Abstract
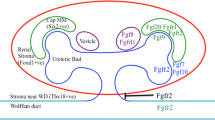
Members of the fibroblast growth factor (FGF) family play diverse roles during the development and patterning of various organs. In human and mice, 22 FGFs and four receptors derived from several splice variants are present. Redundant expression and function of FGF genes in organogenesis have been reported, but their roles in embryonic external genitalia, genital tubercle (GT), development have not been studied in detail. To address the role of FGF during external genitalia development, we have analyzed the expression of FGF genes (Fgf8, 9, 10) and receptor genes (Fgfr1, r2IIIb, r2IIIc) in GT of mice. Furthermore, Fgf10 and Fgfr2IIIb mutant mice were analyzed to elucidate their roles in embryonic external genitalia development. Fgfr2IIIb was expressed in urethral plate epithelium during GT development. Fgfr2IIIb mutant mice display urethral dysmorphogenesis. Marker gene analysis for urethral plate and bilateral mesenchymal formation suggests the existence of epithelial-mesenchymal interaction during urethral morphogenesis. Therefore, FGF10/FGFR2IIIb signals seem to constitute a developmental cascade for such morphogenesis.




Similar content being viewed by others
References
Arman E, Haffner-Krausz R, Gorivodsky M, Lonai P (1999) Fgfr2 is required for limb outgrowth and lung-branching morphogenesis. Proc Natl Acad Sci USA 96:11895–11899
Barlow AJ, Francis-West PH (1997) Ectopic application of recombinant BMP-2 and BMP-4 can change patterning of developing chick facial primordia. Development 124:391–398
Baskin LS (2004) Hypospadias. Adv Exp Med Biol 545:3-22
Baskin LS, Liu W, Bastacky J, Yucel S (2004) Anatomical studies of the mouse genital tubercle. Adv Exp Med Biol 545:103–121
Chen Y, Zhao X (1998) Shaping limbs by apoptosis. J Exp Zool 282:691–702
Dassule HR, McMahon AP (1998) Analysis of epithelial-mesenchymal interactions in the initial morphogenesis of the mammalian tooth. Dev Biol 202:215–227
De Moerlooze L, Spencer-Dene B, Revest J, Hajihosseini M, Rosewell I, Dickson C (2000) An important role for the IIIb isoform of fibroblast growth factor receptor 2 (FGFR2) in mesenchymal-epithelial signalling during mouse organogenesis. Development 127:483–492
Dolle P, Izpisua-Belmonte JC, Brown JM, Tickle C, Duboule D (1991) HOX-4 genes and the morphogenesis of mammalian genitalia. Genes Dev 5:1767–7
Dravis C, Yokoyama N, Chumley MJ, Cowan CA, Silvany RE, Shay J, Baker LA, Henkemeyer M (2004) Bidirectional signaling mediated by ephrin-B2 and EphB2 controls urorectal development. Dev Biol 271:272–290
Duboule D (1994) How to make a limb? Science 266:575–576
Eswarakumar VP, Monsonego-Ornan E, Pines M, Antonopoulou I, Morriss-Kay GM, Lonai P (2002) The IIIc alternative of Fgfr2 is a positive regulator of bone formation. Development 129:3783–3793
Ganan Y, Macias D, Duterque-Coquillaud M, Ros MA, Hurle JM (1996) Role of TGF beta s and BMPs as signals controlling the position of the digits and the areas of interdigital cell death in the developing chick limb autopod. Development 122:2349–2357
Grothe C, Brand-Saberi B, Wilting J, Christ B (1996) Fibroblast growth factor receptor 1 in skeletal and heart muscle cells: expression during early avian development and regulation after notochord transplantation. Dev Dyn 206:310–317
Hajihosseini MK, Lalioti MD, Arthaud S, Burgar HR, Brown JM, Twigg SR, Wilkie AO, Heath JK (2004) Skeletal development is regulated by fibroblast growth factor receptor 1 signalling dynamics. Development 131:325–335
Haraguchi R, Mo R, Hui C, Motoyama J, Makino S, Shiroishi T, Gaffield W, Yamada G (2001) Unique functions of Sonic hedgehog signaling during external genitalia development. Development 128:4241–4250
Haraguchi R, Suzuki K, Murakami R, Sakai M, Kamikawa M, Kengaku M, Sekine K, Kawano H, Kato S, Ueno N, Yamada G (2000) Molecular analysis of external genitalia formation: the role of fibroblast growth factor (Fgf) genes during genital tubercle formation. Development 127:2471–2479
