Abstract
Lymph nodes with acellular mucin harvested from treated colorectal cancers (CRC) are staged as pN0. However, there is variability among pathologists while reporting the pN stage when acellular mucin is found within nodes of untreated CRCs. While the UICC guidelines suggest staging them as pN1, the AJCC and CAP do not offer any recommendations. In order to characterize their clinicopathologic features and outcome, we compared 16 untreated CRCs (study group; mean age: 68 years) harboring nodes with acellular mucin with 34 pN0 and 25 pN1 untreated CRC controls. All tumors were unifocal; 12 (75%) were right-sided lesions. Most cases (75%) showed one node with acellular mucin (range: 1–3). MMR-deficient tumors were significantly more common in the study group (83%) compared to pN0 (33%; p = 0.006) and pN1 controls (8%; p < 0.001). The overall survival of study group patients was closer to pN0 compared to pN1 controls; however, this difference was not statistically significant. In conclusion, untreated CRC that harbor acellular mucin within lymph nodes commonly present as right-sided, MMR-deficient tumors in older women that show a non-mucinous phenotype. While the limited number of cases precludes us from making any formal recommendations about staging, we suggest that the finding of acellular mucin in a node should prompt evaluation of deeper levels (with or without cytokeratin immunohistochemistry) and submission of all pericolonic fat for additional lymph node harvest. Whether acellular mucin in nodes of untreated CRCs is related to the indolent biology of the disease, a robust local immune response or MMR deficiency requires further investigation.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Booth CM, Nanji S, Wei X, Peng Y, Biagi JJ, Hanna TP, Krzyzanowska MK, Mackillop WJ (2016) Use and effectiveness of adjuvant chemotherapy for stage III colon cancer: a population-based study. J Natl Compr Cancer Netw 14:47–56. https://doi.org/10.6004/jnccn.2016.0006
Haller DG, Catalano PJ, Macdonald JS, O'Rourke MA, Frontiera MS, Jackson DV, Mayer RJ (2005) Phase III study of fluorouracil, leucovorin, and levamisole in high-risk stage II and III colon cancer: final report of Intergroup 0089. J Clin Oncol 23:8671–8678. https://doi.org/10.1200/JCO.2004.00.5686
Sargent D, Sobrero A, Grothey A, O'Connell MJ, Buyse M, Andre T, Zheng Y, Green E, Labianca R, O'Callaghan C, Seitz JF, Francini G, Haller D, Yothers G, Goldberg R, de Gramont A (2009) Evidence for cure by adjuvant therapy in colon cancer: observations based on individual patient data from 20,898 patients on 18 randomized trials. J Clin Oncol 27:872–877. https://doi.org/10.1200/JCO.2008.19.5362
Franke AJ, Parekh H, Starr JS, Tan SA, Iqbal A, George TJ Jr (2018) Total neoadjuvant therapy: a shifting paradigm in locally advanced rectal cancer management. Clin Colorectal Cancer 17:1–12. https://doi.org/10.1016/j.clcc.2017.06.008
Jessup JM, Goldberg RM, Asare EA et al (2017) Colon and rectum. In: Amin MB (ed) AJCC Cancer Staging Manual, 8th edn. Springer, Chicago (IL), pp 251–274
College of American Pathologists (CAP) protocol for the examination of resection specimens from patients with primary carcinoma of the colon and rectum. https://documents.cap.org/protocols/cp-gilower-colonrectum-resection-20-4100.pdf. Accessed 10/6/2021
Smith KD, Tan D, Das P, Chang GJ, Kattepogu K, Feig BW, Skibber JM, Rodriguez-Bigas MA (2010) Clinical significance of acellular mucin in rectal adenocarcinoma patients with a pathologic complete response to preoperative chemoradiation. Ann Surg 251:261–264. https://doi.org/10.1097/SLA.0b013e3181bdfc27
Wittekind C B, JD, Compton C, Sobin L (2021) https://www.uicc.org/sites/main/files/atoms/files/TNM_FAQs_EN.pdf - TNM Supplement: A Commentary on Uniform Use Accessed February 17, 2021
Fortea-Sanchis C, Martinez-Ramos D, Escrig-Sos J (2018) The lymph node status as a prognostic factor in colon cancer: comparative population study of classifications using the logarithm of the ratio between metastatic and nonmetastatic nodes (LODDS) versus the pN-TNM classification and ganglion ratio systems. BMC Cancer 18:1208. https://doi.org/10.1186/s12885-018-5048-4
Holleczek B, Rossi S, Domenic A, Innos K, Minicozzi P, Francisci S, Hackl M, Eisemann N, Brenner H, Group E-W (2015) On-going improvement and persistent differences in the survival for patients with colon and rectum cancer across Europe 1999-2007 - results from the EUROCARE-5 study. Eur J Cancer 51:2158–2168. https://doi.org/10.1016/j.ejca.2015.07.024
