Abstract
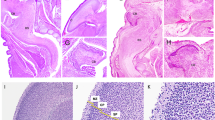
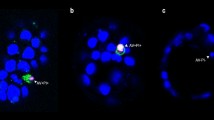
Cell death in the developing human spinal cord was investigated in 5–12 week human conceptuses using immunohistochemical and TUNEL methods. Expression of pro-apoptotic (Fas-receptor, caspase-3) and anti-apoptotic (bcl-2) markers and marker for internucleosomal fragmentation (TUNEL) were analysed in the cranial and caudal parts of the human spinal cord. In early developmental stages (5–6 weeks) of the cranial spinal cord, bcl-2 positive cells were seen in the ventricular zone and in the roof plate, while in the caudal part they were seen surrounding the central lumen. Subsequently, bcl-2 expression appeared in the basal plates of the grey matter and in the spinal ganglia, and from the seventh week on they also appeared in the intermediate horn of the grey matter. In the fetal period, bcl-2 expression appeared in the dorsal horns of the grey matter (9 weeks) but ceased in the ventricular zone (12 weeks) . In the trunk region, TUNEL-positive cells were found in ventricular and mantle zones along the whole length of the spinal cord. Caspase-3 positive cells and Fas-receptor positive cells appeared only in the grey matter of the cranial segments (head and trunk) of the spinal cord, but they were missing in the caudal parts. Caspase-3 dependant pathway, probably activated by Fas-receptor, seems to operate only in the cranial part of the human spinal cord. In the caudal (sacrococcygeal and tail) parts, cells seem to die by caspase-3 independent pathway. The interplay of pro-apoptotic and anti-apoptotic factors may be associated with cranial spinal cord morphogenesis, adjustment of cells number and selective survival of neurons, while in the caudal regions these factors cause massive cell death associated with regression of the caudal spinal cord.







Similar content being viewed by others
References
Barde YA (1989) Trophic factors and neuronal survival. Neuron 2(6):1525–1534
Casha S, Yu WR, Fehlings MG (2001) Oligodendroglial apoptosis occurs along degenerating axons and is associated with FAS and p75 expression following spinal cord injury in the rat. Neuroscience 103(1):203–218
Catala M (2002) Genetic control of caudal development. Clin Genet 61(2):89–96
Chen J, Graham SH, Nakayama M, Zhu RL, Jin K, Stetler A, Simon RP (1997) Apoptosis repressor genes bcl-2 and bcl-x-long are expressed in the rat brain following global ischemia. J Cereb Blood Flow Metab 17:2–10
Clarke PGH (1990) Developmental cell death. Anat Embryol 181:195–213
Coen L, du Pasquier D, Le Mevel S, Brown S, Tata J, Mazabraud A, Demeneix BA (2001) Xenopus Bcl-X(L) selectively protects Rohon-Beard neurons from metamorphic degeneration. Proc Natl Acad Sci USA 98(14):7869–7874
Dryden RJ (1980) Duplication of the spinal cord: a discussion of possible embyogenesis of diplomyelia. Dev Med Child Neurol 22:234–243
Fallon JF, Simandl BK (1978) Evidence of a role for cell death in the disappearance of the embryonic human tail. Am J Anat 152:111–130
FitzGerald MJT, FitzGerald M (1994) Human Embryology. Bailliere Tindal, London
Fujita E, Urase K, Egashira J, Miho Y, Isahara K, Uchiyama Y, Isoai A, Kumagai H, Kuida K, Motoyama N, Momoi T (2000) Detection of caspase-9 activation in the cell death of the Bcl-x-deficient mouse embryo nervous system by cleavage sites-directed antisera. Brain Res Dev Brain Res 122(2):135–147
Gont LK, Steinbeisser H, Blumberg B, DeRobertis EM (1993) Tail formation as a continuation of gastrulation: the multiple cell populations of the Xenopus tailbud derive from the late blastopore lip. Development 119:991–1004
Gonzales-Garcia M, Garcia I, Ding L, O’Shea S, Boise LH, Thompson CB, Nunez G (1995) Bcl-x is expressed in embryonic and postnatal neural tissues and functions to prevent neuronal cell death. Proc Natl Acad Sci 92:4304–4308
Griffith CM, Hay ED (1992) Epithelial-mesenchymal transformation during palatal fusion: carboxyfluorescein traces cells at light and electron microscopic levels. Development 116(4):1087–1099
Handrigan GR (2003) Concordia discors: duality in the origin of the vertebrate tail. J Anat 202:255–267
Jacobson KA, Moore SE, Yang B, Doherty P, Gordon GW, Walsh FS (1997) Cellular determinants of the lateral mobility of neural cell adhesion molecules. Biochim Biophys Acta 1330(2):138–144
