Abstract
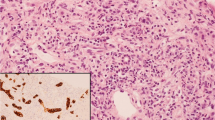
Hemophagocytic lymphohistiocytosis (HLH) is a rare disease with high mortality. Liver involvement is common (based on elevated liver function tests) with most patients demonstrating acute hepatitis. Liver biopsies are frequently obtained in the setting of suspected HLH for the purpose of identification of erythrophagocytosis, and if present, this finding is thought to suggest or support the diagnosis of HLH. However, there are problems with this approach; in particular, we do not know whether this finding is reproducible or whether it is specific to HLH. Therefore, we conducted a multi-institutional study in which experienced liver pathologists reviewed images taken from liver biopsies from patients with normal liver, acute hepatitis, possible HLH, and clinical HLH to determine if there was agreement about the presence or absence of erythrophagocytosis, and to ascertain whether the finding corresponds to a clinical diagnosis of HLH. Twelve liver pathologists reviewed 141 images in isolation (i.e., no clinical information or diagnosis provided). These came from 32 patients (five normal, 17 acute hepatitis, six HLH, four possible HLH). The pathologists classified each image as negative, equivocal, or positive for erythrophagocytosis. Kappa was .08 (no agreement) for case-level and 0.1 for image-level (1.4% agreement, based on two images which were universally considered negative). There was no difference in the proportion of pathologists who diagnosed erythrophagocytosis among those with different diagnoses at case or image-level (p = 0.82 and p = 0.82, respectively). Thus, erythrophagocytosis is an entirely unreliable histologic parameter in liver, as it is irreproducible and not demonstrably associated with a clinical disease (namely, HLH). Unless and until more reliable guidelines can be established, pathologists should refrain from commenting on the presence or absence of erythrophagocytosis in liver biopsy.





Similar content being viewed by others
Data Availability
Raw data was collected at Columbia University. Derived data supporting the findings of this study are available from the corresponding author SL upon reasonable request.
References
Birndt S, Schenk T, Heinevetter B et al (2020) Hemophagocytic lymphohistiocytosis in adults: collaborative analysis of 137 cases of a nationwide German registry. J Cancer Res Clin Oncol 146(4):1065–1077. https://doi.org/10.1007/s00432-020-03139-4
Risma K, Jordan MB (2012) Hemophagocytic lymphohistiocytosis: updates and evolving concepts. Curr Opin Pediatr 24(1):9–15. https://doi.org/10.1097/MOP.0b013e32834ec9c1
Kikuchi A, Singh K, Gars E, Ohgami RS (2022) Pathology updates and diagnostic approaches to haemophagocytic lymphohistiocytosis. Histopathology 80(4):616–626. https://doi.org/10.1111/his.14591
ÖstNilsson‐Ardnor, Henter (1998) Autopsy findings in 27 children with haemophagocytic lymphohistiocytosis. Histopathology 32(4):310–316. https://doi.org/10.1046/j.1365-2559.1998.00377.x
Tsui WMS, Wong KF, Tse CCH (2008) Liver changes in reactive haemophagocytic syndrome. Liver 12(6):363–367. https://doi.org/10.1111/j.1600-0676.1992.tb00590.x
De Kerguenec C, Hillaire S, Molinie V et al (2001) Hepatic manifestations of hemophagocytic syndrome: a study of 30 cases. Am J Gastroenterol 96(3):852–857. https://doi.org/10.1111/j.1572-0241.2001.03632.x
Eife R, Janka GE, Belohradsky BH, Holtmann H (1989) Natural killer cell function and interferon production in familial hemophagocyticlymphohistiocytosis. Pediatr Hematol Oncol 6(3):265–272. https://doi.org/10.3109/08880018909034296
Henter JI, Elinder G, Ost A (1991) Diagnostic guidelines for hemophagocytic lymphohistiocytosis. The FHL Study Group of the Histiocyte Society. Semin Oncol 18(1):29–33
Henter JI, Horne A, Aricó M et al (2007) HLH-2004: Diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatr Blood Cancer 48(2):124–131. https://doi.org/10.1002/pbc.2103
Fardet L, Galicier L, Lambotte O et al (2014) Development and validation of the H score, a score for the diagnosis of reactive hemophagocytic syndrome: score for reactive hemophagocytic syndrome. Arthritis Rheumatol 66(9):2613–2620. https://doi.org/10.1002/art.38690
Debaugnies F, Mahadeb B, Ferster A et al (2016) Performances of the H-score for diagnosis of hemophagocytic lymphohistiocytosis in adult and pediatric patients. Am J Clin Pathol 145(6):862–870. https://doi.org/10.1093/ajcp/aqw076
Cron RQ, Behrens EM, Shakoory B, Ramanan AV, Chatham WW (2015) Does viral hemorrhagic fever represent reactive hemophagocytic syndrome? J Rheumatol 42(7):1078–1080. https://doi.org/10.3899/jrheum.150108
Li J, Wang Q, Zheng W et al (2014) Hemophagocytic lymphohistiocytosis: clinical analysis of 103 adult patients. Medicine (Baltimore) 93(2):100–105. https://doi.org/10.1097/MD.0000000000000022
Campo M, Berliner N (2015) Hemophagocytic lymphohistiocytosis in adults. Hematol Oncol Clin North Am 29(5):915–925. https://doi.org/10.1016/j.hoc.2015.06.009
Chen JH, Fleming MD, Pinkus GS et al (2010) Pathology of the liver in familial hemophagocytic lymphohistiocytosis. Am J Surg Pathol 34(6):852–867. https://doi.org/10.1097/PAS.0b013e3181dbbb17
