Abstract
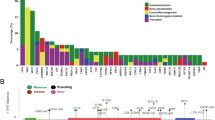
Activating mutations of the epidermal growth factor receptor (EGFR) confers sensitivity to tyrosine kinase inhibitors (TKIs). In colorectal cancer and in lung adenocarcinomas, clinical trials have shown a lack of response to anti-EGFR therapy when KRAS gene mutations are present. In this study, the mutation status of specified exons of the EGFR and KRAS genes was profiled in patients with prostate cancer (PCa). Direct Sanger sequencing was used to screen for mutations in exons 19–21 of EGFR and in exon 2 of KRAS in 88 Chinese patients diagnosed with prostate adenocarcinomas. Mutations were detected in 11 patients. In nine cases (10 %), activating mutations in the region of EGFR encoding the tyrosine kinase (TK) domain were present. Deletions in exon 19 and the L858R substitution in exon 21 were “hotspot” mutations, together accounting for five (55 %) of nine cases. Many synonymous substitutions were also detected. KRAS mutations were found in two cases (2.3 % of 88). There were no cases with mutations in both EGFR and KRAS, suggesting that mutations in the two genes might be mutually exclusive. Although prognostic relevance of EGFR expression by immunohistochemistry (IHC) was observed in PCa patients in previous studies, we found no statistically significant association between EGFR or KRAS mutations and clinicopathological features (including age, smoking status, preoperative prostate-specific antigen, Gleason scores, and tumor stage). We contend that accurate profiling of the mutation status of EGFR and KRAS could improve prognostic stratification, and we suggest a potential anti-EGFR therapy for patients with PCa with EGFR mutations.

Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA cancer J clin 61(2):69–90. doi:10.3322/caac.20107
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA cancer J clin 62(1):10–29. doi:10.3322/caac.20138
Mimeault M, Batra SK (2006) Recent advances on multiple tumorigenic cascades involved in prostatic cancer progression and targeting therapies. Carcinogenesis 27(1):1–22. doi:10.1093/carcin/bgi229
Djakiew D (2000) Dysregulated expression of growth factors and their receptors in the development of prostate cancer. Prostate 42(2):150–160
Shah RB, Ghosh D, Elder JT (2006) Epidermal growth factor receptor (ErbB1) expression in prostate cancer progression: correlation with androgen independence. Prostate 66(13):1437–1444. doi:10.1002/pros.20460
Bonaccorsi L, Carloni V, Muratori M, Formigli L, Zecchi S, Forti G, Baldi E (2004) EGF receptor (EGFR) signaling promoting invasion is disrupted in androgen-sensitive prostate cancer cells by an interaction between EGFR and androgen receptor (AR). Int J cancer J Int du Cancer 112(1):78–86. doi:10.1002/ijc.20362
Di Lorenzo G, Tortora G, D’Armiento FP, De Rosa G, Staibano S, Autorino R, D’Armiento M, De Laurentiis M, De Placido S, Catalano G, Bianco AR, Ciardiello F (2002) Expression of epidermal growth factor receptor correlates with disease relapse and progression to androgen-independence in human prostate cancer. Clin Cancer Res Off J Am Assoc Cancer Res 8(11):3438–3444
Robinson DR, Wu YM, Lin SF (2000) The protein tyrosine kinase family of the human genome. Oncogene 19(49):5548–5557. doi:10.1038/sj.onc.1203957
