Abstract
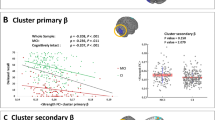
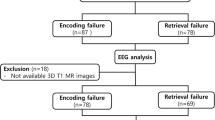
Episodic memory is typically affected during the course of Alzheimer’s disease (AD). Due to the pronounced heterogeneity of functional neuroimaging studies on episodic memory impairments in mild cognitive impairment (MCI) and AD regarding their methodology and findings, we aimed to delineate consistent episodic memory-related brain activation patterns. We performed a systematic, quantitative, coordinate-based whole-brain activation likelihood estimation meta-analysis of 28 functional magnetic resonance imaging (fMRI) studies comprising 292 MCI and 102 AD patients contrasted to 409 age-matched control subjects. We included episodic encoding and/or retrieval phases, investigated the effects of group, verbal or image stimuli and correlated mean Mini-Mental-Status-Examination (MMSE) scores with the modelled activation estimates. MCI patients presented increased right hippocampal activation during memory encoding, decreased activation in the left hippocampus and fusiform gyrus during retrieval tasks, as well as attenuated activation in the right anterior insula/inferior frontal gyrus during verbal retrieval. In AD patients, however, stronger activation within the precuneus during encoding tasks was accompanied by attenuated right hippocampal activation during retrieval tasks. Low cognitive performance (MMSE scores) was associated with stronger activation of the precuneus and reduced activation of the right (para)hippocampus and anterior insula/inferior frontal gyrus. This meta-analysis provides evidence for a specific and probably disease stage-dependent brain activation pattern related to the pathognomonic AD characteristic of episodic memory loss.




Similar content being viewed by others
References
Bäckman L, Andersson JLR, Nyberg L et al (1999) Brain regions associated with episodic retrieval in normal aging and Alzheimer’ s disease. Neurology
Belleville S, Clément F, Mellah S et al (2011) Training-related brain plasticity in subjects at risk of developing Alzheimer’s disease. Brain 134:1623–1634. doi:10.1093/brain/awr037
Bokde ALW, Lopez-Bayo P, Meindl T et al (2006) Functional connectivity of the fusiform gyrus during a face-matching task in subjects with mild cognitive impairment. Brain 129:1113–1124. doi:10.1093/brain/awl051
Bokde ALW, Karmann M, Born C et al (2010) Altered brain activation during a verbal working memory task in subjects with amnestic mild cognitive impairment. J Alzheimers Dis 21:103–118. doi:10.3233/JAD-2010-091054
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Braak H, Alafuzoff I, Arzberger T et al (2006) Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112:389–404. doi:10.1007/s00401-006-0127-z
Browndyke JN, Giovanello K, Petrella J et al (2012) Phenotypic regional functional imaging patterns during memory encoding in mild cognitive impairment and Alzheimer’s disease. Alzheimers Dement 9:284–294. doi:10.1016/j.jalz.2011.12.006
Buckner RL, Raichle ME, Miezin FM, Petersen SE (1996) Functional anatomic studies of memory retrieval for auditory words and visual pictures. J Neurosci 16:6219–6235
Buckner RL, Snyder AZ, Shannon BJ et al (2005) Molecular, structural, and functional characterization of Alzheimer’s disease: evidence for a relationship between default activity, amyloid, and memory. J Neurosci 25:7709–7717. doi:10.1523/JNEUROSCI.2177-05.2005
Cabeza R, Dolcos F, Graham R, Nyberg L (2002) Similarities and differences in the neural correlates of episodic memory retrieval and working memory. Neuroimage 16:317–330. doi:10.1006/nimg.2002.1063
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129:564–583. doi:10.1093/brain/awl004
Celone KA, Calhoun VD, Dickerson BC et al (2006) Alterations in memory networks in mild cognitive impairment and Alzheimer’s disease: an independent component analysis. J Neurosci 26:10222–10231. doi:10.1523/JNEUROSCI.2250-06.2006
Clément F, Belleville S (2010) Compensation and disease severity on the memory-related activations in mild cognitive impairment. Biol Psychiatry 68:894–902. doi:10.1016/j.biopsych.2010.02.004
