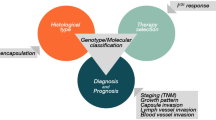
Abstract
Prognostic factors indicative of papillary thyroid carcinoma (PTC) aggressive behaviour remain incompletely established partially due to the different composition of the series on record regarding the relative proportion of classic PTC (CPTC) and follicular variant PTC (FVPTC) subtypes. Several clinico-morphological features of PTC, together with the occurrence of BRAF mutations, are still not fully accepted as markers of aggressiveness. In the present clinico-pathological study of a series of 75 CPTC and FVPTC cases, we evaluated the relative contribution of the morphological features of the tumours and their BRAF and N-RAS status for the occurrence of nodal metastases. The morphological features most closely related to the occurrence of nodal metastases were extra-thyroid extension and poorly circumscribed growth pattern, in both CPTC and FVPTC. Additional features significantly associated to nodal metastases were multicentricity in the CPTC and vascular invasion in the FVPTC group. BRAF V600E mutation was detected in 29% of tumours, 41% of CPTC and 16% of FVPTC; N-RAS Q61R mutation was detected in 6% of tumours, 3% of CPTC and 10% of FVPTC. BRAF mutation was significantly more frequent in the CPTC group and in females, and it was detected only in patients older than 20 years, suggesting a late tumourigenic effect in the development of PTC. BRAF mutation was not significantly associated to any of the other studied features related to aggressiveness or nodal metastases. These results highlight the importance of infiltrative growth pattern and invasiveness over the presence of BRAF mutation in classic and follicular variant PTC for the development of nodal metastases.
Similar content being viewed by others
References
DeLellis RA, Lloyd RV, Heitz PU, Eng C (2004) World Health Organization classification of tumors of endocrine organs. IARC, Lyon
Sobrinho-Simões M, Preto A, Rocha AS, Castro P, Máximo V, Fonseca E, Soares P (2005) Molecular pathology of well-differentiated thyroid carcinomas. Virchows Arch 447:787–793
Rivera M, Ricarte-Filho J, Knauf J, Shaha A, Tuttle RM, Fagin JA, Ghossein RA (2010) Molecular genotyping of papillary thyroid carcinoma follicular variant according to its histologic subtypes (encapsulated vs infiltrative) reveals distinct BRAF and RAS mutation patterns. Mod Pathol 23:1191–1200
Di Cristofaro J, Marcy M, Vasko V, Sebag F, Fakhry N, Wynford-Thomas D, De Micco C (2006) Molecular genetic study comparing follicular variant versus classic papillary thyroid carcinomas: association of N-RAS mutation in codon 61 with follicular variant. Hum Pathol 37:824–830
Liu J, Singh B, Tallini G, Carlson DL, Katabi N, Shaha A, Tuttle RM, Ghossein RA (2006) Follicular variant of papillary thyroid carcinoma: a clinicopathologic study of a problematic entity. Cancer 107:1255–1264
Carcangiu ML, Zampi G, Pupi A, Castagnoli A, Rosai J (1985) Papillary carcinoma of the thyroid. A clinicopathologic study of 241 cases treated at the University of Florence, Italy. Cancer 55:805–828
Gilliland FD, Hunt WC, Morris DM, Key CR (1997) Prognostic factors for thyroid carcinoma. A population-based study of 15,698 cases from surveillance, epidemiology and end results (SEER) program 1973–1991. Cancer 79:564–573
Piana S, Frasoldati A, Di Felice E, Gardini G, Tallini G, Rosai J (2010) Encapsulated well-differentiated follicular-patterned thyroid carcinomas do not play a significant role in the fatality rates from thyroid carcinoma. Am J Surg Pathol 34:868–872
Sobrinho-Simões M, Eloy C, Vinagre J, Soares P (2010) Molecular pathology of thyroid tumors: diagnostic and prognostic relevance. Int J Surg Pathol 18:209S–212S