Krajewska M, Mai JK, Zapata JM, Ashwell KW, Schendel SL, Reed JC, Krajewski S (2002) Dynamics of expression of apoptosis-regulatory proteins Bid, Bcl-2, Bcl-X, Bax and Bak during development of murine nervous system. Cell Death Differ 9(2):145–157
Li GL, Brodin G, Farooque M, Funa K, Holtz A, Wang WL, Olsson Y (1996) Apoptosis and expression of bcl-2 after compression trauma to the rat spinal cord. J Neuropathol Exp Neurol 55:280–289
Milligan CE, Schwartz LM (1996) Programmed cell death during development of animals. In: Holdbrook NJ, Martin GR, Lockshin RA (eds) Cellular aging and cell death. Wiley-Liss, New York, pp 181–208
Moore KL, Persaud TVN (1993) The developing human: clinically oriented embryology. Saunders, Philadelphia
Motoyama N, Wang F, Roth KA, Sawa H, Nakayama KI, Nakayama K, Negishi I, Senju S, Zhang Q, Fujii S, Loh DY (1995) Massive cell death of immature hematopoietic cells and neurons in Bcl-x-deficient mice. Science 267:1506–1510
Nagata S, Golstein P (1995) The Fas death factor. Science 267:1449–1455
Naruse I, Keino H (1995) Apoptosis in the developing CNS. Prog Neurobiol 47(2):135–155
Nievelstein RA, Hartwig NG, Vermelj-Keersc C, Valk J (1993) Embryonic development of the mammalian caudal neural tube. Teratology 48:21–31
Okuno S, Shimizu S, Ito T, Nomura M, Hamada E, Tsujimoto Y, Matsuda H (1998) Bcl-2 prevents caspase-independent cell death. J Biol Chem 273(51):34272–34277
Oppenheim RW (1989) The Neurotrophic theory and naturally occurring motoneuronal death. Trends Neurosc 12:252–255
O’Rahilly R, Muller F (1987) Developmental stages in human embryos. Carnegie Istit. of Washington 637:246
Raoul C, Henderson CE, Pettmann B (1999) Programmed cell death of embryonic motoneurons triggered through the Fas death receptor. J Cell Biol 147:1049–1061
Saraga-Babić M, Stefanović V, Saraga M, Wartiovaara J, Lehtonen E (2002) Expression of intermediate filaments and desmosomal proteins during differentiation of the human spinal cord. Acta Histochem 104(2):157–166
Sapunar D, Vilović K, England MA, Saraga-Babić M (2001). Morphological diversity of dying cells during regression of human tail. Ann Anat 183:217–222
Sato N, Sakuma C, Kato H, Milligan CE, Oppenheim RW, Yaginuma H (2002) Bcl-2 rescues motoneurons from early cell death in the cervical spinal cord of the chicken embryo. J Neurobiol 53(3):381–390
Schoenwolf GC (1977) Tail (end) bud contributions to the posterior region of the chick embryo. J Exp Zool 201:227–246
Schoenwolf GC (1978) Effects of complete tail bud extirpation on early development of the posterior region of the chick embryo. Anat Rec 192:289–296
Schoenwolf GC, Delongo J (1980) Ultrastructure of secondary neurulation in the chick embryo. Am J Anat 158(1):43–63
Schoenwolf GC (1984) Histological and ultrastructural studies of secondary neurulation in mouse embryos. Am J Anat 169(4):361–376
Schubert FR, Tremblay P, Mansouri A, Faisst AM, Kammandel B, Lumsden A, Gruss P, Dietrich S (2001) Early mesodermal phenotypes in splotch suggest a role for Pax3 in the formation of epithelial somites. Dev Dyn 222(3):506–521
Schweichel JU, Merker HJ (1973) The morphology of various types of cell death in prenatal tissues. Teratology 7:253–266
Tam PPL (1984) The histogenetic capacity of tissue in the caudal end of the embryonic axis of the mouse. J Embryol Exp Morphol 82:253–266
Vilović K, Sapunar D, Ilijić E, Mimica MD, England MA, Saraga-Babić M (2001). Morphological characteristics of daying cells in axial structures of developing human embryos. Cells Tissues Organs 169: 347–354
White CA, McCombe PA, Pender MP (1998) The roles of Fas, Fas ligand and Bcl-2 in T cell apoptosis in the central nervous system in experimental autoimmune encephalomyelitis. J Neuroimmunol 82(1):47–55
Wilson V, Beddington RS (1996) Cell fate and morphogenetic movement in the late mouse primitive streak. Mech Dev 55(1):79–89
Yachnis AT, Giovanini MA, Eskin TE, Reier PJ, Anderson DK (1998) Developmental patterns of BCL-2 and BCL-X polypeptide expression in the human spinal cord. Exp Neurol 150:82–97
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vilović, K., Ilijić, E., Glamočlija, V. et al. Cell death in developing human spinal cord. Anat Embryol 211, 1–9 (2006). https://doi.org/10.1007/s00429-005-0044-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-005-0044-2