Hogan BL (1999) Morphogenesis. Cell 96:225–233
Hsu TY, Chang SY, Wang TJ, Ou CY, Chen ZH, Hsu PH (2001) Prenatal sonographic appearance of Beare-Stevenson cutis gyrata syndrome: two- and three-dimensional ultrasonographic findings. Prenat Diagn 21:665–667
Johnson RL, Tabin CJ (1997) Molecular models for vertebrate limb development. Cell 90:979–990
Kawakami Y, Capdevila J, Buscher D, Itoh T, Rodriguez Esteban C, Izpisua Belmonte JC (2001) WNT signals control FGF-dependent limb initiation and AER induction in the chick embryo. Cell 104:891–900
Kurzrock EA, Baskin LS, Li Y, Cunha GR (1999) Epithelial-mesenchymal interactions in development of the mouse fetal genital tubercle. Cells Tissues Organs 164:125–130
Macatee TL, Hammond BP, Arenkiel BR, Francis L, Frank DU, Moon AM (2003) Ablation of specific expression domains reveals discrete functions of ectoderm- and endoderm-derived FGF8 during cardiovascular and pharyngeal development. Development 130:6361–6374
Min H, Danilenko DM, Scully SA, Bolon B, Ring BD, Tarpley JE, DeRose M, Simonet WS (1998) Fgf-10 is required for both limb and lung development and exhibits striking functional similarity to Drosophila branchless. Genes Dev 12:3156–3161
Morgan EA, Nguyen SB, Scott V, Stadler HS (2003) Loss of Bmp7 and Fgf8 signaling in Hoxa13-mutant mice causes hypospadia. Development 130:3095–3109
Murakami R, Mizuno T (1986) Proximal-distal sequence of development of the skeletal tissues in the penis of rat and the inductive effect of epithelium. J Embryol Exp Morphol 92:133–143
Naiche LA, Papaioannou VE (2003) Loss of Tbx4 blocks hindlimb development and affects vascularization and fusion of the allantois. Development 130:2681–2693
Ng JK, Kawakami Y, Buscher D, Raya A, Itoh T, Koth CM, Rodriguez Esteban C, Rodriguez-Leon J, Garrity DM, Fishman MC, Izpisua Belmonte JC (2002) The limb identity gene Tbx5 promotes limb initiation by interacting with Wnt2b and Fgf10. Development 129:5161–5170
Ohuchi H, Hori Y, Yamasaki M, Harada H, Sekine K, Kato S, Itoh N (2000) FGF10 acts as a major ligand for FGF receptor 2 IIIb in mouse multi-organ development. Biochem Biophys Res Commun 277:643–649
Ohuchi H, Nakagawa T, Yamamoto A, Araga A, Ohata T, Ishimaru Y, Yoshioka H, Kuwana T, Nohno T, Yamasaki M, Itoh N, Noji S (1997) The mesenchymal factor, FGF10, initiates and maintains the outgrowth of the chick limb bud through interaction with FGF8, an apical ectodermal factor. Development 124:2235–2244
Ornitz DM, Itoh N (2001) Fibroblast growth factors. Genome Biol 2:REVIEWS3005
Ornitz DM, Xu J, Colvin JS, McEwen DG, MacArthur CA, Coulier F, Gao G, Goldfarb M (1996) Receptor specificity of the fibroblast growth factor family. J Biol Chem 271:15292–15297
Orr-Urtreger A, Givol D, Yayon A, Yarden Y, Lonai P (1991) Developmental expression of two murine fibroblast growth factor receptors, flg and bek. Development 113:1419-1434
Othman-Hassan K, Patel K, Papoutsi M, Rodriguez-Niedenfuhr M, Christ B, Wilting J (2001) Arterial identity of endothelial cells is controlled by local cues. Dev Biol 237:398–409
Partanen J, Schwartz L, Rossant J (1998) Opposite phenotypes of hypomorphic and Y766 phosphorylation site mutations reveal a function for Fgfr1 in anteroposterior patterning of mouse embryos. Genes Dev 12:2332–2344
Perriton CL, Powles N, Chiang C, Maconochie MK, Cohn MJ (2002) Sonic hedgehog signaling from the urethral epithelium controls external genital development. Dev Biol 247:26–46
Peters KG, Werner S, Chen G, Williams LT (1992) Two FGF receptor genes are differentially expressed in epithelial and mesenchymal tissues during limb formation and organogenesis in the mouse. Development 114:233–243
Plikus M, Wang WP, Liu J, Wang X, Jiang TX, Chuong CM (2004) Morpho-regulation of ectodermal organs: integument pathology and phenotypic variations in K14-Noggin engineered mice through modulation of bone morphogenic protein pathway. Am J Pathol 164:1099–1114