NCCN (2021) National Comprehensive Cancer Network Esophageal and Esophagogastric Junction Cancers https://www.nccn.org/store/login/login.aspx?ReturnURL=https://www.nccn.org/professionals/physician_gls/pdf/esophageal.pdf. Accessed 10/6/2021
Rullier A, Laurent C, Vendrely V, Le Bail B, Bioulac-Sage P, Rullier E (2005) Impact of colloid response on survival after preoperative radiotherapy in locally advanced rectal carcinoma. Am J Surg Pathol 29:602–606. https://doi.org/10.1097/01.pas.0000153120.80385.29
Shia J, McManus M, Guillem JG, Leibold T, Zhou Q, Tang LH, Riedel ER, Weiser MR, Paty PB, Temple LK, Nash G, Kolosov K, Minsky BD, Wong WD, Klimstra DS (2011) Significance of acellular mucin pools in rectal carcinoma after neoadjuvant chemoradiotherapy. Am J Surg Pathol 35:127–134. https://doi.org/10.1097/PAS.0b013e318200cf78
de Campos-Lobato LF, Dietz DW, Stocchi L, Vogel JD, Lavery IC, Goldblum JR, Skacel M, Pelley RJ, Kalady MF (2012) Clinical implications of acellular mucin pools in resected rectal cancer with pathological complete response to neoadjuvant chemoradiation. Color Dis 14:62–67. https://doi.org/10.1111/j.1463-1318.2010.02532.x
Karamchandani DM, Chetty R, King TS, Liu X, Westerhoff M, Yang Z, Yantiss RK, Driman DK (2020) Challenges with colorectal cancer staging: results of an international study. Mod Pathol 33:153–163. https://doi.org/10.1038/s41379-019-0344-3
Kirsch RMDSN, Dawson H, Driman DK (2019) Wide variability in assessment and reporting of colorectal cancer specimens among North American pathologists: results of a Canada-US survey Can. J Pathol 11:59–69
Foong KS, Mishra A, Guy R, Wang LM, Shepherd NA (2016) How do we stage acellular mucin in lymph nodes of colorectal cancer specimens without neo-adjuvant therapy? Histopathology 69:527–528. https://doi.org/10.1111/his.12970
Gandhi J, Avery N, Strang J (2015) Acellular mucinous lymph nodes in colonic mucinous adenocarcinoma. Int Arch Med 8:1–3
Cohen R, Buhard O, Cervera P, Hain E, Dumont S, Bardier A, Bachet JB, Gornet JM, Lopez-Trabada D, Dumont S, Kaci R, Bertheau P, Renaud F, Bibeau F, Parc Y, Vernerey D, Duval A, Svrcek M, Andre T (2017) Clinical and molecular characterisation of hereditary and sporadic metastatic colorectal cancers harbouring microsatellite instability/DNA mismatch repair deficiency. Eur J Cancer 86:266–274. https://doi.org/10.1016/j.ejca.2017.09.022
Popat S, Hubner R, Houlston RS (2005) Systematic review of microsatellite instability and colorectal cancer prognosis. J Clin Oncol 23:609–618. https://doi.org/10.1200/JCO.2005.01.086
Author information
Authors and Affiliations
Contributions
Study concept, design, and draft preparation: JEL, LR, VD, JS, DTP
Sample retrieval, laboratory work, data collection: JEL, AK, CF, HL, KP, EL, JS, VD, MM, DTP
Interpretation of data, scoring, analysis: JEL, LR, AK, CF, HL, KP, EL, JS, VD, MM, and DTP
Critical revision, intellectual contribution, and supervision: JEL, LR, AK, CF, HL, KP, EL, JS, VD, MM, and DTP
All authors have approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Alok A. Khorana, M.D. - Receives Institutional grant support from NIH, CDC, Leap, BMS, Celgene, Merck; Consulting fees from Janssen, Bayer, Pfizer, Anthos, Sanofi, BMS, Seagen, honorarium from Medscape for CME events, Support for attending meetings and/or travel from Janssen, Bayer, Seagen, BMS; Participated on a Data Safety Monitoring Board or Advisory Board - Bayer, BMS, Janssen; Medical and Scientific Advisory Board, NBCA (unpaid). All remaining authors have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information

Supplementary Fig. 1
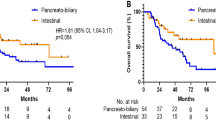
Kaplan-Meier curve comparing survival outcomes of all MMR-D tumors in three groups shows that the clinical outcome of patients with acellular mucin (cases; study group) was more similar to pN0 controls compared to pN+ (N1) colon cancer controls. The number of cases and events in this sub-analysis were too low to calculate p values. (PNG 11 kb)
Rights and permissions
About this article
Cite this article
Lapinski, J.E., Khorana, A.A., Rybicki, L. et al. Acellular mucin in lymph nodes isolated from treatment-naïve colorectal cancer resections: a clinicopathologic analysis of 16 cases. Virchows Arch 481, 63–72 (2022). https://doi.org/10.1007/s00428-022-03332-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-022-03332-5