van Heerden JA, Hay ID, Goellner JR, Salomao D, Ebersold JR, Bergstralh EJ, Grant CS (1992) Follicular thyroid carcinoma with capsular invasion alone: a nonthreatening malignancy. Surgery 112:1130–1136
Tubiana M, Schlumberger M, Rougier P, Laplanche A, Benhamou E, Gardet P, Caillou B, Travagli JP, Parmentier C (1985) Long-term results and prognostic factors in patients with differentiated thyroid carcinoma. Cancer 55:794–804
Elisei R, Ugolini C, Viola D, Lupi C, Biagini A, Giannini R, Romei C, Miccoli P, Pinchera A, Basolo F (2008) BRAF(V600E) mutation and outcome of patients with papillary thyroid carcinoma: a 15-year median follow-up study. J Clin Endocrinol Metab 93:3943–3949
Rosai J, LiVolsi VA, Sobrinho-Simões M, Williams ED (2003) Renaming papillary microcarcinoma of the thyroid gland: the Porto proposal. Int J Surg Pathol 11:249–251
Niemeier LA, Kuffner HA, Carty S, Stewart AF, Nikiforov YE (2010) A combined molecular-pathological score to predict aggressiveness of thyroid papillary microcarcinoma. Mod Pathol. Abst 578, p. 131A
Basolo F, Torregrossa L, Giannini R, Miccoli M, Lupi C, Sensi E, Berti P, Elisei R, Vitti P, Baggiani A, Miccoli P (2010) Correlation between the BRAF V600E mutation and tumor invasiveness in papillary thyroid carcinomas smaller than 20 millimeters: analysis of 1060 cases. J Clin Endocrinol Metab 95:4197–4205
Trovisco V, Soares P, Preto A, de Castro IV, Lima J, Castro P, Máximo V, Botelho T, Moreira S, Meireles AM, Magalhães J, Abrosimov A, Cameselle-Teijeiro J, Sobrinho-Simões M (2005) Type and prevalence of BRAF mutations are closely associated with papillary thyroid carcinoma histotype and patients' age but not with tumour aggressiveness. Virchows Arch 446:589–595
Trovisco V, Couto JP, Cameselle-Teijeiro J, de Castro IV, Fonseca E, Soares P, Sobrinho-Simões M (2008) Acquisition of BRAF gene mutations is not a requirement for nodal metastasis of papillary thyroid carcinoma. Clin Endocrinol (Oxf) 69:683–685
Soares P, Trovisco V, Rocha AS, Lima J, Castro P, Preto A, Máximo V, Botelho T, Seruca R, Sobrinho-Simões M (2003) BRAF mutations and RET/PTC rearrangements are alternative events in the etiopathogenesis of PTC. Oncogene 22:4578–4580
Kim KH, Kang DW, Kim SH, Seong IO, Kang DY (2004) Mutations of the BRAF gene in papillary thyroid carcinoma in a Korean population. Yonsei Med J 45:818–821
Namba H, Nakashima M, Hayashi T, Hayashida N, Maeda S, Rogounovitch TI, Ohtsuru A, Saenko VA, Kanematsu T, Yamashita S (2003) Clinical implication of hot spot BRAF mutation, V599E, in papillary thyroid cancers. J Clin Endocrinol Metab 88:4393–4397
Puxeddu E, Moretti S, Elisei R, Romei C, Pascucci R, Martinelli M, Marino C, Avenia N, Rossi ED, Fadda G, Cavaliere A, Ribacchi R, Falorni A, Pontecorvi A, Pacini F, Pinchera A, Santeusanio F (2004) BRAF(V599E) mutation is the leading genetic event in adult sporadic papillary thyroid carcinomas. J Clin Endocrinol Metab 89:2414–2420
Xu X, Quiros RM, Gattuso P, Ain KB, Prinz RA (2003) High prevalence of BRAF gene mutation in papillary thyroid carcinomas and thyroid tumor cell lines. Cancer Res 63:4561–4567
Nikiforova MN, Kimura ET, Gandhi M, Biddinger PW, Knauf JA, Basolo F, Zhu Z, Giannini R, Salvatore G, Fusco A, Santoro M, Fagin JA, Nikiforov YE (2003) BRAF mutations in thyroid tumors are restricted to papillary carcinomas and anaplastic or poorly differentiated carcinomas arising from papillary carcinomas. J Clin Endocrinol Metab 88:5399–5404
Trovisco V, Soares P, Preto A, Castro P, Máximo V, Sobrinho-Simões M (2007) Molecular genetics of papillary thyroid carcinoma—great expectations… Arq Bras. Endocrinol Metab 51:643–653
