Abstract
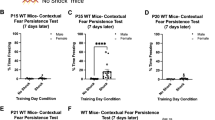
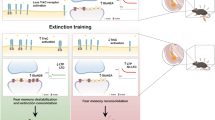
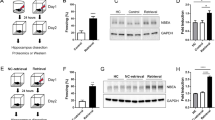
The process of learning mainly depends on the ability to store new information, while the ability to retrieve this information and express appropriate behaviors are also crucial for the adaptation of individuals to environmental cues. Thereby, all three components contribute to the cognitive fitness of an individual. While a lack of behavioral adaptation is a recurrent trait of intellectually disabled patients, discriminating between memory formation, memory retrieval or behavioral expression deficits is not easy to establish. Here, we report some deficits in contextual fear behavior in knockout mice for the intellectual disability gene Il1rapl1. Functional in vivo experiments revealed that the lack of conditioned response resulted from a local inhibitory to excitatory (I/E) imbalance in basolateral amygdala (BLA) consecutive to a loss of excitatory drive onto BLA principal cells by caudal hippocampus axonal projections. A normalization of the fear behavior was obtained in adult mutant mice following opsin-based in vivo synaptic priming of hippocampo-BLA synapses in adult il1rapl1 knockout mice, indicating that synaptic efficacy at hippocampo-BLA projections is crucial for contextual fear memory expression. Importantly, because this restoration was obtained after the learning phase, our results suggest that some of the genetically encoded cognitive deficits in humans may originate from a lack of restitution of genuinely formed memories rather than an exclusive inability to store new memories.




Similar content being viewed by others
References
Anagnostaras SG, Gale GD, Fanselow MS (2001) Hippocampus and contextual fear conditioning: recent controversies and advances. Hippocampus 11:8–17 doi:10.1002/1098-1063(2001) 11:1<8:AID-HIPO1015>3.0.CO;2-7
Baroncelli L, Braschi C, Spolidoro M et al (2011) Brain plasticity and disease: a matter of inhibition. Neural Plast 2011:1–11. doi:10.1155/2011/286073
Bienvenu TCM, Busti D, Magill PJ et al (2012) Cell-type-specific recruitment of amygdala interneurons to hippocampal theta rhythm and noxious stimuli in vivo. Neuron 74:1059–1074. doi:10.1016/j.neuron.2012.04.022
Carrié A, Jun L, Bienvenu T et al (1999) A new member of the IL-1 receptor family highly expressed in hippocampus and involved in X-linked mental retardation. Nat Genet 23:25–31. doi:10.1038/12623
Felix-Ortiz AC, Tye KM (2014) Amygdala inputs to the ventral hippocampus bidirectionally modulate social behavior. J Neurosci 34:586–595. doi:10.1523/JNEUROSCI.4257-13.2014
Felix-Ortiz AC, Beyeler A, Seo C et al (2013) BLA to vHPC inputs modulate anxiety-related behaviors. Neuron 79:658–664. doi:10.1016/j.neuron.2013.06.016
Gambino F, Pavlowsky A, Begle A et al (2007) IL1-receptor accessory protein-like 1 (IL1RAPL1), a protein involved in cognitive functions, regulates N-type Ca2+-channel and neurite elongation. Proc Natl Acad Sci 104:9063–9068. doi:10.1073/pnas.0701133104
Gatto CL, Broadie K (2010) Genetic controls balancing excitatory and inhibitory synaptogenesis in neurodevelopmental disorder models. Front Synaptic Neurosci 2:4. doi:10.3389/fnsyn.2010.00004
Goosens KA (2011) Hippocampal regulation of aversive memories. Curr Opin Neurobiol 21:460–466. doi:10.1016/j.conb.2011.04.003
Goshen I, Brodsky M, Prakash R et al (2011) Dynamics of retrieval strategies for remote memories. Cell 147:678–689. doi:10.1016/j.cell.2011.09.033
Hayashi T, Yoshida T, Ra M et al (2013) IL1RAPL1 associated with mental retardation and autism regulates the formation and stabilization of glutamatergic synapses of cortical neurons through RhoA signaling pathway. PLoS One 8:e66254. doi:10.1371/journal.pone.0066254.s002
Herry C, Mons N (2004) Resistance to extinction is associated with impaired immediate early gene induction in medial prefrontal cortex and amygdala. Eur J Neurosci 20:781–790. doi:10.1111/j.1460-9568.2004.03542.x
