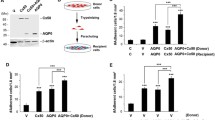
Abstract
The aim of the study was to examine gap-junction-mediated intercellular communication after experimentally induced aggregations of microtubules in cultured bovine lens epithelial cells. Intercellular communication between lens cells appears to be crucial for normal lens homeostasis. However, investigations on the maintenance of direct ion and metabolite exchange via gap junctions and its quantified dependency of cytoskeletal microtubules have not been available under conditions leading to bundling of microtubules. Thus, metabolic coupling of neighboring lens epithelial cells was quantified following microinjections of neurobiotin into single cells under various conditions. In controls, intensive gap-junction-mediated intercellular communication could be documented by dye-spreading of microinjected neurobiotin. In contrast, taxol treatment for 1–3 days impaired, but did not completely block gap-junction-mediated intercellular communication. After depletion of taxol, a complete recovery of intercellular communication was achieved. In addition, confocal laser scanning microscopy and rapid-freeze deep-etch electron microscopy revealed a displacement of actin-filaments from the perinuclear cytoplasm, accompanied by an abnormal aggregation of microtubules after taxol treatment, including impeded translocation of connexin 43 from the cytoplasm into the plasma membrane. Incubation of cells with nocodazole destroyed the microtubule network, accompanied by a clear reduction of plasma-membrane-integrated connexin 43 and significant impairment of dye spreading. Thus, in lens epithelial cells intercellular communication at gap junctions made by connexin 43 depends on the integrity of the microtubule network through the translocation of connexins to the plasma membrane.






Similar content being viewed by others
References
Berthoud VM, Cook AJ, Beyer EC (1994) Characterization of the gap junction protein connexin56 in the chicken lens by immunofluorescence and immunoblotting. Invest Ophthalmol Vis Sci 35:4109–4117
Berthoud VM, Bassnett S, Beyer EC (1999) Cultured chicken embryo lens cells resemble differentiating fiber cells in vivo and contain two kinetic pools of connexin 56. Exp Eye Res 68:475–484
Beyer EC, Kistler J, Paul DL, Goodenough DA (1989) Antisera directed against connexin 43 peptides react with a 43-kD protein localized to gap junctions in myocardium and other tissues. J Cell Biol 108:595–605
Bloomfield SA, Xin D, Persky SE (1995) A comparison of receptive field and tracer coupling size of horizontal cells in the rabbit retina. Vis Neurosci 12:985–999
Breipohl W, Leyendecker M, von Tiesenhausen K, Grus FH (1995) Effects of UV-B on the growth pattern of bovine passage I and II lens epithelial cells in vitro. Ophthalmic Res 27:62–68
Dahm R, van Marle J, Prescott AR, Quinlan RA (1999) Gap junctions containing alpha8-connexin (MP70) in the adult mammalian lens epithelium suggests a re-evaluation of its role in the lens. Exp Eye Res 69:45–56
Gao Y, Spray DC (1998) Structural changes in lenses of mice lacking the gap junction protein connexin43. Invest Ophthalmol Vis Sci 39: 1198–1209
George CH, Kendall JM, Evans WH (1999) Intracellular trafficking pathways in the assembly of connexins into gap junctions. J Biol Chem 274:8678–8685
Giepmans BN, Verlaan I, Hengeveld T, Janssen H, Calafat J, Falk MM, Moolenaar WH (2001) Gap junction protein connexin 43 interacts directly with microtubules. Curr Biol 11:1364–1368
Giessmann D, Theiss C, Breipohl W, Meller K (2003) Microinjection of anti-actin antibodies impaired gap junctional intercellular communication in lens epithelial cells in vitro. Curr Eye Res 27: 157–164
Goldberg GS, Lampe PD, Nicholson BJ (1999) Selective transfer of endogenous metabolites through gap junctions composed of different connexins. Nat Cell Biol 1:457–459