Ito Y, Yoshida H, Maruo R, Morita S, Takano T, Hirokawa M, Yabuta T, Fukushima M, Inoue H, Tomoda C, Kihara M, Uruno T, Higashiyama T, Takamura Y, Miya A, Kobayashi K, Matsuzuka F, Miyauchi A (2009) BRAF mutation in papillary thyroid carcinoma in a Japanese population: its lack of correlation with high-risk clinicopathological features and disease-free survival of patients. Endocr J 56:89–97
Castro P, Rebocho AP, Soares RJ, Magalhães J, Roque L, Trovisco V, Vieira de Castro I, Cardoso de Oliveira M, Fonseca E, Soares P, Sobrinho-Simões M (2006) PAX8-PPARg rearrangement is frequently detected in the follicular variant of papillary thyroid carcinoma. J Clin Endocrinol Metab 91:213–220
Zhu Z, Gandhi M, Nikiforova MN, Fischer AH, Nikiforov YE (2003) Molecular profile and clinical-pathologic features of the follicular variant of papillary thyroid carcinoma. An unusual high prevalence of RAS mutations. Am J Clin Pathol 120:71–77
Karga H, Lee JK, Vickery AL Jr, Thor A, Gaz RD, Jameson JL (1991) RAS oncogene mutations in benign and malignant thyroid neoplasms. J Clin Endocrinol Metab 73:832–836
Shi YF, Zou MJ, Schmidt H, Juhasz F, Stensky V, Robb D, Farid NR (1991) High rates of RAS codon 61 mutation in thyroid tumors in an iodine deficient area. Cancer Res 51:2690–2693
Xing M, Clarck D, Guan H, Ji M, Dackiw A, Carson KA, Kim M, Tufaro A, Ladenson P, Zeiger M, Tufano R (2009) BRAF mutation testing of thyroid fine-needle aspiration biopsy specimens for preoperative risk stratification in papillary thyroid cancer. J Clin Oncol 27:2977–2982
Rivera M, Ricarte-Filho J, Tuttle RM, Ganly I, Shaha A, Knauf J, Fagin J, Ghossein R (2010) Molecular, morphologic, and outcome analysis of thyroid carcinomas according to degree of extrathyroid extension. Thyroid 20:1085–1093
Sobrinho-Simões M, Eloy C, Lobo C, Magalhães J, Amaro T (2011) Follicular thyroid carcinoma. Mod Pathol 24:S10–S18
Widder S, Guggisberg K, Khalil M, Pasieka JL (2008) A pathologic re-review of follicular thyroid neoplasms: the impact of changing the threshold for the diagnosis of the follicular variant of papillary thyroid carcinoma. Surgery 144:80–85
Fugazzola L, Pilotti S, Pinchera A, Vorontsova TV, Mondellini P, Bongarzone I, Greco A, Astakahova L, Butti MG, Demidchik EP, Pacini F, Pierotti MA (1995) Oncogenic rearrangements of the RET proto-oncogene in papillary thyroid carcinomas from children exposed to Chernobyl nuclear accident. Cancer Res 55:5617–5620
Nikiforov YE, Rowland JM, Bove KE, Monforte-Munoz H, Fagin JA (1997) Distinct pattern of ret oncogene rearrangements in morphological variants of radiation-induced and sporadic thyroid papillary carcinomas in children. Cancer Res 57:1690–1694
Rhoden KJ, Unger K, Salvatore G, Yilmaz Y, Vovk V, Chiappetta G, Qumsiyeh MB, Rothstein JL, Fusco A, Santoro M, Zitzelsberger H, Tallini G (2006) RET/Papillary thyroid cancer rearrangement in nonneoplastic thyrocytes: follicular cells of Hashimoto's thyroiditis share low-level recombination events with a subset of papillary carcinoma. J Clin Endocrinol Metab 91:2414–23
Xing M, Vasko V, Tallini G, Larin A, Wu G, Udelsman R, Ringel MD, Ladenson PW, Sidransky D (2004) BRAF T1796A transversion mutation in various thyroid neoplasms. J Clin Endocrinol Metab 89:1365–8
Dhomen N, Reis-Filho JS, da Rocha Dias S, Hayward R, Savage K, Delmas V, Larue L, Pritchard C, Marais R (2009) Oncogenic Braf induces melanocyte senescence and melanoma in mice. Cancer Cell 15:294–303
Dankort D, Curley DP, Cartlidge RA, Nelson B, Karnezis AN, Damsky WE Jr, You MJ, DePinho RA, McMahon M, Bosenberg M (2009) Braf(V600E) cooperates with Pten loss to induce metastatic melanoma. Nat Genet 41:544–52