Houbaert X, Zhang CL, Gambino F et al (2013) Target-specific vulnerability of excitatory synapses leads to deficits in associative memory in a model of intellectual disorder. J Neurosci 33:13805–13819. doi:10.1523/JNEUROSCI.1457-13.2013
Hübner C, Bosch D, Gall A et al (2014) Ex vivo dissection of optogenetically activated mPFC and hippocampal inputs to neurons in the basolateral amygdala: implications for fear and emotional memory. Front Behav Neurosci 8:64. doi:10.3389/fnbeh.2014.00064
Knapska E, Macias M, Mikosz M et al (2012) Functional anatomy of neural circuits regulating fear and extinction. Proc Natl Acad Sci 109:17093–17098. doi:10.1073/pnas.1202087109
Kullmann DM, Moreau AW, Bakiri Y, Nicholson E (2012) Plasticity of inhibition. Neuron 75:951–962. doi:10.1016/j.neuron.2012.07.030
Ledoux JE (2000) Emotion circuits in the brain. Annu Rev Neurosci 23:155–184. doi:10.1146/annurev.neuro.23.1.155
Lisman JE, Coyle JT, Green RW et al (2008) Circuit-based framework for understanding neurotransmitter and risk gene interactions in schizophrenia. Trends Neurosci 31:234–242. doi:10.1016/j.tins.2008.02.005
Liu X, Ramirez S, Pang PT et al (2012) Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature 484:381–385. doi:10.1038/nature11028
Maren S, Phan KL, Liberzon I (2013) The contextual brain: implications for fear conditioning, extinction and psychopathology. Nat Rev Neurosci. doi:10.1038/nrn3492
Morozov A, Sukato D, Ito W (2011) Selective suppression of plasticity in amygdala inputs from temporal association cortex by the external capsule. J Neurosci 31:339–345
Muller J, Corodimas KP, Fridel Z, Ledoux JE (1997) Functional inactivation of the lateral and basal nuclei of the amygdala by muscimol infusion prevents fear conditioning to an explicit conditioned stimulus and to contextual stimuli. Behav Neurosci 111:683–691
Orsini CA, Kim JH, Knapska E, Maren S (2011) Hippocampal and prefrontal projections to the basal amygdala mediate contextual regulation of fear after extinction. J Neurosci 31:17269–17277. doi:10.1523/JNEUROSCI.4095-11.2011
Pavlowsky A, Gianfelice A, Pallotto M et al (2010) A postsynaptic signaling pathway that may account for the cognitive defect due to IL1RAPL1 mutation. Curr Biol 20:103–115. doi:10.1016/j.cub.2009.12.030
Pavlowsky A, Chelly J, Billuart P (2011) Emerging major synaptic signaling pathways involved in intellectual disability. Mol Psychiatry. doi:10.1038/mp.2011.139
Pentkowski NS, Blanchard DC, Lever C et al (2006) Effects of lesions to the dorsal and ventral hippocampus on defensive behaviors in rats. Eur J Neurosci 23:2185–2196. doi:10.1111/j.1460-9568.2006.04754.x
Petreanu L, Huber D, Sobczyk A, Svoboda K (2007) Channelrhodopsin-2-assisted circuit mapping of long-range callosal projections. Nat Neurosci 10:663–668. doi:10.1038/nn1891
Pitkänen A, Pikkarainen M, Nurminen N, Ylinen A (2000) Reciprocal connections between the amygdala and the hippocampal formation, perirhinal cortex, and postrhinal cortex in rat. A review. Ann N Y Acad Sci 911:369–391
Piton A, Michaud JL, Peng H et al (2008) Mutations in the calcium-related gene IL1RAPL1 are associated with autism. Hum Mol Genet 17:3965–3974. doi:10.1093/hmg/ddn300
Rudy JW, Huff NC, Matus-Amat P (2004) Understanding contextual fear conditioning: insights from a two-process model. Neurosci Biobehav Rev 28:675–685. doi:10.1016/j.neubiorev.2004.09.004
Rumpel S, LeDoux J, Zador A, Malinow R (2005) Postsynaptic receptor trafficking underlying a form of associative learning. Science 308:83–88. doi:10.1126/science.1103944
Valnegri P, Montrasio C, Brambilla D et al (2011) The X-linked intellectual disability protein IL1RAPL1 regulates excitatory synapse formation by binding PTP and RhoGAP2. Hum Mol Genet 20:4797–4809. doi:10.1093/hmg/ddr418
Yoshida T, Yasumura M, Uemura T et al (2011) IL-1 receptor accessory protein-like 1 associated with mental retardation and autism mediates synapse formation by trans-synaptic interaction with protein tyrosine phosphatase. J Neurosci 31:13485–13499. doi:10.1523/JNEUROSCI.2136-11.2011
Acknowledgments
