Abstract
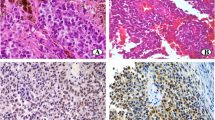
The aim of this study was to investigate the expression of p16 in relation with the histopathologic features and the clinical course in patients with sinonasal melanoma. Thirty-seven sinonasal melanomas were immunostained for p16. Seventeen tumours were investigated for loss of the 9p21 region using interphase fluorescence in situ hybridization (FISH). Twenty-seven melanomas (72.9%) showed loss of p16 expression. All cases with spindle or mixed cytology showed loss of p16, whereas this was present in 50% of epithelioid tumours (p=0.01). Loss of p16 expression was more frequently seen in melanomas with alveolar architecture (87.5%) than in tumours with diffuse architecture (68.9%) (p=0.4). There was no correlation between p16 expression and presence of lymph node or distant metastases (p=0.57 and 0.24, respectively). In addition, p16 status did not influence overall survival (p=0.2). The FISH results were in good agreement with immunohistochemistry: 11 tumours out of 17 showed deletion of the 9p21 region and 10 of these showed loss of protein expression. Loss of p16 expression is a frequent event in sinonasal melanoma and it is mainly related to deletion of 9p21 region. At variance from cutaneous melanoma, loss of p16 is not correlated with the prognosis of patients affected by sinonasal melanoma.


Similar content being viewed by others
References
Batsakis JG, Regezi JA, Solomon AR, Rice DH (1982) The pathology of head and neck tumors: mucosal melanomas. Head Neck Surg 4:404–418
Batsakis JG, Cove H (1994) Melanosis, melanocytic hyperplasia, and primary malignant melanoma of the nasal cavity. Cancer 44:1424–1433
Berthelsen A, Andersen AP, Jensen S, Hansen HA (1984) Melanomas of the mucosa in the oral cavity and the upper respiratory passages. Cancer 54:907–912
Chang AE, Karnell LH, Menck HR (1998) The National Cancer Data Base report on cutaneous and noncutaneous melanoma: a summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer 83:1664–1678
Curtin JA, Fridlyand J, Kageshita T, Patel HN, Busam KJ, Kutzner H, Cho KH, Aiba S, Brocker EB, LeBoit PE, Pinkel D, Bastian BC (2005) Distinct sets of genetic alterations in melanoma. N Engl J Med 353:2135–2147
Franquemont DW, Mills SE (1991) Sinonasal malignant melanoma. A clinicopathologic and immunohistochemical study of 14 cases. Am J Clin Pathol 96:689–697
Freedman HM, DeSanto LW, Devine KD, Weiland LH (1973) Malignant melanoma of the nasal cavity and paranasal sinuses. Arch Otolaryngol 97:322–325
Hofbauer GF, Boni R, Simmen D, Mihic D, Nestle FO, Burg G, Dummer R (2002) Histological, immunological and molecular features of a nasal mucosa primary melanoma associated with nasal melanosis. Melanoma Res 12:77–82
Kamb A, Gruis NA, Weaver-Feldhaus J, Liu Q, Harshman K, Tavtigian SV, Stockert E, Day RS III, Johnson BE, Skolnick MH (1994) A cell cycle regulator potentially involved in genesis of many tumor types. Science 264:436–440
Lund V (1982) Malignant melanoma of the nasal cavity and paranasal sinuses. J Laryngol Otol 96:347–355
Mihic-Probst D, Mnich CD, Oberholzer PA, Seifert B, Sasse B, Moch H, Dummer R (2006). p16 expression in primary malignant melanoma is associated with prognosis and lymph node status. Int J Cancer 118:2262–2268
Padilla Parrado M, Saez Castillo AI, Galan Morales JT, Orradre Romeo JL, Delgado Quero AL, Sanchez Carrion S (2001) Melanoma maligno de fosas nasales. Estudio retrospectivo. Analisis de la expresion de p53 y p16INK4. An Otorrinolaringol Ibero Am 28:303–316
Piccinin S, Doglioni C, Maestro R, Vukosavljevic T, Gasparotto D, D’Orazi C, Boiocchi M (1997) p16/CDKN2 and CDK4 gene mutations in sporadic melanoma development and progression. Int J Cancer 74:26–30
Reed JA, Loganzo F Jr, Shea CR, Walker GJ, Flores JF, Glendening JM, Bogdany JK, Shiel MJ, Haluska FG, Fountain JW et al (1995) Loss of expression of the p16/cyclin-dependent kinase inhibitor 2 tumor suppressor gene in melanocytic lesions correlates with invasive stage of tumor progression. Cancer Res 55:2713–2718
Serrano M, Hannon GJ, Beach D (1993) A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature 366:704–707
Sharpless NE, Chin L (2003) The INK4a/ARF locus and melanoma. Oncogene 22:3092–3098
Sparrow LE, Eldon MJ, English DR, Heenan PJ (1998) p16 and p21WAF1 protein expression in melanocytic tumors by immunohistochemistry. Am J Dermatopathol 20:255–261
Straume O, Sviland L, Akslen LA (2000). Loss of nuclear p16 protein expression correlates with increased tumor cell proliferation (Ki-67) and poor prognosis in patients with vertical growth phase melanoma. Clin Cancer Res 6:1845–1853
Trapp TK, Fu YS, Calcaterra TC (1987) Melanoma of the nasal and paranasal sinus mucosa. Head Neck Surg 113:1086–1089
Thompson LD, Wieneke JA, Miettinen M (2003) Sinonasal tract and nasopharyngeal melanomas: a clinicopathologic study of 115 cases with a proposed staging system. Am J Surg Pathol 27:594–611
van Dijk M, Sprenger S, Rombout P, Marres H, Kaanders J, Jeuken J, Ruiter D (2003) Distinct chromosomal aberrations in sinonasal mucosal melanoma as detected by comparative genomic hybridization. Genes Chromosomes Cancer 36:151–158
Wenig BM, Dulguerov P, Kapadia SB, Prasad ML, Fanburg Smith JC, Thompson LDR (2005) Neuroectodermal tumours. In: Barnes L, Eveson JW, Reichart P, Sidranski D (eds) World Health Organization classification of tumours. Pathology and genetics of head and neck tumours. IARC Press, Lyon, pp 65–75
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Franchi, A., Alos, L., Gale, N. et al. Expression of p16 in sinonasal malignant melanoma. Virchows Arch 449, 667–672 (2006). https://doi.org/10.1007/s00428-006-0288-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-006-0288-0