Gong X, Li E, Klier G, Huang Q, Wu Y, Lei H, Kumar NM, Horwitz J, Gilula NB (1997) Disruption of alpha3 connexin gene leads to proteolysis and cataractogenesis in mice. Cell 91:833–843
Goodenough DA (1979) Lens gap junctions: a structural hypothesis for nonregulated low-resistance intercellular pathways. Invest Ophthalmol Vis Sci 18:1104–1122
Goodenough DA (1992) The crystalline lens. A system networked by gap junctional intercellular communication. Semin Cell Biol 3:49–58
Graham RC, Karnovsky MJ (1966) The early stages of absorption of injected horseradish peroxidase in the proximal tubles of mouse kidney; ultrastructural cytochemistry by a new technique. J Histochem Cytochem 14:291–302
Hernandez Galindo EE, Theiss C, Steuhl KP, Meller D (2003) Gap junctional communication in microinjected human limbal and peripheral corneal epithelial cells cultured on intact amniotic membrane. Exp Eye Res 76:303–314
Jiang JX, White TW, Goodenough DA (1995) Changes in connexin expression and distribution during chick lens development. Dev Biol 168:649–661
Johnson RG, Meyer RA, Li XR, Preus DM, Tan L, Grunenwald H, Paulson AF, Laird DW, Sheridan JD (2002) Gap junctions assemble in the presence of cytoskeletal inhibitors, but enhanced assembly requires microtubules. Exp Cell Res 275:67–80
Kinsey VE, Reddy DVN (1964) Studies of the crystalline lens, XI: the relative role of the epithelium and capsule in transport. Invest Ophthalmol 4:104–116
Kistler JB, Kirkland B, Bullivant S (1985) Identification of a 70,000-D protein in lens membrane junctional domains. J Cell Biol 101:28–35
Kumar NM, Gilula NB (1996) The gap junction communication channel. Cell 84:381–388
Laird DW, Puranam KL, Revel JP (1991) Turnover and phosphorylation dynamics of connexin 43 gap junction protein in cultured cardiac myocytes. Biochem J 273:67–72
Lauf U, Giepmans BN, Lopez P, Braconnot S, Chen SC, Falk MM (2002) Dynamic trafficking and delivery of connexons to the plasma membrane and accretion to gap junctions in living cells. Proc Natl Acad Sci USA 99:10446–10451
Loewenstein WR (1981) Junctional intercellular communication: the cell-to-cell membrane channel. Physiol Rev 61:829–913
Lonchampt MO, Laurent M, Coutois Y, Trenchev P, Hughes RC (1976) Microtubules and microfilaments of bovine lens epithelial cells: electron microscopy and immunofluorescence staining with specific antibodies. Exp Eye Res 23:505–518
Martin PEM, Blundell G, Ahmad S, Errington RJ, Evans WH (2001) Multiple pathways in the trafficking and assembly of connexin 26, 32 and 43 into gap junction intercellular communication channels. J Cell Sci 114:3845–3855
Masurovsky EB, Peterson ER, Crain SM, Horwitz SB (1981) Microtubule arrays in taxol-treated mouse dorsal root ganglion-spinal cord cultures. Brain Res 217:392–398
Masurovsky EB, Peterson ER, Crain SM, Horwitz SB (1983) Morphological alterations in dorsal root ganglion neurons and supporting cells of organotypic mouse spinal cord-ganglion cultures exposed to taxol. Neuroscience 10:491–509
Mathias RT, Rae JL, Baldo GJ (1997) Physiological properties of the normal lens. Physiol Rev 77:21–50
Meller K (1989) Chromatolysis of dorsal root ganglion cells studied by cryofixation. Cell Tissue Res 256:59–66
Meller K (1992) Axoplasmic transport of horseradish peroxidase in single neurons of the dorsal root ganglion studied in vitro by microinjection. Cell Tissue Res 270:139–148
Meller K (1994) Intracellular injection of transport markers and antibodies into in vitro cultured nerve cells. Neurosci Protocols 50:1–11
Miller TM, Goodenough DA (1985) Gap junction structures after experimental alteration of junctional channel conductance. J Cell Biol 101:1741–1748
Mills SL, Massey SC (2000) A series of biotinylated tracers distinguishes three types of gap junction in retina. J Neurosci 20:8629–8636
