Abstract
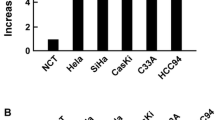
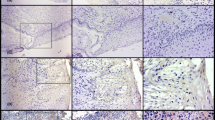
Ubiquitin and telomerase immunohistochemical expression patterns in cervical cancer were compared with normal cervical tissue samples. Eighty-one cervical cancer cases and 22 normal exo–endocervical tissue were examined with polyclonal antibody for ubiquitin and 44G12 clone for telomerase using tissue microarrays. The results were interpreted using a semiquantitative scale The average age of patients was 50.67 years. The most frequent histological types were moderately differentiated epidermoid carcinoma (43.5%), according to the degree of differentiation, and endocervical adenocarcinoma (42.1%). Immunohistochemical findings were as follows: 98.7% of cervical cancers showed immunoexpression for ubiquitin and 52.6% for telomerase. Statistically significant differences were found in tumor immunoreactivity when compared with control tissue (p < 0.0007) for both biomarkers. There was no significant difference in biomarker expression at different histological types of tumors, although telomerase was less expressed in endocervical adenocarcinoma. Our findings confirm that abnormal immunoexpression pattern of ubiquitin and telomerase is common in HPV-positive cervical cancer, indicating the existence of an intense degradation of proteins, subsequent cellular immortalization and maintenance of the malignant phenotype.

Similar content being viewed by others
References
Muñoz N, Bosch FX, de Sanjosé S et al (2003) Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med 348:518–527
Moscicki A-B, Schiffman M, Kjaer S et al (2006) Updating the natural history of HPV and anogenital cancer. Vaccine 24S3:S3/42–S3/51
zur Hausen H (2000) Papillomaviruses causing cancer: evasion from host-cell control in early events in carcinogenesis. J Natl Cancer Inst 92:690–698
von Knebel-Doeberitz M (2001) Aspects of molecular pathogenesis of cervical cancer in establishing new tumor markers for early detection and diagnosis. Zentralbl Gynakol 123:186–191
Scheffner M (1998) Ubicuitin, E6-AP, and their role in p53 inactivation. Pharmacol Ther 78:129–139
Jarboe EA, Liaw K-L, Thompson LC et al (2002) Analysis of telomerase as a diagnostic biomarker of cervical dysplasia and carcinoma. Oncogene 21:664–673
Scheffner M, Whitaker NJ (2003) Human papillomavirus-induced carcinogenesis and the ubicutin-proteosome system. Sem Cancer Biol 13:59–67
Kim YT, Zhao M (2005) Aberrant cell cycle regulation in cervical cancer. Yonsei Medical J 46:597–613
Hanahan D, Weinberg R (2000) The hallmark of cancer. Cell 100:57–70
Hiyama E, Hiyama K (2007) Telomere and telomerase in stem cell. British J Cancer 96:1020–1024
Reesink-Peters N, Helder MN, Wisman GBA et al (2003) Detection of telomerase, its components, and human papillomavirus in cervical scrapings as a tool for triage in women with cervical dysplasia. J Clin Pathol 56:31–35
Braviccini S, Sanchini MA, Amadori A et al (2005) Potencial of telomerase expresión and activity in cervical specimens as a diagnostic tool. J Clin Pathol 58:911–914
Jarboe EA, Thompson LC, Heinz D et al (2004) Telomerase and human papillomavirus as diagnosis adjuncts for cervical dysplasia and carcinoma. Hum Pathol 35:396–402
Pinto-Tang CT, Premoli G (2005) Detection of telomerase activity in cervical lesions by non-radioactive telomeric repeat amplification protocol (TRAP). Invest Clin 46:255–263
Gewin L, Galloway DA (2001) E box-dependent activation of telomerase by human papillomavirus type 16 E6 does not require induction of c-myc. J Virol 75:7198–7201
Oh ST, Kyo S, Laimins LA (2001) Telomerase activation by human papillomavirus type 16 E6 protein: induction of human telomerase reverse transcrptase expression through Myc and GC-rich Sp1 binding sites. J Virol 75:5559–5566
Branca M, Giorgi C, Ciotto M et al (2006) Upregulation of telomerase (hTERT) is related to the grade of cervical intraepithelial neoplasia, but is not an independent predictor of high-risk human papilomavirus, virus persistence, or disease outcome in cervical cancer. Diagn Cytopathol 34:739–748
Takakura M, Saturo K, Taro K et al (1998) Expression of human telomerase subunits and correlation with telomerase activity in cervical cancer. Cancer Res 58:1558–1561
Wang S-Z, Sun J-H, Zhang W et al (2004) Telomerase activity in cervical intraepithelial neoplasia. Chinese Med J 117:202–206