Clément F, Belleville S (2012) Effect of disease severity on neural compensation of item and associative recognition in mild cognitive impairment. J Alzheimers Dis 29:109–123. doi:10.3233/JAD-2011-110426
Clément F, Belleville S, Mellah S (2010) Functional neuroanatomy of the encoding and retrieval processes of verbal episodic memory in MCI. Cortex 46:1005–1015. doi:10.1016/j.cortex.2009.07.003
Dannhauser TM, Shergill SS, Stevens T et al (2008) An fMRI study of verbal episodic memory encoding in amnestic mild cognitive impairment. Cortex 44:869–880. doi:10.1016/j.cortex.2007.04.005
Dere E, Pause BM, Pietrowsky R (2010) Emotion and episodic memory in neuropsychiatric disorders. Behav Brain Res 215:162–171. doi:10.1016/j.bbr.2010.03.017
Derflinger S, Sorg C, Gaser C et al (2011) Grey-matter atrophy in Alzheimer’s disease is asymmetric but not lateralized. J Alzheimers Dis 25:347–357. doi:10.3233/JAD-2011-110041
Dubois B, Feldman HH, Jacova C et al (2010) Revising the definition of Alzheimer’s disease: a new lexicon. Lancet Neurol 9:1118–1127. doi:10.1016/S1474-4422(10)70223-4
Duvernoy H, Cattin F, Naidich T et al (2005) Sectional anatomy and magnetic resonance imaging. In: Duvernoy HM (ed) Hum. Hippocampus, 3rd edn. Springer, Heidelberg, pp 129–217
Eickhoff SB, Stephan KE, Mohlberg H et al (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25:1325–1335. doi:10.1016/j.neuroimage.2004.12.034
Eickhoff SB, Paus T, Caspers S et al (2007) Assignment of functional activations to probabilistic cytoarchitectonic areas revisited. Neuroimage 36:511–521. doi:10.1016/j.neuroimage.2007.03.060
Eickhoff SB, Laird AR, Grefkes C et al (2009) Coordinate-based activation likelihood estimation meta-analysis of neuroimaging data: a random-effects approach based on empirical estimates of spatial uncertainty. Hum Brain Mapp 30:2907–2926. doi:10.1002/hbm.20718
Eickhoff SB, Bzdok D, Laird AR et al (2012) Activation likelihood estimation meta-analysis revisited. Neuroimage 59:2349–2361. doi:10.1016/j.neuroimage.2011.09.017
Espinosa A, Alegret M, Valero S et al (2013) A longitudinal follow-up of 550 mild cognitive impairment patients: evidence for large conversion to dementia rates and detection of major risk factors involved. J Alzheimers Dis 34:769–780. doi:10.3233/JAD-122002
Folstein MF, Folstein SE, McHugh PR (1975) Mini-mental state: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198. doi:10.1016/0022-3956(75)90026-6
Frings L, Dressel K, Abel S et al (2010) Reduced precuneus deactivation during object naming in patients with mild cognitive impairment, Alzheimer’s disease, and frontotemporal lobar degeneration. Dement Gerieatr Cogn 30:334–343. doi:10.1159/000320991
Giovanello KS, De Brigard F, Hennessey Ford J et al (2012) Event-related functional magnetic resonance imaging changes during relational retrieval in normal aging and amnestic mild cognitive impairment. J Int Neuropsychol Soc 18:886–897. doi:10.1017/S1355617712000689
Golby A, Silverberg G, Race E et al (2005) Memory encoding in Alzheimer’s disease: an fMRI study of explicit and implicit memory. Brain 128:773–787. doi:10.1093/brain/awh400
Golby AJ, Poldrack RA, Brewer JB et al (2001) Material-specific lateralization in the medial temporal lobe and prefrontal cortex during memory encoding. Brain 124:1841–1854
Gould RL, Brown RG, Owen AM et al (2005) Functional neuroanatomy of successful paired associate learning in Alzheimer’s disease. Am J Psychiatry 162:2049–2060
Grady C (2012) The cognitive neuroscience of ageing. Nat Rev Neurosci 13:491–505. doi:10.1038/nrn3256
Grön G, Bittner D, Schmitz B et al (2002) Subjective memory complaints: objective neural markers in patients with Alzheimer’s disease and major depressive disorder. Ann Neurol 51:491–498. doi:10.1002/ana.10157
Gusnard DA, Raichle ME (2001) Searching for a baseline: functional imaging and the resting human brain. Nat Rev Neurosci 2:685–694
Hämäläinen A, Pihlajamäki M, Tanila H et al (2007) Increased fMRI responses during encoding in mild cognitive impairment. Neurobiol Aging 28:1889–1903. doi:10.1016/j.neurobiolaging.2006.08.008
