b), low density of 5-HT-ir fibers (c), and high density of CR-ir fibers (d). Scale bars 200 µm (c, d); 500 µm (a, b) (TIFF 2620 kb)
429_2015_998_MOESM2_ESM.tif
Supplementary material 2 Transverse section of the caudal hindbrain in Scyliorhinus canicula at stage 29, showing a reduction in the expression of proliferating cell nuclear marker (PCNA) in the zones where grooves are forming (indicated with arrowheads). Scale bar 300 µm (TIFF 366 kb)
Rights and permissions
About this article
Cite this article
Pose-Méndez, S., Candal, E., Mazan, S. et al. Morphogenesis of the cerebellum and cerebellum-related structures in the shark Scyliorhinus canicula: insights on the ground pattern of the cerebellar ontogeny. Brain Struct Funct 221, 1691–1717 (2016). https://doi.org/10.1007/s00429-015-0998-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-015-0998-7