Abstract
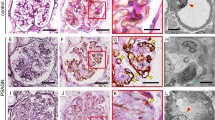
CD34, traditionally a marker of hematopoietic stem cells (HSCs), was found on endothelial cells and fibroblasts as well. At the level of the extraglomerular or intraglomerular mesangium, CD34 may signal either the presence of HSCs or, conversely, may be a marker of transdifferentiation. CD34-positive cells of the extraglomerular mesangium could migrate into the intraglomerular mesangium and participate in reparative processes at this level. The aim of our study was to analyze the presence of CD34 at the level of the extraglomerular and intraglomerular mesangium and its relationship with histological markers of activity and chronicity, as well as with other immunohistochemical markers in glomerulonephritis (GN). A cross-sectional study of 36 patients with GN was conducted. Conventional stains: hematoxylin–eosin, periodic acid Schiff, and Trichrome Gömöri, as well as immunohistochemistry: CD34, alpha smooth muscle actin (alpha SMA), vimentin, and proliferating cell nuclear antigen (PCNA) were employed. Activity and chronicity of GN were evaluated according to a scoring system initially used for lupus nephritis and antineutrophil-cytoplasmic-antibody-associated vasculitis. Immunohistochemistry was assessed using a semiquantitative score. The mean age was 46.44 ± 12.97 years; 22 were male and14 were female. The extraglomerular mesangium was visible on specimens in 30 patients. CD34 was present in the extraglomerular mesangium in 15 patients: 11 of these patients showed concomitant intraglomerular and extraglomerular mesangial CD34 immunostaining, while four showed only extraglomerular mesangial immunostaining. In three patients, CD34 immunostaining was present only in the intraglomerular mesangium. Twelve patients showed negative immunostaining in both the extraglomerular and the intraglomerular mesangium. Overall, there was a fair degree of relationship, which did not reach statistical significance between CD34 in the extraglomerular mesangium and CD34 in the intraglomerular mesangium across the 36 patients. In the intraglomerular mesangium, CD34 did not significantly correlate with mesangial alpha SMA, vimentin, PCNA, and activity or chronicity index. In the extraglomerular mesangium, CD34 did not show a significant correlation with alpha SMA, vimentin, or PCNA. The activity index and the chronicity index showed a good correlation with serum creatinine. Mesangial cell proliferation correlated well with the mesangial matrix increase, while interstitial vimentin showed a good correlation with interstitial alpha SMA. We demonstrated the presence of CD34 in the extraglomerular mesangium, which could be related to transdifferentiated mesangial cells or to HSCs in the absence of blood vessels at this level. Our study shows the value of histological indices for evaluating GN but cannot assign significance to CD34 immunolabeling for the assessment of GN.


Similar content being viewed by others
References
Goligorsky MS, Iijima K, Krivenko Y et al (1997) Role of mesangial cells in macula densa to afferent arteriole information transfer. Clin Exp Pharmacol Physiol 24(7):527–531
Nishio H, Tada J, Hashiyama M et al (1997) MC7. CD34 workshop panel report. In: Kishimoto T, Kitukani H, von dem Borne AEG et al (eds) Leucocyte typing VI. White cell differentiation antigens. Proceedings of the 6th International Workshop and Conference; 1996 Nov 10–14; Kobe, Japan. Garland Publishing Inc, New York, pp 974–976
Krause DS, Fackler MJ, Civin CI et al (1996) CD34: structure, biology and clinical utility (Review). Blood; 87:1–13
Naruse K, Fujieda M, Hayashi Y et al (1999) CD34 expression as a novel marker of transformed mesangial cells in biopsied glomerular diseases. J Pathology 189(1):105–111
Chebotareva NV, Proppe D, Rudolf P et al (2002) Clinical significance of expression of the smooth muscle-actin alpha and CD34 antigen in mesangial cells in glomerulonephritis (Article in Russian). Ter Arkh 74(6):27–31
Natkunam Y, Rouse R, Zhu S et al (2000) Immunoblot analysis of CD34 expression in histologically diverse neoplasms. Am J Pathol 156:21–27
Imasawa T, Utsunomiya Y, Kawamura T et al (2001) The potential of bone marrow-derived cells to differentiate into glomerular mesangial cells. J Am Soc Nephrol 12(7):1401–1409
Nishimura M, Toki J, Sugiura K et al (1994) Focal segmental glomerular sclerosis, a type of intractable chronic glomerulonephritis, is a stem cell disorder? J Exp Med 179(3):1053–1058
Cornacchia F, Fornoni A, Plati AR et al (2001) Glomerulosclerosis is transmitted by bone marrow-derived mesangial cell progenitors. J Clin Invest 108(11):1649–1656
Striker GE (2002) BM-derived progenitors deliver both their genotype and phenotype. Syllabus. ASN Renal Week; PGE Course: Advances in Cell and Molecular Biology: Integration with Renal Medicine. October 30–31, Philadelphia, Pennsylvania