We thank Drs. Cyril Herry, Andréas Luthi and Andrew Penn for fruitful discussions and corrections of the manuscript. We are grateful to Dr. P. Billuart and J. Chelly for their help and for providing us with Il1rapl1 mutant mice, and Dr. Jiyun Peng for helping in the tracking of mouse activities. We acknowledge Ed Boyden, Ph.D., and the Massachusetts Institute of Technology for kindly providing AAV-ArchT constructs to the research community and Dr. C. Herry for providing us with AAV viruses. We also thank the Pole in vivo and animal facilities of the Bordeaux University for the animal care. The microscopy was done in the Bordeaux Imaging Centre of the University of Bordeaux, with the help of Sébastien Marais. This study was supported by grants from the Agence Nationale pour la Recherche (ANR-10-BLAN-1434, ANR-12-JSV4-0005-01, ANR-12-SAMA-001-03 and ANR-10-LABX-43 BRAIN to E.H. and Y.H.), the European Neuroscience Institutes Network (Y.H.), the Gencodys FP7 program (Y.H.).
Author information
Authors and Affiliations
Corresponding author
Additional information
C.-L. Zhang and X. Houbaert contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2014_882_MOESM1_ESM.pdf
Supplementary Fig. 1: Effect of functional inactivation of cHPC or BLA on contextual fear expression. a Experimental protocol. WT mice were conditioned, and tested for contextual fear response 24 h later (“before”). In vivo muscimol applications were done 24 h later, and mice tested 1-2 h and 24 h later (“after”). b Cannula positions of included animals for cHPC (left) or BLA (right) inactivation. c cHPC and BLA impairment by muscimol infusion temporally affects the intensity of the contextual fear conditioned response.Supplementary Fig. 2: Pharmacological BLA activation during contextual fear expression in Il1rapl1 WT and KO mice. a Experimental protocol. b Cannula positions in Il1rapl1 KO and WT animals. Pharmacological activation of BLA induced a transient increase of freezing levels in KO animals (Fig. 7C). Two-way ANOVA revealed a significant effect of genotype (ANOVA F (1,45) = 12,362; p = 0.001) and of treatment (ANOVA F (2,45) = 5,555; p = 0.007), but no interaction between genotype and treatment (ANOVA F (2,45) = 0,318; p = 0.729) as the treatment did not have an significant effect on WT freezing levels. SNK post hoc multiple comparisons were used to test for differences between groups. They showed an effect of treatment on KO freezing levels (SNK before vs. 1–2 h after; p < 0.05). The difference observed between WT and KO animals before treatment (SNK before WT vs. KO; p < 0.05) disappeared after illumination, KO animals reaching similar levels to WT animals (SNK 1–2 h after WT vs. KO; p > 0.05). 24 h after treatment, a difference between WT and KO animals is observed again (SNK 24 h later WT vs. KO; p < 0.05), showing that BLA activation transiently rescues KO phenotype.Supplementary Fig. 3: Infection sites of in vitro optogenetic experiments. The maximal surface of each infection site was assessed by GFP expression, and reported according to the bregma position for all used WT and KO animals. Note that infection sites always included the caudal CA1 region of the hippocampus and often the ventral subiculum.Supplementary Fig. 4: Lack of BLA neuronal activation following hippocampal projections in Il1rapl1 KO mice. A: Experimental protocol. WT and KO GAD-67eGPF mice were infected with AAV-ChR2 in the cHPC. Three weeks later, fiber optics were positioned under anesthesia and repetitive bursts of 460 nm light pulses were applied at the infection sites. 90 min after illumination, animals were sacrificed and prepared for C-Fos labeling in the BLA. B: counting of C-Fos positive cells expressing or not eGFP was performed in WT and KO mice (3 and 3), and showed a higher proportion of double labeled cells in the KO mice. (PDF 4466 kb)
Rights and permissions
About this article
Cite this article
Zhang, CL., Houbaert, X., Lepleux, M. et al. The hippocampo-amygdala control of contextual fear expression is affected in a model of intellectual disability. Brain Struct Funct 220, 3673–3682 (2015). https://doi.org/10.1007/s00429-014-0882-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-014-0882-x