Anderson S, Shera K, Ihle J et al (1997) Telomerase activation in cervical cancer. Am J Pathol 151:25–31
Yashima K, Ashfaq R, Nowak J et al (1998) Telomerase activity and expression of its RNA component in cervical lesions. Cancer 82:1319–1327
Pagano M (1997) Cell cycle regulation by the ubicutin pathway. FASEB J 11:1067–1075
Rolfe M, Chiu I, Pagano M (1997) The ubicutin-mediated proteolytic pathway as a therapeutic area. J Mol Med 75:5–17
Haglund K, Dikic I (2005) Ubicuitylation and cell signaling. EMBO J 24:3353–3359
Huibregtse JM, Scheffner M, Beaudenon S et al (1995) A family of proteins structurally and functionally related to the E6-AP ubiquitin-protein ligase. Proc Natl Acad Sci USA 92:2563–2567
Mitch WE, Goldberg AL (1996) Mechanisms of muscle wasting- the role of the ubiquitin-proteosome pathway. New Engl J Med 335:1897–1905
Boyer SN, Wazer DE, Band V (1996) E7 protein of human papilloma virus-16 induces degradation of retinoblastoma protein through the ubicutin-proteosome pathway. Cancer Res 56:4620–4624
González SL, Stremlau M, He X et al (2001) Degradation of the retinoblastoma tumor suppressor by the human papillomavirus type 16 E7 oncoprotein is important for functional inactivation and is separable from proteosomal degradation of E7. J Virol 75:7583–7591
Berezutskaya E, Bagchi S (1997) The human papillomavirus E7 oncoprotein functionally interacts with the S4 subunit of the 26S proteosome. J Biol Chem 272:30135–30140
Kao WH, Beaudenon SL, Talis AL et al (2000) Human papillomavirus type 16 E6 induces self-ubicuitination of the E6AP ubiquitin-protein ligase. J Virol 74:6408–6417
Hengstermann A, Linares LK, Ciechanover A et al (2001) Complete switch from Mdm2 to human papillomavirus E6-mediated degradation of p53 in cervical cancer cells. Proc Natl Acad Sci USA 98:1218–1223
Shay JW, Bacchetti S (1997) A survey of telomerase activity in human cancer. Eur J Cancer 33:787–791
Yokoyama Y, Takahashi Y, Shinohara A et al (1998) Telomerase activity in the female reproductive tract and neoplasms. Gynecol Oncol 68:145–149
Shroyer KR, Thompson LC, Enomoto T et al (1998) Telomerase expression in normal epithelium, reactive atypia, squamous dysplasia, and squamous cell carcinoma of the uterine cervix. Am J Clin Pathol 109:153–162
Yan P, Benhattar J, Seelentag W et al (2004) Immunohistochemical localization of hTERT protein in human tissues. Histochem Cell Biol 121:391–397
Kyo S, Masutomi K, Maida Y et al (2003) Significance of immunological detection of human telomerase reverse transcriptase. Re-evaluation of expression and localization of human telomerase reverse transcriptase. Am J Pathol 163:859–867
Liu K, Hodes RJ, Weng NP (2001) Cutting edge: telomerase activation in human T lymphocytes does not require increase in telomerase reverse transcriptase (hTERT) protein but is associated with hTERT phosphorylation and nuclear translocation. J Immunol 166:4826–4830
Kakihana M, Yahata N, Hirano T et al (2002) Telomerase activity during carcinogenesis in the bronchus. Oncol Rep 9:43–49
Veldman T, Horikawa I, Barrett JC et al (2001) Transcriptional activation of the telomerase hTERT gene by human papillomavirus type 16 E6 oncoprotein. J Virol 75:4467–4472
Fehrmann F, Laimins LA (2003) Human papillomaviruses: targeting differentiation epithelial cells malignant transformation. Oncogene 22:5201–5207
Veldman T, Liu X, Yuan H et al (2003) Human papillomavirus E6 and Myc proteins associate in vivo and bind to and cooperatively activate the telomerase reverse transcriptase promoter. PNAS 100:8211–8216
Acknowledgments
The authors thank Alejo Sempere and Laura Martínez from the Department of Pathology, School of Medicine, University of Valencia, for their invaluable support and training immunohistochemical technicians.
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This study was carried out with support from the AECC (Junta Provincial) and Fundación Instituto Valenciano de Oncología (FIVO), Valencia, Spain.
Rights and permissions
About this article
Cite this article
de Méndez Morelva, T., Antonio, L.B. Immunohistochemical expression of ubiquitin and telomerase in cervical cancer. Virchows Arch 455, 235–243 (2009). https://doi.org/10.1007/s00428-009-0818-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-009-0818-7