Knauf JA, Sartor MA, Medvedovic M, Lundsmith E, Ryder M, Salzano M, Nikiforov YE, Giordano TJ, Ghossein RA, Fagin JA (2011) Progression of BRAF-induced thyroid cancer is associated with epithelial-mesenchymal transition requiring concomitant MAP kinase and TGFβ signaling. Oncogene. doi:10.1038/onc.2011.44
Franco AT, Malaguarnera R, Refetoff S, Liao XH, Lundsmith E, Kimura S, Pritchard C, Marais R, Davies TF, Weinstein LS, Chen M, Rosen N, Ghossein R, Knauf JA, Fagin JA (2011) Thyrotrophin receptor signaling dependence of BRAF-induced thyroid tumor initiation in mice. Proc Natl Acad Sci USA 108:1615–1620
Sensi M, Nicolini G, Petti C, Bersani I, Lozupone F, Molla A, Vegetti C, Nonaka D, Mortarini R, Parmiani G, Fais S, Anichini A (2006) Mutually exclusive NRASQ61R and BRAFV600E mutations at the single-cell level in the same human melanoma. Oncogene 25:3357–3364
Soares P, Sobrinho-Simões M (2009) Is BRAF mutation screening useful for preoperative risk stratification in papillary thyroid cancer? Future Oncol 5:1225–1229
Kim SK, Hwang TS, Yoo YB, Han HS, Kim DL, Song KH, Lim SD, Kim WS, Paik NS (2011) Surgical results of thyroid nodules according to a management guideline based on the BRAF(V600E) mutation status. J Clin Endocrinol Metab 96:658–664
Tang KT, Lee CH (2010) BRAF mutation in papillary thyroid carcinoma: pathologic role and clinical implications. J Chin Med Assoc 73:113–128
Melck AL, Yip L, Carty SE (2010) The utility of BRAF testing in the management of papillary thyroid cancer. Oncologist 15:1285–1293
Soares P, Sobrinho-Simões M (2011) Small papillary thyroid cancers—is BRAF of prognostic value? Nat Rev Endocrinol 7:9–10
Couto JP, Prazeres H, Castro P, Lima J, Máximo V, Soares P, Sobrinho-Simões M (2009) How molecular pathology is changing and will change the therapeutics of patients with follicular cell-derived thyroid cancer? J Clin Pathol 62:414–421
Gupta-Abramson V, Troxel AB, Nellore A, Puttaswamy K, Redlinger M, Ransone K, Mandel SJ, Flaherty KT, Loevner LA, O'Dwyer PJ, Brose MS (2008) Phase II trial of sorafenib in advanced thyroid cancer. J Clin Oncol 26:4714–4719
Wada N, Duh QY, Sugino K, Iwasaki H, Kameyama K, Mimura T, Ito K, Takami H, Takanashi Y (2003) Lymph node metastasis from 259 papillary thyroid microcarcinomas: frequency, pattern of occurrence and recurrence, and optimal strategy for neck dissection. Ann Surg 237:399–407
Mitsutake N, Knauf JA, Mitsutake S, Mesa C Jr, Zhang L, Fagin JA (2005) Conditional BRAFV600E expression induces DNA synthesis, apoptosis, dedifferentiation, and chromosomal instability in thyroid PCCL3 cells. Cancer Res 65:2465–2473
Castro P, Fonseca E, Magalhaes J, Sobrinho-Simoes M (2002) Follicular, papillary, and "hybrid" carcinomas of the thyroid. Endocr Pathol 13:313–320
Acknowledgements
This work was partially supported by the grant Prof. Edward Limbert SPEND/GENZYME–Year 2007. Part of the data reported in this study were obtained in the project “Identificação de factores prognósticos e de selecção terapêutica em carcinomas diferenciados da tireoide”, which is supported by Fundação Calouste Gulbenkian. IPATIMUP is an Associate Laboratory of the Portuguese Ministry of Science, Technology and Higher Education and is partly supported by the Fundação para a Ciência e Tecnologia.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eloy, C., Santos, J., Soares, P. et al. The preeminence of growth pattern and invasiveness and the limited influence of BRAF and RAS mutations in the occurrence of papillary thyroid carcinoma lymph node metastases. Virchows Arch 459, 265–276 (2011). https://doi.org/10.1007/s00428-011-1133-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-011-1133-7