Abstract
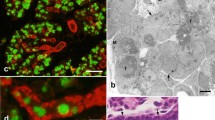
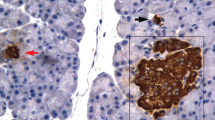
The mature rat parotid gland shows hardly any cell bodies of myoepithelial cells around the acini, only a few cell processes being visible. However, in the early postnatal period, the rat parotid gland shows many myoepithelial cell bodies around the acini, including the intercalated ducts. In order to clarify the reason for the disappearance of myoepithelial cells from the area around the acinus during postnatal development, changes in the number and distribution of myoepithelial cells in the rat parotid gland were examined histochemically and chronologically, with particular reference to cell proliferation and cell death. From day 7 to day 14, many myoepithelial cells showing a positive reaction with anti-actin antiserum were found around the acini and intercalated ducts, but thereafter the number of such cells decreased gradually, particularly around the acini, and had almost disappeared after day 35. BrdU/PCNA-positive myoepithelial cells surrounding the acini were easily detected on day 14, but disappeared by day 21, whereas BrdU/PCNA-positive acinar cells remained numerous even after day 21. TUNEL/ISEL staining showed no positive myoepithelial cells throughout the observation period. Transmission electron microscopy also demonstrated no myoepithelial cells with chromatin condensation characteristic of apoptosis through the observation period. These findings suggest that the main reason for the disappearance of myoepithelial cells from the area around the acinus during postnatal development is the large difference between the number of myoepithelial cells and that of acinar cells, because the acinar cells retain their proliferative activity even after myoepithelial cells have become quiescent.












Similar content being viewed by others
References
Deugnier M-A, Moiseyeva EP, Thiery JP Glukhova M (1995) Myoepithelial cell differentiation in the developing mammary gland: progressive acquisition of smooth muscle phenotype. Dev Dyn 201:107–117
Drenckhahn D, Groochel-Stewart U, Unsicker K (1997) Immunofluorescence-microscopic demonstration of myosin and actin in salivary glands and exocrine pancreas of the rat. Cell Tissue Res 183:273–279
Garrett JA, Emmelin N (1979) Activities of salivary myoepithelial cells: a review. Med Biol 57:1–24
Garrett JR, Parsons PA (1973) Alkaline phosphatase and myoepithelial cells in the parotid gland of the rat. Histochem J 5:463–471
Gugliotta P, Sapino A, Macri L, Skalli O, Gabbiani D, Bussolati G (1988) Specific demonstration of myoepithelial cells by anti-alpha smooth muscle actin antibody. J Histochem Cytochem 36:659–663
Ikeda R, Aiyama S (1997) Developmental changes in mucous cells of the early postnatal rat parotid gland: an ultrastructural and histochemical study. Arch Histol Cytol 60:185–193
Ikeda R, Aiyama S (1999) Developmental changes of sugar residues and secretory protein in mucous cells of the early postnatal rat parotid gland. Anat Rec 255:155–161
Komori A, Takagi I, Suzuki M, Ishikawa G (1979) Frequency and distribution of mucous cells in human parotid glands. J Stomatol Soc Jpn 46:11–18
Lawson KA (1970) Morphogenesis and functional differentiation of the rat parotid gland in vivo and in vitro. J Embryol Exp Morphol 24:411–424
Lazard D, Sastre X, Frid MG, Glukhova MA, Thiery JP, Koteliansky VE (1993) Expression of smooth muscle-specific proteins in myoepithelium and stromal myofibroblasts of normal and malignant human breast tissue. Proc Natl Acad Sci USA 90:999–1003
Longtine JA, Pinkus GS, Fujiwara K, Corson JM (1985) Immunohistochemical localization of smooth muscle myosin in normal human tissues. J Histochem Cytochem 33:179–184
Nagashima Y, Ono K (1985) Myoepithelial cell ultrastructure in the submandibular gland of man. Anat Embryol 171:259–265
Nagato T, Yoshida H, Yoshida A, Uehara Y (1980) A scanning electron microscope study of myoepithelial cells in exocrine glands. Cell Tissue Res 209:1–10
Ogawa Y, Yamauchi S, Ohnishi A, Ito R, Ijuhin N (1999) Immunohistochemistry of myoepithelial cells during development of the rat salivary glands. Anat Embryol 200:215–228
Pinkstaff CA (1980) The cytology of salivary glands. Int Rev Cytol 63:141–261
Redman RS (1994) Myoepithelium of salivary glands. Microsc Res Tech 27:25–45
Redman RS, Sreebny LM (1970) Proliferative behavior of differentiating cells in the developing rat parotid gland. J Cell Biol 46:81–87
Redman RS, Sreebny LM (1971) Morphologic and biochemical observations on the development of the rat parotid gland. Dev Biol 25:248–279
Redman RS, Sweney LR, Mclauglin ST (1980) Differentiation of myoepithelial cells in the developing rat parotid gland. Am J Anat 158:299–320
Riva A, Tandler B, Testa Riva F (1988) Ultrastructural observations on human sublingual gland. Am J Anat 181:385–392
Riva A, Valentino L, Lantini MS, Floris A, Testa-Riva F (1993) 3D-structure of cells of human salivary glands as seen by SEM. Microsc Res Tech 26:5–20
Satoh Y, Oomori Y, Ishikawa K, Ono K (1994) Configuration of myoepithelial cells in various exocrine glands of guinea pigs. Anat Embryol 189:227–236
Schneyer CA, Hall HD (1969) Growth pattern of postnatally developing rat parotid gland. Proc Soc Exp Biol Med 130:603–607
Segawa A (1994) Structure and development of normal salivary glands. J Dent Med 40:783–792
Segawa A, Shoi N, Yamashina S (1995) Function of myoepithelial cells in salivary secretion: reevaluation of the expulsion theory. Acta Anat Nippon 70:330–337
Shear M (1966) The structure and function of myoepithelial cells in salivary glands. Arch Oral Biol 11:769–780
Shear M (1968) Histological and histochemical studies of intercalated ducts in rat parotid glands. S Afr J Med Sci 33:97–103
Taga R, Sesso A (1979) Ultrastructural studies on developing parotid gland of the rat at early postnatal periods. Arch Histol Jpn 42:427–444
Acknowledgment
The authors deeply thank Mrs. Sumie Satoh for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsujimura, T., Ikeda, R. & Aiyama, S. Changes in the number and distribution of myoepithelial cells in the rat parotid gland during postnatal development. Anat Embryol 211, 567–574 (2006). https://doi.org/10.1007/s00429-006-0111-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-006-0111-3