Hynes NE, Lane HA (2005) ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 5(5):341–354. doi:10.1038/nrc1609
Whang YE, Armstrong AJ, Rathmell WK, Godley PA, Kim WY, Pruthi RS, Wallen EM, Crane JM, Moore DT, Grigson G, Morris K, Watkins CP, George DJ (2013) A phase II study of lapatinib, a dual EGFR and HER-2 tyrosine kinase inhibitor, in patients with castration-resistant prostate cancer. Urol Oncol 31(1):82–86. doi:10.1016/j.urolonc.2010.09.018
Bellezza I, Bracarda S, Caserta C, Minelli A (2006) Targeting of EGFR tyrosine kinase by ZD1839 ("Iressa") in androgen-responsive prostate cancer in vitro. Mol Genet metab 88(2):114–122. doi:10.1016/j.ymgme.2005.12.014
Dhupkar P, Dowling M, Cengel K, Chen B (2010) Effects of anti-EGFR antibody cetuximab on androgen-independent prostate cancer cells. Anticancer Res 30(6):1905–1910
Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat SM, Supko JG, Haluska FG, Louis DN, Christiani DC, Settleman J, Haber DA (2004) Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. New England J Med 350(21):2129–2139. doi:10.1056/NEJMoa040938
Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, Naoki K, Sasaki H, Fujii Y, Eck MJ, Sellers WR, Johnson BE, Meyerson M (2004) EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304(5676):1497–1500. doi:10.1126/science.1099314
Pao W, Miller V, Zakowski M, Doherty J, Politi K, Sarkaria I, Singh B, Heelan R, Rusch V, Fulton L, Mardis E, Kupfer D, Wilson R, Kris M, Varmus H (2004) EGF receptor gene mutations are common in lung cancers from "never smokers" and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc Natl Acad Sci U S A 101(36):13306–13311. doi:10.1073/pnas.0405220101
Sequist LV, Joshi VA, Jänne PA, Bell DW, Fidias P, Lindeman NI, Louis DN, Lee JC, Mark EJ, Longtine J, Verlander P, Kucherlapati R, Meyerson M, Haber DA, Johnson BE, Lynch TJ (2006) Epidermal growth factor receptor mutation testing in the care of lung cancer patients. Clin Cancer Res Off J Am Assoc Cancer Res 12(14 Pt 2):4403s–4408s. doi:10.1158/1078-0432.ccr-06-0099
Uchida A, Hirano S, Kitao H, Ogino A, Rai K, Toyooka S, Takigawa N, Tabata M, Takata M, Kiura K, Tanimoto M (2007) Activation of downstream epidermal growth factor receptor (EGFR) signaling provides gefitinib-resistance in cells carrying EGFR mutation. Cancer Sci 98(3):357–363. doi:10.1111/j.1349-7006.2007.00387.x
Eberhard DA, Johnson BE, Amler LC, Goddard AD, Heldens SL, Herbst RS, Ince WL, Jänne PA, Januario T, Johnson DH, Klein P, Miller VA, Ostland MA, Ramies DA, Sebisanovic D, Stinson JA, Zhang YR, Seshagiri S, Hillan KJ (2005) Mutations in the epidermal growth factor receptor and in KRAS are predictive and prognostic indicators in patients with non-small-cell lung cancer treated with chemotherapy alone and in combination with erlotinib. J Clin Oncol Off J Am Soc Clin Oncol 23(25):5900–5909. doi:10.1200/jco.2005.02.857
Karapetis CS, Khambata-Ford S, Jonker DJ, O’Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD, Robitaille S, Price TJ, Shepherd L, Au HJ, Langer C, Moore MJ, Zalcberg JR (2008) K-ras mutations and benefit from cetuximab in advanced colorectal cancer. New England J Med 359(17):1757–1765. doi:10.1056/NEJMoa0804385
Shigematsu H, Gazdar AF (2006) Somatic mutations of epidermal growth factor receptor signaling pathway in lung cancers. Int J Cancer J Int du Cancer 118(2):257–262. doi:10.1002/ijc.21496