Powers CJ, McLeskey SW, Wellstein A (2000) Fibroblast growth factors, their receptors and signaling. Endocr Relat Cancer 7:165–197
Revest JM, Spencer-Dene B, Kerr K, De Moerlooze L, Rosewell I, Dickson C (2001) Fibroblast growth factor receptor 2-IIIb acts upstream of Shh and Fgf4 and is required for limb bud maintenance but not for the induction of Fgf8, Fgf10, Msx1, or Bmp4. Dev Biol 231:47–62
Sekine K, Ohuchi H, Fujiwara M, Yamasaki M, Yoshizawa T, Sato T, Yagishita N, Matsui D, Koga Y, Itoh N, Kato S (1999) Fgf10 is essential for limb and lung formation. Nat Genet 21:138–141
Suzuki K, Bachiller D, Chen YP, Kamikawa M, Ogi H, Haraguchi R, Ogino Y, Minami Y, Mishina Y, Ahn K, Crenshaw EB, III, Yamada G (2003) Regulation of outgrowth and apoptosis for the terminal appendage: external genitalia: development by concerted actions of BMP signaling. Development 130:6209–6220
Suzuki K, Ogino Y, Murakami R, Satoh Y, Bachiller D, Yamada G (2002) Embryonic development of mouse external genitalia: insights into a unique mode of organogenesis. Evol Dev 4:133–141
Thesleff I, Partanen AM, Vainio S (1991) Epithelial-mesenchymal interactions in tooth morphogenesis: the roles of extracellular matrix, growth factors, and cell surface receptors. J Craniofac Genet Dev Biol 11:229–237
Tickle C, Eichele G (1994) Vertebrate limb development. Ann Rev Cell Biol 10:121–152
Tucker AS, Yamada G, Grigoriou M, Pachnis V, Sharpe PT (1999) Fgf-8 determines rostral-caudal polarity in the first branchial arch. Development 126:51–61
van der Werff JF, Nievelstein RA, Brands E, Luijsterburg AJ, Vermeij-Keers C (2000) Normal development of the male anterior urethra. Teratology 61:172–183
Vargas RA, Maegawa GH, Taucher SC, Leite JC, Sanz P, Cifuentes J, Parra M, Munoz H, Maranduba CM, Passos-Bueno MR (2003) Beare-Stevenson syndrome: Two South American patients with FGFR2 analysis. Am J Med Genet 121:41–46
Wilkinson DG (1995) RNA detection using non-radioactive in situ hybridization. Curr Opin Biotechnol 6:20–23
Wright TJ, Mansour SL (2003) Fgf3 and Fgf10 are required for mouse otic placode induction. Development 130:3379–3390
Xu X, Weinstein M, Li C, Naski M, Cohen RI, Ornitz DM, Leder P, Deng C (1998) Fibroblast growth factor receptor 2 (FGFR2)-mediated reciprocal regulation loop between FGF8 and FGF10 is essential for limb induction. Development 125:753–765
Yamada G, Satoh Y, Baskin LS, Cunha GR (2003) Cellular and molecular mechanisms of development of the external genitalia. Differentiation 71:445–460
Yamaguchi TP, Bradley A, McMahon AP, Jones S (1999) A Wnt5a pathway underlies outgrowth of multiple structures in the vertebrate embryo. Development 126:1211–1223
Yonei-Tamura S, Endo T, Yajima H, Ohuchi H, Ide H, Tamura K (1999) FGF7 and FGF10 directly induce the apical ectodermal ridge in chick embryos. Dev Biol 211:133–143
Acknowledgements
We would like to thank Drs. John K. Heath, Clive Dickson, Tsutomu Ogata, Shigeaki Kato, Janet Rossant, Chi-Chung Hui, Yiping Chen, Gail Martin, Cheng-Ming Chuong, Brigid Hogan, Uli Ruether, Karen M. Lyons, Elaine Fuchs, Nobuyuki Itoh, Anne M. Moon, and David Ornitz for reagents and/or suggestions, and Takashi Tanaharu, Ryu Gejima, Daisuke Sasagawa, and Shiho Kitagawa for assistance. This research was supported by a Grant-in-Aid for Scientific Research on Priority Areas (1), General Promotion of Cancer Research in Japan, Mechanisms of Sex Differentiation, and by the 21st Century COE Research Program and Child Health and Development (14C-1) from the Ministry of Health, Labour and Welfare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Satoh and R. Haraguchi contributed equally to this work
Rights and permissions
About this article
Cite this article
Satoh, Y., Haraguchi, R., Wright, T.J. et al. Regulation of external genitalia development by concerted actions of FGF ligands and FGF receptors. Anat Embryol 208, 479–486 (2004). https://doi.org/10.1007/s00429-004-0419-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-004-0419-9