Musil LS, Beyer EC, Goodenough DA (1990) Expression of the gap junction protein connexin 43 in embryonic chick lens: molecular cloning, ultrastructural localization, and post-translational phosphorylation. J Membr Biol 116:63–175
Parekh H, Simpkins H (1997) The transport and binding of taxol. Gen Pharmacol 29:167–172
Paul DL, Ebihara L, Takemoto LJ, Swenson KI, Goodenough DA (1991) Connexin 46, a novel lens gap junction protein, induces voltage-gated currents in nonjunctional plasma membrane of Xenopus oocytes. J Cell Biol 115:1077–1089
Paulson AF, Lampe PD, Meyer RA, TenBroek E, Atkinson MM, Walseth TF, Johnson RG (2000) Cyclic AMP and LDL trigger a rapid enhancement in gap junction assembly through a stimulation of connexin trafficking. J Cell Sci 113:3037–3049
Peracchia C (1978) Calcium effects on gap junction structure and cell coupling. Nature 271:669–671
Piatigorsky J, Webster H, Wollberg M (1972) Cell elongation in the cultured embryonic chick lens epithelium with and without protein synthesis. Involvement of microtubules. J Cell Biol 55:82–92
Rup DM, Veenstra RD, Wang HZ, Brink PR, Beyer EC (1993) Chick connexin-56, a novel lens gap junction protein. Molecular cloning and functional expression. J Biol Chem 268:706–712
Saez JC, Connor JA, Spray DC, Bennett MV (1989) Hepatocyte gap junctions are permeable to the second mess inositol 1,4,5-trisphosphate, and to calcium ions. Proc Natl Acad Sci USA 86:2708–2712
Saez JC, Nairn AC, Czernik AJ, Spray DC, Hertzberg EL, Greengard P, Bennett MVL (1990) Phosphorylation of connexin 32, a hepatocyte gap-junction protein, by cAMP-dependent protein kinase, protein kinase C and Ca2+/calmodulin-dependent protein kinase II. Eur J Biochem 192:263–273
Saleh SM, Takemoto DJ (2000) Overexpression of protein kinase C gamma inhibits gap junctional intercellular communication in the lens epithelial cells. Exp Eye Res 71:99–102
Schiff PB, Horwitz SB (1980) Taxol stabilizes microtubules in mouse fibroblast cells. Proc Natl Acad Sci USA 77:1561–1565
Spray DC, White RL, Campos de Carvalho A, Harris AL, Bennett MVL (1984) Gating of gap junction channels. Biophys J 45:219–230
TenBroek EM, Johnson R, Louis CF (1994) Cell-to-cell communication in a differentiating ovine lens culture system. Invest Ophthalmol Vis Sci 35:215–228
Theiss C, Meller K (2000) Taxol impairs anterograde axonal transport of microinjected horse radish peroxidase in dorsal root ganglia neurons in vitro. Cell Tissue Res 299:213–224
Theiss C, Meller K (2002a) Aluminum impairs gap junctional intercellular communication between astroglial cells in vitro. Cell Tissue Res 310: 143–154
Theiss C, Meller K (2002b) Microinjected anti-actin antibodies impaired cell-communication in astrocytes in vitro. Exp Cell Res 281:197–204
White TW, Bruzzone R, Goodenough DA, Paul DL (1992) Mouse Cx50, a functional member of the connexin family of gap junction proteins, is the lens fibre protein MP70. Mol Biol Cell 3:711–720
White TW, Goodenough DA, Paul DL (1998) Targeted ablation of connexin50 in mice results in microphthalmia and zonular pulverulent cataracts. J Cell Biol 143:815–825
Zampighi GA, Simon SA, Hall JE (1992) The specialized junctions of the lens. Int Rev Cytol 136:185–225
Acknowledgements
The authors especially thank M. Leyendecker for her help in setting up the initial tissue culture conditions and A. Ambrosat for secretarial work. We also wish to thank Dr. Schmidt and Dr. Masalme for providing eyes from slaughtered cattle.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Giessmann, D., Theiss, C., Breipohl, W. et al. Decreased gap junctional communication in neurobiotin microinjected lens epithelial cells after taxol treatment. Anat Embryol 209, 391–400 (2005). https://doi.org/10.1007/s00429-005-0456-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-005-0456-z