Fujiwara F, Hibi S, Imashuku S (1993) Hypercytokinemia in hemophagocytic syndrome. J Pediatr Hematol Oncol 15(1):92–98. https://doi.org/10.1097/00043426-199302000-00012
Bilzer M, Roggel F, Gerbes AL (2006) Role of Kupffer cells in host defense and liver disease. Liver Int 26:1175–1186. https://doi.org/10.1111/j.1478-3231.2006.01342.x1175
(2017) R: a language and environment for statistical computing. Author: R Core Team Organization: R foundation for statistical computing. Location: Vienna, Austria. Year. url: https://www.R-project.org/
Prendki V, Stirnemann J, Lemoine M et al (2011) Prevalence and clinical Significance of Küpffer cell hyperplasia with hemophagocytosis in liver biopsies. Am J Surg Pathol 35(3):337–345. https://doi.org/10.1097/PAS.0b013e318209c681
Desai N, Del Portillo A, Fazlollahi L, Lee MJ, Remotti HE, Kudose S, Lagana SM (2023) Hemophagocytosis (or Pseudohemophagocytosis) is questionably reproducible in liver biopsy but is common in cases of acute hepatitis without clinical hemophagocytic lymphohistiocytosis. Liver Pathology. Laboratory Investigation 103(3). https://doi.org/10.1016/j.labinv.2023.100095
Barry M, Sinha SK, Leader MB, Kay EW (2001) Poor agreement in recognition of abnormal mitoses: requirement for standardized and robust definitions. Histopathology 38(1):68–72. https://doi.org/10.1046/j.1365-2559.2001.01034.x
O’Malley FP, Mohsin SK, Badve S, Bose S, Collins LC, Ennis M, Kleer CG, Pinder SE, Schnitt SJ (2006) Interobserver reproducibility in the diagnosis of flat epithelial atypia of the breast. Mod Pathol 19(2):172–179. https://doi.org/10.1038/modpathol.3800514
Tsuda H, Yoshida M, Akiyama F, Ohi Y, Kinowaki K, Kumaki N, Kondo Y, Saito A, Sasaki E, Nishimura R, Fujii S, Homma K, Horii R, Murata Y, Itami M, Kajita S, Kato H, Kurosumi M, Sakatani T, Shimizu S, Taniguchi K, Tamiya S, Nakamura H, Kanbayashi C, Shien T, Iwata H (2021) Nuclear grade and comedo necrosis of ductal carcinoma in situ as histopathological eligible criteria for the Japan Clinical Oncology Group 1505 trial: an interobserver agreement study. Jpn J Clin Oncol 51(3):434–443. https://doi.org/10.1093/jjco/hyaa235
Bilzer M, Roggel F, Gerbes AL (2006) Role of Kupffer cells in host defense and liver disease. Liver Int 26(10):1175–1186. https://doi.org/10.1111/j.1478-3231.2006.01342.x
Downs-Kelly E, Mendelin JE, Bennett AE, Castilla E, Henricks WH, Schoenfield L, Skacel M, Yerian L, Rice TW, Rybicki LA, Bronner MP, Goldblum JR (2008) Poor interobserver agreement in the distinction of high-grade dysplasia and adenocarcinoma in pretreatment Barrett’s esophagus biopsies. Am J Gastroenterol 103(9):2333–40. https://doi.org/10.1111/j.1572-0241.2008.02020.x. (quiz 2341)
Alpert L, Setia N, Ko HM, Lagana SM, Pittman ME, Johncilla M, Drage MG, Zhao L, Salomao MA, Liao X, Choi WT, Jenkins SM, Hart J, Harpaz N, Voltaggio L, Lauwers GY, Odze R, Remotti H, Smyrk TC, Graham RP (2021) Interobserver agreement and the impact of mentorship on the diagnosis of inflammatory bowel disease-associated dysplasia among subspecialist gastrointestinal pathologists. Virchows Arch 478(6):1061–1069. https://doi.org/10.1007/s00428-020-02998-z
Funding
SK is being supported by the Young Investigator Grant of the National Kidney Foundation. This work was supported, in part, by the Intramural Research Program of the National Institutes of Health, National Cancer Institute.
Author information
Authors and Affiliations
Contributions
Niyati Desai: conceptualization, methodology, data curation, writing—original draft, writing—review and editing, visualization. Satoru Kudose: methodology, data curation, writing—original draft, writing—review and editing. Helen E. Remotti: case review, writing—review and editing. Armando Del Portillo: case review, writing—review and editing. Ladan Fazlollahi: case review, writing—review and editing. Michael J. Lee: case review, writing—review and editing. Yuqing Xiong: writing—review and editing. Roger K. Moreira: case review, writing—review and editing. Marcela Salomao: case review, writing—review and editing. Maria Isabel Fiel: case review, writing—review and editing. Raul S. Gonzalez: case review, writing—review and editing. Joseph Misdraji: case review, writing—review and editing. Ryan M. Gill: case review, writing—review and editing. John Hart: case review, writing—review and editing. David E. Kleiner: case review, writing—review and editing. Uta Drebber: case review, writing—review and editing. Andrew M. Bellizzi: case review, writing—review and editing. Stephen M. Lagana: conceptualization, methodology, case review, data curation, writing—original draft, writing—review and editing, visualization, supervision, project administration.
Corresponding author
Ethics declarations
The study complies with ethical standards and was approved by the Columbia University Irving Medical Center IRB.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Desai, N., Kudose, S., Remotti, H.E. et al. Erythrophagocytosis is not a reproducible finding in liver biopsies, and is not associated with clinical diagnosis of hemophagocytic lymphohistiocytosis. Virchows Arch 484, 61–69 (2024). https://doi.org/10.1007/s00428-023-03683-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-023-03683-7