Abstract
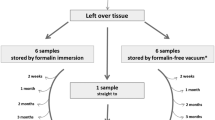
Diagnostic pathology activities are largely based on fixation of tissues in 4% formaldehyde, which has recently been re-classified as a carcinogenic compound and banned in several countries. Hospitals that do not have in-house pathology services need to send surgical and biopsy specimens to referral centers. These are generally transferred in liquid containers, under suboptimal safety conditions, as accidental spillage of potentially dangerous substances may occur. A safe, innovative, two-step procedure for pathology sample transportation is presented. Formalin-fixed material from ten surgical cases was dissected (including surrogate biopsies) and preserved in liquid-free plastic bags under vacuum for up to 30 days and subsequently processed for conventional histology, several immunohistochemical markers, and molecular tests (e.g., RAS mutation). The data were compared with the corresponding routine analyses. Formalin-fixed specimens after up to 30 days under vacuum storage gave equivalent results compared to standard histopathological slides and molecular tests, regarding both hematoxylin-eosin, immuno-stained slides and also nucleic acid extracted for molecular tests. The proposal of under-vacuum sealing pathology specimens that were previously formalin fixed can be adopted to transfer liquid-free biopsy and surgical specimens to referral pathology services. In fact, it is easy to perform, less expensive (both plastic bags and domestic-type vacuum chamber machines are at affordable costs), and above all is fully safe and adequate in the pre-analytical processing of pathology specimens.



Similar content being viewed by others
References
Hewitt SM, Lewis FA, Cao Y, Conrad RC, Cronin M, Danenberg KD, Goralski TJ, Langmore JP, Raja RG, Williams PM, Palma JF, Warrington JA (2008) Tissue handling and specimen preparation in surgical pathology: issues concerning the recovery of nucleic acids from formalin-fixed, paraffin-embedded tissue. Arch Pathol Lab Med 132:1929–1935. https://doi.org/10.1043/1543-2165-132.12.1929
Khoury T, Sait S, Hwang H, Chandrasekhar R, Wilding G, Tan D, Kulkarni S (2009) Delay to formalin fixation effect on breast biomarkers. Mod Pathol 22:1457–1467. https://doi.org/10.1038/modpathol.2009.117
Adesina A, Chumba D, Nelson AM, Orem J, Roberts DJ, Wabinga H, Wilson M, Rebbeck TR (2013) Improvement of pathology in sub-Saharan Africa. Lancet Oncol 14:e152–e157. https://doi.org/10.1016/S1470-2045(12)70598-3
Fox CH, Johnson FB, Whiting J, Roller PP (1985) Formaldehyde fixation. J Histochem Cytochem 33:845–853. https://doi.org/10.1177/33.8.3894502
Cheng J, Zhang L, Tang Y, Li Z (2016) The toxicity of continuous long-term low-dose formaldehyde inhalation in mice. Immunopharmacol Immunotoxicol 38:495–501. https://doi.org/10.1080/08923973.2016.1248844
Han RT, Back SK, Lee H, Lee J, Kim HY, Kim HJ, Na HS (2016) Formaldehyde-induced aggravation of pruritus and dermatitis is associated with the elevated expression of Th1 cytokines in a rat model of atopic dermatitis. PLoS One 11:e0168466. https://doi.org/10.1371/journal.pone.0168466
European Parliament and Council of European Union (2008) Regulation (EC) No 1272/2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. Off J Eur Union L 353:1–1355
International Agency for Research and Cancer, IARC (2012) Monographs on the evaluation of carcinogenic risks to humans. Chemical Agents and Related Occupations 100F:401–430 Lyon
Bussolati G, Annaratone L, Berrino E, Miglio U, Panero M, Cupo M, Gugliotta P, Venesio T, Sapino A, Marchio C (2017) Acid-free glyoxal as a substitute of formalin for structural and molecular preservation in tissue samples. PLoS One 12:e0182965. https://doi.org/10.1371/journal.pone.0182965
Bussolati G, Chiusa L, Cimino A, D’Armento G (2008) Tissue transfer to pathology labs: under vacuum is the safe alternative to formalin. Virchows Arch 452:229–231. https://doi.org/10.1007/s00428-007-0529-x
Fischer AH, Jacobson KA, Rose J, Zeller R (2008) Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. pdb.prot4986. doi: https://doi.org/10.1101/pdb.prot4986