Hampstead BM, Stringer AY, Stilla RF et al (2011) Where did I put that? Patients with amnestic mild cognitive impairment demonstrate widespread reductions in activity during the encoding of ecologically relevant object-location associations. Neuropsychologia 49:2349–2361. doi:10.1016/j.neuropsychologia.2011.04.008
Hanseeuw B, Dricot L, Kavec M et al (2011) Associative encoding deficits in amnestic mild cognitive impairment: a volumetric and functional MRI study. Neuroimage 56:1743–1748. doi:10.1016/j.neuroimage.2011.03.034
Hashimoto M, Masliah E (2003) Cycles of aberrant synaptic sprouting and neurodegeneration in Alzheimer’s and dementia with Lewy bodies. Neurochem Res 28:1743–1756
Hayes SM, Ryan L, Schnyer DM, Nadel L (2004) An fMRI study of episodic memory: retrieval of object, spatial, and temporal information. Behav Neurosci 118:885–896. doi:10.1037/0735-7044.118.5.885
Heun R, Freymann K, Erb M et al (2007) Mild cognitive impairment (MCI) and actual retrieval performance affect cerebral activation in the elderly. Neurobiol Aging 28:404–413. doi:10.1016/j.neurobiolaging.2006.01.012
Huijbers W, Vannini P, Sperling RA et al (2012) Explaining the encoding/retrieval flip: memory-related deactivations and activations in the posteromedial cortex. Neuropsychologia 50:3764–3774. doi:10.1016/j.neuropsychologia.2012.08.021
Hynd MR, Scott HL, Dodd PR (2004) Glutamate-mediated excitotoxicity and neurodegeneration in Alzheimer’s disease. Neurochem Int 45:583–595. doi:10.1016/j.neuint.2004.03.007
Ikonomovic MD, Klunk WE, Abrahamson EE et al (2011) Precuneus amyloid burden is associated with reduced cholinergic activity in Alzheimer disease. Neurology 77:39–47. doi:10.1212/WNL.0b013e3182231419
Jin M, Pelak VS, Curran T et al (2012) A preliminary study of functional abnormalities in aMCI subjects during different episodic memory tasks. Magn Reson Imaging 30:459–470. doi:10.1016/j.mri.2011.12.014
Jonides J, Wager T, Badre DT (2002) Memory, neuroimaging. In: Ramachandran VS (ed) Encycl. Hum. Brain. Elsevier Science, San Diego, pp 797–833
Jungwirth S, Zehetmayer S, Hinterberger M et al (2012) The validity of amnestic MCI and non-amnestic MCI at age 75 in the prediction of Alzheimer’s dementia and vascular dementia. Int Psychogeriatr 24:959–966. doi:10.1017/S1041610211002870
Kelley WM, Miezin FM, McDermott KB et al (1998) Hemispheric specialization in human dorsal frontal cortex and medial temporal lobe for verbal and nonverbal memory encoding. Neuron 20:927–936
Kircher TT, Weis S, Freymann K et al (2007) Hippocampal activation in patients with mild cognitive impairment is necessary for successful memory encoding. J Neurol Neurosurg Psychiatry 78:812–818. doi:10.1136/jnnp.2006.104877
Kochan N, Breakspear M, Slavin MJ et al (2010) Functional alterations in brain activation and deactivation in mild cognitive impairment in response to a graded working memory challenge. Dement Geriatr Cogn 30:553–568. doi:10.1159/000322112
Kühn S, Gallinat J (2013) Segregating cognitive functions within hippocampal formation: a quantitative meta-analysis on spatial navigation and episodic memory. Hum Brain Mapp. doi:10.1002/hbm.22239
Kurth F, Zilles K, Fox PT et al (2010) A link between the systems: functional differentiation and integration within the human insula revealed by meta-analysis. Brain Struct Funct 214:519–534. doi:10.1007/s00429-010-0255-z
Laird AR, Eickhoff SB, Kurth F et al (2009) ALE meta-analysis workflows via the brainmap database: progress towards a probabilistic functional brain atlas. Front Neuroinform 3:11. doi:10.3389/neuro.11.023.2009
Lim TS, Lee HY, Barton JJS, Moon SY (2011) Deficits in face perception in the amnestic form of mild cognitive impairment. J Neurol Sci 309:123–127. doi:10.1016/j.jns.2011.07.001
Machulda MM, Senjem ML, Weigand SD et al (2009) Functional MRI changes in amnestic and non-amnestic MCI during encoding and recognition tasks. J Int Neuropsychol Soc 15:372–382. doi:10.1017/S1355617709090523
Mandzia JL, McAndrews MP, Grady CL et al (2009) Neural correlates of incidental memory in mild cognitive impairment: an fMRI study. Neurobiol Aging 30:717–730. doi:10.1016/j.neurobiolaging.2007.08.024
