Abstract
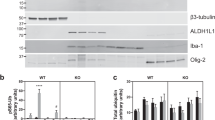
SUCLA2 encodes the ATP-forming β subunit (A-SUCL-β) of succinyl-CoA ligase, an enzyme of the citric acid cycle. Mutations in SUCLA2 lead to a mitochondrial disorder manifesting as encephalomyopathy with dystonia, deafness and lesions in the basal ganglia. Despite the distinct brain pathology associated with SUCLA2 mutations, the precise localization of SUCLA2 protein has never been investigated. Here, we show that immunoreactivity of A-SUCL-β in surgical human cortical tissue samples was present exclusively in neurons, identified by their morphology and visualized by double labeling with a fluorescent Nissl dye. A-SUCL-β immunoreactivity co-localized >99 % with that of the d subunit of the mitochondrial F0–F1 ATP synthase. Specificity of the anti-A-SUCL-β antiserum was verified by the absence of labeling in fibroblasts from a patient with a complete deletion of SUCLA2. A-SUCL-β immunoreactivity was absent in glial cells, identified by antibodies directed against the glial markers GFAP and S100. Furthermore, in situ hybridization histochemistry demonstrated that SUCLA2 mRNA was present in Nissl-labeled neurons but not glial cells labeled with S100. Immunoreactivity of the GTP-forming β subunit (G-SUCL-β) encoded by SUCLG2, or in situ hybridization histochemistry for SUCLG2 mRNA could not be demonstrated in either neurons or astrocytes. Western blotting of post mortem brain samples revealed minor G-SUCL-β immunoreactivity that was, however, not upregulated in samples obtained from diabetic versus non-diabetic patients, as has been described for murine brain. Our work establishes that SUCLA2 is expressed exclusively in neurons in the human cerebral cortex.








Similar content being viewed by others
References
Brekke E, Walls AB, Norfeldt L, Schousboe A, Waagepetersen HS, Sonnewald U (2012) Direct measurement of backflux between oxaloacetate and fumarate following pyruvate carboxylation. Glia 60:147–158
Carrozzo R, Dionisi-Vici C, Steuerwald U, Lucioli S, Deodato F, Di Giandomenico S, Bertini E, Franke B, Kluijtmans LAJ, Meschini MC, Rizzo C, Piemonte F, Rodenburg R, Santer R, Santorelli FM, van Rooij A, Vermunt-de Koning D, Morava E, Wevers RA (2007) SUCLA2 mutations are associated with mild methylmalonic aciduria, leigh-like encephalomyopathy, dystonia and deafness. Brain 130:862–874
Chinopoulos C (2011a) Mitochondrial consumption of cytosolic ATP: not so fast. FEBS Lett 585:1255–1259
Chinopoulos C (2011b) The “B Space” of mitochondrial phosphorylation. J Neurosci Res 89:1897–1904
Chinopoulos C (2013) Which way does the citric acid cycle turn during hypoxia? The critical role of alpha-ketoglutarate dehydrogenase complex. J Neurosci Res 91:1030–1043
Chinopoulos C, Adam-Vizi V (2010) Mitochondria as ATP consumers in cellular pathology. Biochim Biophys Acta 1802:221–227
Chinopoulos C, Gerencser AA, Mandi M, Mathe K, Torocsik B, Doczi J, Turiak L, Kiss G, Konrad C, Vajda S, Vereczki V, Oh RJ, Adam-Vizi V (2010) Forward operation of adenine nucleotide translocase during F0F1-ATPase reversal: critical role of matrix substrate-level phosphorylation. FASEB J 24:2405–2416
Conti F, Melone M, De BS, Minelli A, Brecha NC, Ducati A (1998) Neuronal and glial localization of GAT-1, a high-affinity gamma-aminobutyric acid plasma membrane transporter, in human cerebral cortex: with a note on its distribution in monkey cortex. J Comp Neurol 396:51–63
Conti F, Zuccarello LV, Barbaresi P, Minelli A, Brecha NC, Melone M (1999) Neuronal, glial, and epithelial localization of gamma-aminobutyric acid transporter 2, a high-affinity gamma-aminobutyric acid plasma membrane transporter, in the cerebral cortex and neighboring structures. J Comp Neurol 409:482–494
Dringen R, Bishop GM, Koeppe M, Dang TN, Robinson SR (2007) The pivotal role of astrocytes in the metabolism of iron in the brain. Neurochem Res 32:1884–1890
Elpeleg O, Miller C, Hershkovitz E, Bitner-Glindzicz M, Bondi-Rubinstein G, Rahman S, Pagnamenta A, Eshhar S, Saada A (2005) Deficiency of the ADP-forming succinyl-CoA synthase activity is associated with encephalomyopathy and mitochondrial DNA depletion. Am J Hum Genet 76:1081–1086