Engstrom PF, Arnoletti JP, Benson AB 3rd, Chen YJ, Choti MA, Cooper HS, Covey A, Dilawari RA, Early DS, Enzinger PC, Fakih MG, Fleshman J Jr, Fuchs C, Grem JL, Kiel K, Knol JA, Leong LA, Lin E, Mulcahy MF, Rao S, Ryan DP, Saltz L, Shibata D, Skibber JM, Sofocleous C, Thomas J, Venook AP, Willett C, National Comprehensive Cancer N (2009) NCCN Clinical Practice Guidelines in Oncology: colon cancer. J National Compr Cancer Netw JNCCN 7(8):778–831
Franiel T, Ludemann L, Taupitz M, Rost J, Asbach P, Beyersdorff D (2009) Pharmacokinetic MRI of the prostate: parameters for differentiating low-grade and high-grade prostate cancer. RoFo Fortschr auf dem Gebiete der Rontgenstrahlen und der Nuklearmedizin 181(6):536–542. doi:10.1055/s-0028-1109168
Tsao MS, Sakurada A, Cutz JC, Zhu CQ, Kamel-Reid S, Squire J, Lorimer I, Zhang T, Liu N, Daneshmand M, Marrano P, da Cunha SG, Lagarde A, Richardson F, Seymour L, Whitehead M, Ding K, Pater J, Shepherd FA (2005) Erlotinib in lung cancer—molecular and clinical predictors of outcome. New England J Med 353(2):133–144. doi:10.1056/NEJMoa050736
Dobashi Y, Suzuki S, Sugawara H, Ooi A (2007) Involvement of epidermal growth factor receptor and downstream molecules in bone and soft tissue tumors. Human Pathol 38(6):914–925. doi:10.1016/j.humpath.2006.12.005
Asahina H, Yamazaki K, Kinoshita I, Yokouchi H, Dosaka-Akita H, Nishimura M (2006) Non-responsiveness to gefitinib in a patient with lung adenocarcinoma having rare EGFR mutations S768I and V769L. Lung Cancer 54(3):419–422. doi:10.1016/j.lungcan.2006.09.005
Peraldo-Neia C, Migliardi G, Mello-Grand M, Montemurro F, Segir R, Pignochino Y, Cavalloni G, Torchio B, Mosso L, Chiorino G, Aglietta M (2011) Epidermal growth factor receptor (EGFR) mutation analysis, gene expression profiling and EGFR protein expression in primary prostate cancer. BMC Cancer 11:31. doi:10.1186/1471-2407-11-31
Schmid K, Oehl N, Wrba F, Pirker R, Pirker C, Filipits M (2009) EGFR/KRAS/BRAF mutations in primary lung adenocarcinomas and corresponding locoregional lymph node metastases. Clin Cancer Res Off J Am Assoc Cancer Res 15(14):4554–4560. doi:10.1158/1078-0432.CCR-09-0089
Nieto Y, Nawaz F, Jones RB, Shpall EJ, Nawaz S (2007) Prognostic significance of overexpression and phosphorylation of epidermal growth factor receptor (EGFR) and the presence of truncated EGFRvIII in locoregionally advanced breast cancer. J Clin Oncol Off J Am Soc Clin Oncol 25(28):4405–4413. doi:10.1200/JCO.2006.09.8822
Itakura Y, Sasano H, Shiga C, Furukawa Y, Shiga K, Mori S, Nagura H (1994) Epidermal growth factor receptor overexpression in esophageal carcinoma. An immunohistochemical study correlated with clinicopathologic findings and DNA amplification. Cancer 74(3):795–804
Li AR, Chitale D, Riely GJ, Pao W, Miller VA, Zakowski MF, Rusch V, Kris MG, Ladanyi M (2008) EGFR mutations in lung adenocarcinomas: clinical testing experience and relationship to EGFR gene copy number and immunohistochemical expression. J Mol Diagn JMD 10(3):242–248. doi:10.2353/jmoldx.2008.070178
Pao W, Kris MG, Iafrate AJ, Ladanyi M, Jänne PA, Wistuba II, Miake-Lye R, Herbst RS, Carbone DP, Johnson BE, Lynch TJ (2009) Integration of molecular profiling into the lung cancer clinic. Clin Cancer Res Off J Am Assoc Cancer Res 15(17):5317–5322. doi:10.1158/1078-0432.ccr-09-0913