McCarthy G, Blamire AM, Rothman DL et al (1993) Echo-planar magnetic resonance imaging studies of frontal cortex activation during word generation in humans. Proc Natl Acad Sci USA 90:4952–4956
McCarthy G, Puce A, Gore JC, Allison T (1996) Face-specific processing in the human fusiforrn gyms. J Cogn Neurosci 9:605–610
McKhann G, Drachman D, Folstein M et al (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
Mueller SG, Schuff N, Yaffe K et al (2010) Hippocampal atrophy patterns in mild cognitive impairment and Alzheimer’s disease. Hum Brain Mapp 31:1339–1347. doi:10.1002/hbm.20934
Nickl-Jockschat T, Kleiman A, Schulz JB et al (2012) Neuroanatomic changes and their association with cognitive decline in mild cognitive impairment: a meta-analysis. Brain Struct Funct 217:115–125. doi:10.1007/s00429-011-0333-x
Nyberg L, McIntosh AR, Houle S et al (1996) Activation of medial temporal structures during episodic memory retrieval. Nature 380:715–717. doi:10.1038/380715a0
O’Brien JL, O’Keefe K, LaViolette PS et al (2010) Longitudinal fMRI in elderly reveals loss of hippocampal activation with clinical decline. Neurology 74:1969–1976
Palop JJ, Chin J, Roberson ED et al (2007) Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of Alzheimer’s disease. Neuron 55:697–711. doi:10.1016/j.neuron.2007.07.025
Pariente J, Cole S, Henson R et al (2005) Alzheimer’s patients engage an alternative network during a memory task. Ann Neurol 58:870–879. doi:10.1002/ana.20653
Pause BM, Zlomuzica A, Kinugawa K et al (2013) Perspectives on episodic-like and episodic memory. Front Behav Neurosci 7:33. doi:10.3389/fnbeh.2013.00033
Peters F, Collette F, Degueldre C et al (2009) The neural correlates of verbal short-term memory in Alzheimer’s disease: an fMRI study. Brain 132:1833–1846. doi:10.1093/brain/awp075
Petersen RC (2011) Mild cognitive impairment. N Engl J Med 364:2227–2234
Petersen RC, Smith GE, Waring SC et al (1999) Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 56:303–308
Petrella JR, Krishnan S, Slavin MJ et al (2006) Mild cognitive impairment: evaluation with 4-T functional MR imaging. Radiology 240:177–186
Petrella JR, Prince SE, Wang L et al (2007) Prognostic value of posteromedial cortex deactivation in mild cognitive impairment. PLoS ONE 2:e1104. doi:10.1371/journal.pone.0001104
Price CJ (2000) The anatomy of language: contributions from functional neuroimaging. J Anat 197:335–359
Price CJ (2010) The anatomy of language: a review of 100 fMRI studies published in 2009. Ann NY Acad Sci 1191:62–88. doi:10.1111/j.1749-6632.2010.05444.x
Rémy F, Mirrashed F, Campbell B, Richter W (2005) Verbal episodic memory impairment in Alzheimer’s disease: a combined structural and functional MRI study. Neuroimage 25:253–266. doi:10.1016/j.neuroimage.2004.10.045
Rosazza C, Minati L, Ghielmetti F et al (2009) Engagement of the medial temporal lobe in verbal and nonverbal memory: assessment with functional MR imaging in healthy subjects. Am J Neuroradiol 30:1134–1141. doi:10.3174/ajnr.A1518
Salehi A, Swaab DF (1999) Diminished neuronal metabolic activity in Alzheimer’s disease. Review article. J Neural Transm 106:955–986
Schacter DL, Curran T, Reiman EM et al (1999) Medial temporal lobe activation during episodic encoding and retrieval: a PET study. Hippocampus 9:575–881. doi:10.1002/(SICI)1098-1063(1999)9:5<575:AID-HIPO11>3.0.CO;2-K
Scheff SW, Price DA, Schmitt FA et al (2013) Synapse stability in the precuneus early in the progression of Alzheimer’s disease. J Alzheimers Dis 35:599–609. doi:10.3233/JAD-122353
Schilbach L, Bzdok D, Timmermans B et al (2012) Introspective minds: using ALE meta-analyses to study commonalities in the neural correlates of emotional processing, social & unconstrained cognition. PLoS ONE 7:e30920. doi:10.1371/journal.pone.0030920
Schroeter ML, Stein T, Maslowski N, Neumann J (2009) Neural correlates of Alzheimer’s disease and mild cognitive impairment: a systematic and quantitative meta-analysis involving 1351 patients. Neuroimage 47:1196–1206. doi:10.1016/j.neuroimage.2009.05.037