Gerencser AA, Chinopoulos C, Birket MJ, Jastroch M, Vitelli C, Nicholls DG, Brand MD (2012) Quantitative measurement of mitochondrial membrane potential in cultured cells: calcium-induced de- and hyperpolarization of neuronal mitochondria. J Physiol 590:2845–2871
Jenkins TM, Weitzman PD (1986) Distinct physiological roles of animal succinate thiokinases. Association of guanine nucleotide-linked succinate thiokinase with ketone body utilization. FEBS Lett 205:215–218
Johnson JD, Mehus JG, Tews K, Milavetz BI, Lambeth DO (1998) Genetic evidence for the expression of ATP- and GTP-specific succinyl-CoA synthetases in multicellular eucaryotes. J Biol Chem 273:27580–27586
Kadrmas EF, Ray PD, Lambeth DO (1991) Apparent ATP-linked succinate thiokinase activity and its relation to nucleoside diphosphate kinase in mitochondrial matrix preparations from rabbit. Biochim Biophys Acta 1074:339–346
Kiss G, Konrad C, Doczi J, Starkov AA, Kawamata H, Manfredi G, Zhang SF, Gibson GE, Beal MF, Adam-Vizi V, Chinopoulos C (2013) The negative impact of alpha-ketoglutarate dehydrogenase complex deficiency on matrix substrate-level phosphorylation. FASEB J 27:2392–2406
Kowluru A, Tannous M, Chen HQ (2002) Localization and characterization of the mitochondrial isoform of the nucleoside diphosphate kinase in the pancreatic beta cell: evidence for its complexation with mitochondrial succinyl-CoA synthetase. Arch Biochem Biophys 398:160–169
Labbe RF, Kurumada T, Onisawa J (1965) The role of succinyl-CoA synthetase in the control of heme biosynthesis. Biochim Biophys Acta 111:403–415
Lambeth DO, Tews KN, Adkins S, Frohlich D, Milavetz BI (2004) Expression of two succinyl-CoA synthetases with different nucleotide specificities in mammalian tissues. J Biol Chem 279:36621–36624
Li X, Wu F, Beard DA (2013) Identification of the kinetic mechanism of succinyl-CoA synthetase. Biosci Rep 33:145–163
Lopes-Cardozo M, Larsson OM, Schousboe A (1986) Acetoacetate and glucose as lipid precursors and energy substrates in primary cultures of astrocytes and neurons from mouse cerebral cortex. J Neurochem 46:773–778
McKee EE, Bentley AT, Smith RM Jr, Ciaccio CE (1999) Origin of guanine nucleotides in isolated heart mitochondria. Biochem Biophys Res Commun 257:466–472
McKee EE, Bentley AT, Smith RM Jr, Kraas JR, Ciaccio CE (2000) Guanine nucleotide transport by atractyloside-sensitive and -insensitive carriers in isolated heart mitochondria. Am J Physiol Cell Physiol 279:C1870–C1879
Miller C, Wang L, Ostergaard E, Dan P, Saada A (2011) The interplay between SUCLA2, SUCLG2, and mitochondrial DNA depletion. Biochim Biophys Acta 1812:625–629
Milon L, Rousseau-Merck MF, Munier A, Erent M, Lascu I, Capeau J, Lacombe ML (1997) nm23-H4, a new member of the family of human nm23/nucleoside diphosphate kinase genes localised on chromosome 16p13. Hum Genet 99:550–557
Minelli A, DeBiasi S, Brecha NC, Zuccarello LV, Conti F (1996) GAT-3, a high-affinity GABA plasma membrane transporter, is localized to astrocytic processes, and it is not confined to the vicinity of GABAergic synapses in the cerebral cortex. J Neurosci 16:6255–6264
Navarro-Sastre A, Tort F, Garcia-Villoria J, Pons MR, Nascimento A, Colomer J, Campistol J, Yoldi ME, Lopez-Gallardo E, Montoya J, Unceta M, Martinez MJ, Briones P, Ribes A (2012) Mitochondrial DNA depletion syndrome: new descriptions and the use of citrate synthase as a helpful tool to better characterise the patients. Mol Genet Metab 107:409–415
Ostergaard E (2008) Disorders caused by deficiency of succinate-CoA ligase. J Inherit Metab Dis 31:226–229
Ostergaard E, Christensen E, Kristensen E, Mogensen B, Duno M, Shoubridge EA, Wibrand F (2007a) Deficiency of the alpha subunit of succinate-coenzyme A ligase causes fatal infantile lactic acidosis with mitochondrial DNA depletion. Am J Hum Genet 81:383–387
Ostergaard E, Hansen FJ, Sorensen N, Duno M, Vissing J, Larsen PL, Faeroe O, Thorgrimsson S, Wibrand F, Christensen E, Schwartz M (2007b) Mitochondrial encephalomyopathy with elevated methylmalonic acid is caused by SUCLA2 mutations. Brain 130:853–861
