Abstract
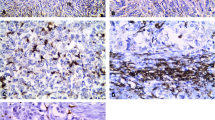
Desmoid-type fibromatosis (DTF) is a rare soft tissue tumor with fibroblastic features affecting two to four individuals per million population per year. Despite its bland microscopic appearance, the tumor behaves aggressively. Although unable to metastasize, DTF tends to recur and local recurrences in anatomically critical sites can be fatal. Tumor-associated macrophages (TAM) play an important role in tumor development through the activation of angiogenesis, particularly in cases of epithelial malignancies. The aim of this study is to investigate the prognostic significance of TAMs and the number of microvessels in DTF. Tumor macrophages (CD163), microvessel density (CD31), and beta-catenin were investigated on 69 primary DTF cases with follow-up information. CTNNB1 mutations were also studied. High density of tumor macrophages and high number of microvessels were associated with a significantly worse recurrence-free survival (P = 0.03 and P = 0.007, respectively). There was a significant correlation between microvessel density and CD163 macrophages (P = 0.02). Furthermore, combination of high number of tumor macrophages and high microvessel density greatly improved the statistical significance (P = 0.000005). Macrophages and microvessels may play an important role in the biologic behavior of DTF. This finding could help in the clinical management of patients with DTF.


Similar content being viewed by others
References
Weiss SW, Goldblum JR (2008) Fibromatoses. In: Weiss SW, Goldblum JR (eds) Ezinger and Weiss’s soft tissue tumors, 5th edn. Mosby, Inc, St Louis, pp 227–256
Condeelis J, Pollard JW (2006) Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124(2):263–266
Lewis CE, Pollard JW (2006) Distinct role of macrophages in different tumor microenvironments. Cancer Res 66(2):605–612
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141(1):39–51
Frank RE, Saclarides TJ, Leurgans S, Speziale NJ, Drab EA, Rubin DB (1995) Tumor angiogenesis as a predictor of recurrence and survival in patients with node-negative colon cancer. Ann Surg 222(6):695–699
Hollingsworth HC, Kohn EC, Steinberg SM, Rothenberg ML, Merino MJ (1995) Tumor angiogenesis in advanced stage ovarian carcinoma. Am J Pathol 147(1):33–41
Olivarez D, Ulbright T, DeRiese W, Foster R, Reister T, Einhorn L, Sledge G (1994) Neovascularization in clinical stage A testicular germ cell tumor: prediction of metastatic disease. Cancer Res 54(10):2800–2802
Weidner N, Carroll PR, Flax J, Blumenfeld W, Folkman J (1993) Tumor angiogenesis correlates with metastasis in invasive prostate carcinoma. Am J Pathol 143(2):401–409
Weidner N, Semple JP, Welch WR, Folkman J (1991) Tumor angiogenesis and metastasis–correlation in invasive breast carcinoma. N Engl J Med 324(1):1–8
Espinosa I, Edris B, Lee CH, Wei Cheng H, Gilks CB, Wang Y, Montgomery KD, Varma S, Li R, Marinelli RJ, West RB, Nielsen T, Beck AH, van de Rijn M (2011) CSF1 expression in nongynecological leiomyosarcoma is associated with increased tumor angiogenesis. Am J Pathol 179(4):2100–7
Lin EY, Li JF, Gnatovskiy L, Deng Y, Zhu L, Grzesik DA, Qian H, Xue XN, Pollard JW (2006) Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res 66(23):11238–11246
Leek RD, Lewis CE, Whitehouse R, Greenall M, Clarke J, Harris AL (1996) Association of macrophage infiltration with angiogenesis and prognosis in invasive breast carcinoma. Cancer Res 56(20):4625–4629
Espinosa I, Jose Carnicer M, Catasus L, Canet B, D'Angelo E, Zannoni GF, Prat J Myometrial invasion and lymph node metastasis in endometrioid carcinomas: tumor-associated macrophages, microvessel density, and HIF1A have a crucial role. Am J Surg Pathol 34 (11):1708-1714
Beck AH, Espinosa I, Edris B, Li R, Montgomery K, Zhu S, Varma S, Marinelli RJ, van de Rijn M, West RB (2009) The macrophage colony-stimulating factor 1 response signature in breast carcinoma. Clin Cancer Res 15(3):778–787
Lee CH, Espinosa I, Vrijaldenhoven S, Subramanian S, Montgomery KD, Zhu S, Marinelli RJ, Peterse JL, Poulin N, Nielsen TO, West RB, Gilks CB, van de Rijn M (2008) Prognostic significance of macrophage infiltration in leiomyosarcomas. Clin Cancer Res 14(5):1423–1430
Pritchard DJ, Nascimento AG, Petersen IA (1996) Local control of extra-abdominal desmoid tumors. J Bone Joint Surg Am 78(6):848–854
Easter DW, Halasz NA (1989) Recent trends in the management of desmoid tumors. Summary of 19 cases and review of the literature. Ann Surg 210(6):765–769
McKinnon JG, Neifeld JP, Kay S, Parker GA, Foster WC, Lawrence W Jr (1989) Management of desmoid tumors. Surg Gynecol Obstet 169(2):104–106
Lazar AJ, Tuvin D, Hajibashi S, Habeeb S, Bolshakov S, Mayordomo-Aranda E, Warneke CL, Lopez-Terrada D, Pollock RE, Lev D (2008) Specific mutations in the beta-catenin gene (CTNNB1) correlate with local recurrence in sporadic desmoid tumors. Am J Pathol 173(5):1518–1527
Kuhnen C, Helwing M, Rabstein S, Homann HH, Muller KM (2005) Desmoid-type fibromatosis (aggressive fibromatosis). Pathologe 26(2):117–126
Kubota Y, Takubo K, Shimizu T, Ohno H, Kishi K, Shibuya M, Saya H, Suda T (2009) M-CSF inhibition selectively targets pathological angiogenesis and lymphangiogenesis. J Exp Med 206(5):1089–1102
Acknowledgments
This work was supported by grant from the Spanish Group for Sarcoma Research (GEIS; ref. GEIS/2008).
Conflicts of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Romero, S., Szafranska, J., Cabrera, E. et al. Role of tumor-associated macrophages and angiogenesis in desmoid-type fibromatosis. Virchows Arch 461, 117–122 (2012). https://doi.org/10.1007/s00428-012-1265-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-012-1265-4