Schwindt GC, Black SE (2009) Functional imaging studies of episodic memory in Alzheimer’s disease: a quantitative meta-analysis. Neuroimage 45:181–190. doi:10.1016/j.neuroimage.2008.11.024
Scoville WB, Milner B (2000) Loss of recent memory after bilateral hippocampal lesions. J Neuropsych Clin Neurosci 12:103–113
Small SA, Schobel SA, Buxton RB et al (2011) A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nat Rev Neurosci 12:585–601. doi:10.1038/nrn3085
Spaniol J, Davidson PSR, Kim ASN et al (2009) Event-related fMRI studies of episodic encoding and retrieval: meta-analyses using activation likelihood estimation. Neuropsychologia 47:1765–1779. doi:10.1016/j.neuropsychologia.2009.02.028
Sperling RA, Bates JF, Chua EF et al (2003) fMRI studies of associative encoding in young and elderly controls and mild Alzheimer’s disease. J Neurol Neurosurg Psychiatry 74:44–50
Sperling RA, Laviolette PS, O’Keefe K et al (2009) Amyloid deposition is associated with impaired default network function in older persons without dementia. Neuron 63:178–188. doi:10.1016/j.neuron.2009.07.003
Squire LR, Stark CEL, Clark RE (2004) The medial temporal lobe. Annu Rev Neurosci 27:279–306. doi:10.1146/annurev.neuro.27.070203.144130
Stark CE, Squire LR (2001) When zero is not zero: the problem of ambiguous baseline conditions in fMRI. Proc Natl Acad Sci USA 98:12760–12766. doi:10.1073/pnas.221462998
Trivedi MA, Murphy CM, Goetz C et al (2008) fMRI activation changes during successful episodic memory encoding and recognition in amnestic mild cognitive impairment relative to cognitively healthy older adults. Dement Geriatr Cogn Disord 26:123–137. doi:10.1159/000148190
Tulving E (1972) Episodic and semantic memory. In: Tulving E (ed) Organ. Mem. Academic Press, New York, pp 381–403
Turkeltaub PE, Eden GF, Jones KM, Zeffiro TA (2002) Meta-analysis of the functional neuroanatomy of single-word reading: method and validation. Neuroimage 16:765–780. doi:10.1006/nimg.2002.1131
Turkeltaub PE, Eickhoff SB, Laird AR et al (2012) Minimizing within-experiment and within-group effects in activation likelihood estimation meta-analyses. Hum Brain Mapp 33:1–13. doi:10.1002/hbm.21186
Van der Meulen M, Lederrey C, Rieger SW et al (2012) Associative and semantic memory deficits in amnestic mild cognitive impairment as revealed by functional magnetic resonance imaging. Cogn Behav Neurol 25:195–215
Wang X, Han Z, He Y et al (2013) Where color rests: spontaneous brain activity of bilateral fusiform and lingual regions predicts object color knowledge performance. Neuroimage 76:252–263. doi:10.1016/j.neuroimage.2013.03.010
Welsh K, Butters N, Hughes J et al (1991) Detection of abnormal memory decline in mild cases of Alzheimer’s disease using CERAD neuropsychological measures. Arch Neurol Chicago 48:278–281
Acknowledgments
We would like to thank B. Hampstead for sharing additional data which lead to the inclusion of one further study (Hampstead et al. 2011). We would like to furthermore thank Imis Dogan for her invaluable input during our data investigation, Ana Costa for providing knowledge and relevant literature about the different human memory systems and Melissa Chung for proofreading the manuscript. NJS and JBS are in part funded by the Helmholtz Alliance ICEMED - Imaging and Curing Environmental Metabolic Diseases, through the Initiative and Network Fund of the Helmholtz Association. SBE was funded by the Human Brain Project (R01-MH074457-01A1) and acknowledges funding by the Initiative and Networking Fund of the Helmholtz Association within the Helmholtz Alliance on Systems Biology (Human Brain Model) and the IRTG 1328. SBE and KR were funded by the Excellence Initiative of the German federal and state governments.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nellessen, N., Rottschy, C., Eickhoff, S.B. et al. Specific and disease stage-dependent episodic memory-related brain activation patterns in Alzheimer’s disease: a coordinate-based meta-analysis. Brain Struct Funct 220, 1555–1571 (2015). https://doi.org/10.1007/s00429-014-0744-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-014-0744-6