Sapino A, Marchiò C, Senetta R, Castellano I, Macrì L, Cassoni P, Ghisolfi G, Cerrato M, D’Ambrosio E, Bussolati G (2006) Routine assessment of prognostic factors in breast cancer using a multicore tissue microarray procedure. Virchows Arch 449:288–296 doi.org/10.1007/s00428-006-0233-2
Marchiò C, Lambros MB, Gugliotta P, Di Cantogno LV, Botta C, Pasini B, Tan DS, Mackay A, Fenwick K, Tamber N, Bussolati G, Ashworth A, Reis-Filho JS, Sapino A (2009) J Pathol 219:16–24. doi: https://doi.org/10.1002/path.2574, Does chromosome 17 centromere copy number predict polysomy in breast cancer? A fluorescence in situ hybridization and microarray-based CGH analysis
van Beers EH, Joosse SA, Ligtenberg MJ, Fles R, Hogervorst FB, Verhoef S, Nederlof PM (2006) A multiplex PCR predictor for a CGH success of FFPE samples. Br J Cancer 94:333–337
Illumina (2014) Expression analysis of FFPE samples. Illumina Technical Note: RNA Sequencing
Mariani S, Bertero L, Osella-Abate S, Di Bello C, Francia di Celle P, Coppola V, Sapino A, Cassoni P, Marchiò C (2017) Extreme assay sensitivity in molecular diagnostics further unveils intratumour heterogeneity in metastatic colorectal cancer as well as artifactual low-frequency mutations in the KRAS gene. Br J Cancer 117:358–366. https://doi.org/10.1038/bjc.2017.170
Berg D, Malinowsky K, Reischauer B, Wolff C, Becker KF (2011) Use of formalin-fixed and paraffin-embedded tissues for diagnosis and therapy in routine clinical settings. Methods Mol Biol 785:109–122. https://doi.org/10.1007/978-1-61779-286-1_8
Denoux Y, Blanc-Vincent MP, Simony-Lafontaine J, Verriele-Beurrier V, Briffod M, Voigt JJ (2002) Standards, options and recommendations: good practice for the management and shipment of histological and cytopathological cancer specimens. Bull Cancer 89:401–409
van der Loos CM (2007) A focus on fixation. Biotech Histochem 82:141–154. https://doi.org/10.1080/10520290701375302
Zarbo RJ (2015) Histologic validation of vacuum sealed, formalin-free tissue preservation, and transport system. Recent Results Cancer Res 199:15–26. https://doi.org/10.1007/978-3-319-13957-9_2
Adeyi OA (2011) Pathology services in developing countries: the West African experience. Arch Pathol Lab Med 135:183–186. https://doi.org/10.1043/2008-0432-CCR.1
Gopal S, Krysiak R, Liomba G (2013) Building a pathology laboratory in Malawi. Lancet Oncol 14:291–292. https://doi.org/10.1016/S1470-2045(13)70109-8
Duong A, Steinmaus C, McHale CM, Vaughan CP, Zhang L (2011) Reproductive and developmental toxicity of formaldehyde: a systematic review. Mutat Res 728:118–138. https://doi.org/10.1016/j.mrrev.2011.07.003
Zhang L, Tang X, Rothman N, Vermeulen R, Ji Z, Shen M, Qiu C, Guo W, Liu S, Reiss B, Freeman LB, Ge Y, Hubbard AE, Hua M, Blair A, Galvan N, Ruan X, Alter BP, Xin KX, Li S, Moore LE, Kim S, Xie Y, Hayes RB, Azuma M, Hauptmann M, Xiong J, Stewart P, Li L, Rappaport SM, Huang H, Fraumeni JF, Smith MT, Lan Q (2010) Occupational exposure to formaldehyde, hematotoxicity, and leukemia-specific chromosome changes in cultured myeloid progenitor cells. Cancer Epidemiol Biomark Prev 19:80–88. https://doi.org/10.1158/1055-9965.EPI-09-0762
Di Novi C, Minniti D, Barbaro S, Zampirolo MG, Cimino A, Bussolati G (2010) Vacuum-based preservation of surgical specimens: an environmentally-safe step towards a formalin-free hospital. Sci Total Environ 408:3092–3095. https://doi.org/10.1016/j.scitotenv.2010.04.022
Author information
Authors and Affiliations
Contributions
All individuals listed as co-authors of this manuscript qualify for every one of the four following criteria:
● Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work
● Drafting the work or revising it critically for important intellectual content
● Final approval of the version to be published
● Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Metovic, J., Bertero, L., Musuraca, C. et al. Safe transportation of formalin-fixed liquid-free pathology specimens. Virchows Arch 473, 105–113 (2018). https://doi.org/10.1007/s00428-018-2383-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-018-2383-4