Neumann I, Kain R, Regele H et al (2005) Histological and clinical predictors of early and late renal outcome in ANCA-associated vasculitis. Nephrol Dial Transpl 20(1):96–104
Davison A, Cameron JS, Grunfeld JP et al (eds) (2005) Oxford textbook of clinical nephrology. 3rd edn. Oxford University Press, Oxford, pp 169–182
Dawson B, Trapp RG (2004) Basic and clinical biostatistics. Lange, New York, p 48
Hugo C, Shankland SJ, Bowen-Pope DF et al (1997) Extraglomerular origin of the mesangial cell after injury. J Clin Invest 100(4):786–794
Haseley LA, Hugo C, Reidy MA et al (1999) Dissociation of mesangial cell migration and proliferation in experimental glomerulonephritis. Kidney Int 56:964–972
Poulsom R, Alison MR, Cook T et al (2003) Bone marrow stem cells contribute to healing of the kidney. J Am Soc Nephrol 14(Suppl 1):S48–S54
Lin F, Cordes K, Li L et al (2003) Hematopoietic stem cells contribute to the regeneration of renal tubules after renal ischemia-reperfusion injury in mice. J Am Soc Nephrol 14:1188–1199
Lin F, Igarashi P (2003) Searching for stem/progenitor cells in the adult mouse kidney. J Am Soc Nephrol 14:3290–3292
Schena FP, Abbattista MR (2003) Stem cells: reparative medicine and nephrology (review). J Nephrol 16(Suppl 7):S1–S5
Ricardo SD, Deane JA (2005) Adult stem cells in renal injury and repair (review). Nephrology 10(3):276–282
Ito T (2003) Stem cells of the adult kidney:where are you from? Nephrol Dial Transplant 18(4):641–644
Julian B (2005) IgA Nephropathy. An update on pathogenesis. In: Clinical Nephrology Conferences. ASN Renal Week Philadelphia. Syllabus, pp 477–490
Baddour N, Adam AG, El Koraie AH et al (2002) The reparative role of stem cells in human glomerulonephritis. XXXIX Congress of the European Renal Association European Dialysis and Transplant Association July 14–17, 2002 Copenhagen Denmark. Nephrol Dial Transplant 17(Suppl 1):9–10 Abstracts
Guo J, Schedl A, Krause DS (2004) Bone marrow transplantation attenuates progression of mesangial sclerosis in mouse. In ASN Congress, October 27–November 1, 2004, St. Louis, MO SA FC 098 J Am Soc Nephrol Abstracts Issue
Yokoo T, Ohoshi Y, Utsunomiya Y et al (2003) Gene delivery using human cord blood-derived CD34+ cells into inflamed glomeruli in NOD/SCID mice. Kydney Int 64(1):102–109
Goligorsky MS, Rabelink T (2006) Meeting report: ISN forefronts in nephrology on endothelial biology and renal disease: from bench to prevention (review). Kidney Int 70(2):258–264
Kang DH, Kanellis J, Hugo C et al (2002) Role of the microvascular endothelium in progressive renal disease. J Am Soc Nephrol 13:806–816
Kitamura H, Shimizu A, Masuda Y et al (1998) Apoptosis in glomerular endothelial cells during the development of glomerulosclerosis in a remnant kidney model. Exp Nephrol 6:328–336
Ohashi R, Kitamura H, Yamanaka N (2000) Peritubular capillary injury during the progression of experimental glomerulonephritis in rats. J Am Soc Nephrol 11:47–56
Bottinger EP, Bitzer M (2002) TGF-beta signaling in renal disease. J Am Soc Nephrol 13:2600–2610
Wada Y, Morioka T, Oyanagi-Tanaka Y et al (2002) Impairment of vascular regeneration precedes progressive glomerulosclerosis in anti-Thy 1.1. glomerulonephritis. Kidney Int 61:432–443
Bohle A, Mackensen-Haen S, Wehrmann M (1996) Significance of postglomerular capillaries in the pathogenesis of chronic renal failure. Kidney Blood Press Res, 19(3–4):191–195
Remuzzi A, Gagliardini E, Sangalli F et al (2006) ACE inhibition reduces glomerulosclerosis and regenerates glomerular tissue in a model of progressive renal disease. Kidney Int 69(7):1124–1130 April (1)
Adamczak M, Gross ML, Amann K et al (2004) Reversal of glomerular lesions involves coordinated restructuring of glomerular microvasculature. J Am Soc Nephrol 15:3063–3072
Okon K, Szumera A, Kuzniewski M (2003) Are CD34+ cells found in renal interstitial fibrosis? Am J Nephrol 23(6):409–414
El Kossi MM, El Nahas AM (2003) Stem cell factor and crescentic glomerulonephritis. Am J Kidney Dis 41(4):785–795
El Nahas AM (2003) Plasticity of kidney cells: role in kidney remodeling and scarring. Kidney Int 64(5):1553–1465
Iwano M, Plieth D, Danoff TM et al (2002) Evidence that fibroblasts derive from epithelium during tissue fibrosis. J Clin Invest 110:341–350
Cantley LG (2005) Adult stem cells in the repair of the injured renal tubule (review). Nat Clin Prac Nephrol 1(1):22–32
Acknowledgements
Preliminary data from this study were presented in a poster session at the Congress of the European Society of Nephrology, European Dialysis, and Transplant Association, July, 2006, Glasgow, UK.
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gluhovschi, C., Gluhovschi, G., Potencz, E. et al. What is the significance of CD34 immunostaining in the extraglomerular and intraglomerular mesangium?. Virchows Arch 453, 321–328 (2008). https://doi.org/10.1007/s00428-008-0647-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-008-0647-0