Ostergaard E, Schwartz M, Batbayli M, Christensen E, Hjalmarson O, Kollberg G, Holme E (2010) A novel missense mutation in SUCLG1 associated with mitochondrial DNA depletion, encephalomyopathic form, with methylmalonic aciduria. Eur J Pediatr 169:201–205
Ottaway JH, McClellan JA, Saunderson CL (1981) Succinic thiokinase and metabolic control. Int J Biochem 13:401–410
Palkovits M (1973) Isolated removal of hypothalamic or other brain nuclei of the rat. Brain Res 59:449–450
Pall ML (1985) GTP: a central regulator of cellular anabolism. Curr Top Cell Regul 25:1–20
Pfaff E, Klingenberg M, Heldt HW (1965) Unspecific permeation and specific exchange of adenine nucleotides in liver mitochondria. Biochim Biophys Acta 104:312–315
Przybyla-Zawislak B, Dennis RA, Zakharkin SO, McCammon MT (1998) Genes of succinyl-CoA ligase from Saccharomyces cerevisiae. Eur J Biochem 258:736–743
Rotig A, Poulton J (2009) Genetic causes of mitochondrial DNA depletion in humans. Biochim Biophys Acta 1792:1103–1108
Shank RP, Bennett GS, Freytag SO, Campbell GL (1985) Pyruvate carboxylase: an astrocyte-specific enzyme implicated in the replenishment of amino acid neurotransmitter pools. Brain Res 329:364–367
Sonnewald U, Rae C (2010) Pyruvate carboxylation in different model systems studied by (13)C MRS. Neurochem Res 35:1916–1921
Strausberg RL, Feingold EA, Grouse LH, Derge JG, Klausner RD, Collins FS, Wagner L, Shenmen CM, Schuler GD, Altschul SF, Zeeberg B, Buetow KH, Schaefer CF, Bhat NK, Hopkins RF, Jordan H, Moore T, Max SI, Wang J, Hsieh F, Diatchenko L, Marusina K, Farmer AA, Rubin GM, Hong L, Stapleton M, Soares MB, Bonaldo MF, Casavant TL, Scheetz TE, Brownstein MJ, Usdin TB, Toshiyuki S, Carninci P, Prange C, Raha SS, Loquellano NA, Peters GJ, Abramson RD, Mullahy SJ, Bosak SA, McEwan PJ, McKernan KJ, Malek JA, Gunaratne PH, Richards S, Worley KC, Hale S, Garcia AM, Gay LJ, Hulyk SW, Villalon DK, Muzny DM, Sodergren EJ, Lu X, Gibbs RA, Fahey J, Helton E, Ketteman M, Madan A, Rodrigues S, Sanchez A, Whiting M, Madan A, Young AC, Shevchenko Y, Bouffard GG, Blakesley RW, Touchman JW, Green ED, Dickson MC, Rodriguez AC, Grimwood J, Schmutz J, Myers RM, Butterfield YS, Krzywinski MI, Skalska U, Smailus DE, Schnerch A, Schein JE, Jones SJ, Marra MA (2002) Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences. Proc Natl Acad Sci USA 99:16899–16903
Tanner S, Shen Z, Ng J, Florea L, Guigo R, Briggs SP, Bafna V (2007) Improving gene annotation using peptide mass spectrometry. Genome Res 17:231–239
Thomson M (1998) What are guanosine triphosphate-binding proteins doing in mitochondria? Biochim Biophys Acta 1403:211–218
Vozza A, Blanco E, Palmieri L, Palmieri F (2004) Identification of the mitochondrial GTP/GDP transporter in Saccharomyces cerevisiae. J Biol Chem 279:20850–20857
Wallace JC, Jitrapakdee S, Chapman-Smith A (1998) Pyruvate carboxylase. Int J Biochem Cell Biol 30:1–5
Westergaard N, Sonnewald U, Schousboe A (1994) Release of alpha-ketoglutarate, malate and succinate from cultured astrocytes: possible role in amino acid neurotransmitter homeostasis. Neurosci Lett 176:105–109
Yu AC, Drejer J, Hertz L, Schousboe A (1983) Pyruvate carboxylase activity in primary cultures of astrocytes and neurons. J Neurochem 41:1484–1487
Zhang Z, Tan M, Xie Z, Dai L, Chen Y, Zhao Y (2011) Identification of lysine succinylation as a new post-translational modification. Nat Chem Biol 7:58–63
Acknowledgments
Thanks are expressed to Katalin Zölde for excellent technical assistance.This work was supported by the Országos Tudományos Kutatási Alapprogram (OTKA) grants NNF2 85612, K 100319 to A.D., the Danish National Health Research Council grant 12-127702 to E.O., OTKA 81983 and the Hungarian Academy of Sciences grant 02001 to V. A.-V, TÁMOP 4.2.1./B-09/1/KMR and BIOINF09TÉT_10-1-2011-0058 to MJM, OTKA grants NNF 78905, NNF2 85658, K 100918, and the MTA-SE Lendület Neurobiochemistry Research Division grant 95003 to C.C.
Conflict of interest
The authors declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dobolyi, A., Ostergaard, E., Bagó, A.G. et al. Exclusive neuronal expression of SUCLA2 in the human brain. Brain Struct Funct 220, 135–151 (2015). https://doi.org/10.1007/s00429-013-0643-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-013-0643-2