Mu XL, Li LY, Zhang XT, Wang MZ, Feng RE, Cui QC, Zhou HS, Guo BQ (2005) Gefitinib-sensitive mutations of the epidermal growth factor receptor tyrosine kinase domain in Chinese patients with non-small cell lung cancer. Clin Cancer Res Off J Am Assoc Cancer Res 11(12):4289–4294. doi:10.1158/1078-0432.CCR-04-2506
Liu Z, Liu L, Li M, Wang Z, Feng L, Zhang Q, Cheng S, Lu S (2011) Epidermal growth factor receptor mutation in gastric cancer. Pathology 43(3):234–238. doi:10.1097/PAT.0b013e328344e61b
Cho NY, Choi M, Kim BH, Cho YM, Moon KC, Kang GH (2006) BRAF and KRAS mutations in prostatic adenocarcinoma. Int J Cancer J Int du Cancer 119(8):1858–1862. doi:10.1002/ijc.22071
Shen Y, Lu Y, Yin X, Zhu G, Zhu J (2010) KRAS and BRAF mutations in prostate carcinomas of Chinese patients. Cancer Genet Cytogenet 198(1):35–39. doi:10.1016/j.cancergencyto.2009.12.003
Shiraishi T, Muneyuki T, Fukutome K, Ito H, Kotake T, Watanabe M, Yatani R (1998) Mutations of ras genes are relatively frequent in Japanese prostate cancers: pointing to genetic differences between populations. Anticancer Res 18(4B):2789–2792
Pergolizzi RG, Kreis W, Rottach C, Susin M, Broome JD (1993) Mutational status of codons 12 and 13 of the N- and K-ras genes in tissue and cell lines derived from primary and metastatic prostate carcinomas. Cancer Investig 11(1):25–32
Ren G, Liu X, Mao X, Zhang Y, Stankiewicz E, Hylands L, Song R, Berney DM, Clark J, Cooper C, Lu YJ (2012) Identification of frequent BRAF copy number gain and alterations of RAF genes in Chinese prostate cancer. Genes Chromosome Cancer 51(11):1014–1023. doi:10.1002/gcc.21984
Agell L, Hernandez S, Salido M, de Muga S, Juanpere N, Arumi-Uria M, Menendez S, Lorenzo M, Lorente JA, Serrano S, Lloreta J (2011) PI3K signaling pathway is activated by PIK3CA mRNA overexpression and copy gain in prostate tumors, but PIK3CA, BRAF, KRAS and AKT1 mutations are infrequent events. Mod Pathol Off J United States Can Acad Pathol Inc 24(3):443–452. doi:10.1038/modpathol.2010.208
Riely GJ, Politi KA, Miller VA, Pao W (2006) Update on epidermal growth factor receptor mutations in non-small cell lung cancer. Clin Cancer Res Off J Am Assoc Cancer Res 12(24):7232–7241. doi:10.1158/1078-0432.CCR-06-0658
Weber DC, Tille JC, Combescure C, Egger JF, Laouiti M, Hammad K, Granger P, Rubbia-Brandt L, Miralbell R (2012) The prognostic value of expression of HIF1alpha, EGFR and VEGF-A, in localized prostate cancer for intermediate- and high-risk patients treated with radiation therapy with or without androgen deprivation therapy. Radiat Oncol 7:66. doi:10.1186/1748-717×-7-66
Acknowledgments
We thank the Chinese Academy of Medical Sciences for support with informatics infrastructure and the Beijing Hospital for providing histology and tissue management support. We are grateful to Jianming Ying and Ling Shan for technical assistance.
Conflict of interest
The authors declare that they have no conflict of interest. All authors have read and approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, M., Zhang, W., Shan, L. et al. Mutation status of somatic EGFR and KRAS genes in Chinese patients with prostate cancer (PCa). Virchows Arch 464, 575–581 (2014). https://doi.org/10.1007/s00428-014-1566-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-014-1566-x