Abstract
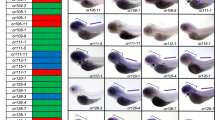
G protein-coupled receptors are critical regulators of diverse developmental processes such as oocyte maturation, fertilization, gastrulation, and organogenesis. To further study the molecular mechanisms underlying these processes, we cloned and characterized the orphan leucine-rich repeat-containing G protein-coupled receptor 6 (LGR6), a stem cell marker in mammalian hair follicles, in medaka fish, Oryzias latipes. To examine the expression pattern of lgr6, we performed whole-mount in situ hybridization (WISH) during embryogenesis. The expression of lgr6 was first detected as a band in the anterior part of the posterior brain vesicle in 0.5–1 day post fertilization (dpf) embryos. This band disappeared by 2 dpf, but new signals appeared in the otic vesicles bordering the original band and also detected in the nasal placode and posterior lateral line primordia. At later stages (3–5 dpf), lgr6 was widely expressed in the brain, otic vesicle, neuromasts, root of the pectoral fin, cranial cartilage, and gut. Then, we conducted more detailed expression analysis of lgr6 in adult gut using WISH and immunohistochemical staining. Lgr6-positive cells were detected in the crypt-like proliferative zone and in parts of the villus. We also performed RT-PCR of mRNAs from different tissues. The lgr6 mRNA was found highest in the kidney and gill. The transcript was also present in the brain, heart, liver, spleen, intestine, skeletal muscle, testis, and ovary, similar to that of mammalian LGR6. These results suggest that medaka lgr6 plays an important role in organ development during embryogenesis and serves as a good molecular marker for future studies of postembryonic organ-specific development in mammals.







Similar content being viewed by others
References
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, Clevers H (2007) Identification of stem cells in small intestine and colon by maker gene Lgr5. Nature 449:1003–1007
Deguchi T, Fujimori KE, Kawasaki T, Ohgushi H, Yuba S (2009) Molecular cloning and gene expression of the prox1a and prox1b genes in the medaka, Oryzias latipes. Gene Expr Patterns 9:341–347
Furutani-Seiki M, Wittbrodt J (2004) Medaka and zebrafish, an evolutionary twin study. Mech Dev 121:629–637
Fraser LR, Adeoya-Osiguwa SA, Baxendale RW (2003) First messenger regulation of capacitation via G protein-coupled receptors. Mol Hum Reprod 9:739–748
Griffin CT, Srinivasan Y, Zheng YW, Huang W, Coughlin SR (2001) A role for thrombin receptor signaling in endothelial cells during embryonic development. Science 293:1666–1670
Haegebarth A, Clevers H (2009) Wnt signaling, lgr5, and stem cells in the intestine and skin. Am J Pathol 174:715–721
Hano T, Oshima Y, Kinoshita M, Tanaka M, Wakamatsu Y, Ozato K, Nassef M, Shimasaki Y, Honjo T (2009) In ovo nanoinjection of nonylphenol affects embryonic development of a transgenic see-through medaka (Oryzias latipes), olvas-GFP/STII-YI strain. Chemosphere 77:1594–1599
Hinton DE, Wakamatsu Y, Ozato K, Kashiwada S (2004) Imaging liver development/remodeling in the see-though medaka fish. Comp Hepatol 14(3 Suppl):S30
Hirose K, Shimoda N, Kikuchi Y (2011) Expression patterns of lgr4 and lgr6 during zebrafish development. Gene Expr Patterns 11:378–383
Hsu SY, Kudo M, Chen T, Nakabayashi K, Bhalla A, van der Spec PJ, van Duin M, Hsueh AJW (2000) The three subfamilies of leucine-rich repeat-containing G protein-coupled receptors (LGR): identification of LGR6 and LGR7 and the signaling mechanism for LGR7. Mol Endocrinol 14:1257–1271
Ishikawa Y, Yasuda T, Kage T, Takashima S, Yoshimoto M, Yamamoto N, Maruyama K, Takeda H, Ito H (2008) Early development of the cerebellum in teleost fishes: a study based on gene expression patterns and histology in the medaka embryo. Zool Sci 25:407–418
Iwamatsu T (2004) Stages of normal development in the medaka Oryzias latipes. Mech Dev 121:605–618
Kasahara M, Naruse K, Sasaki S, Nakatani Y, Qu W, Ahsan B, Yamada T, Nagayasu Y, Doi K, Kasai Y, Jindo T, Kobayashi D, Shimada A, Toyoda A, Kuroki Y, Fujiyama A, Sasaki T, Shimizu A, Asakawa S, Shimizu N, Hashimoto S, Yang J, Lee Y, Matsushima K, Sugano S, Sakaizumi M, Narita T, Ohishi K, Haga S, Ohta F, Nomoto H, Nogata K, Morishita T, Endo T, Shin-I T, Takeda H, Morishita S, Kohara Y (2007) The medaka draft genome and insights into vertebrate genome evolution. Nature 447:714–719
Kashiwada S, Goka K, Shiraishi H, Arizono K, Ozato K, Wakamatsu Y, Hinton DE (2007) Age-dependent in situ hepatic and gill CYP1A activity in the see-through medaka (Oryzias latipes). Comp Biochem Physiol C Toxicol Pharmacol 145:96–102
Kawasaki T, Saito K, Deguchi T, Fujimori K, Tadokoro M, Yuba S, Ohgushi H, Kawarabayasi Y (2008) Pharmacological characterization of isoproterenol-treated medaka fish. Pharmacol Res 58:348–355
Kobayashi D, Jindo T, Naruse K, Takeda H (2006) Development of the endoderm and gut in medaka, Oryzias latipes. Develop Growth Differ 48:283–295.
Kupperman E, An S, Osborne N, Waldron S, Stainier DY (2000) A sphingosine-1-phosphate receptor regulates cell migration during vertebrate heart development. Nature 406:192–195
Leung T, Chen H, Stauffer AM, Giger KE, Sinha S, Horstick EJ, Humbert JE, Hansen CA, Robishaw JD (2006) Zebrafish G protein gamma2 is required for VEGF signaling during angiogenesis. Blood 108:160–166
Leung T, Humbert JE, Stauffer AM, Giger KE, Chen H, Tsai H-J, Wang C, Mirshahi T, Robishaw JD (2008) The orphan G protein-coupled receptor 161 is required for left–right patterning. Dev Biol 323:31–40
Lin F, Sepich DS, Chen S, Topczewski J, Yin C, Solnica-Krezel L, Hamm H (2005) Essential roles of Ga12/13 signaling in distinct cell behaviors driving zebrafish convergence and extension gastrulation movements. J Cell Biol 169:777–787
Loosli F, Koster RW, Carl M, Krone A, Wittbrodt J (1998) Six3, a medaka homologue of the Drosophila homeobox gene sine oculis is expressed in the anterior embryonic shield and the developing eye. Mech Dev 74:159–164
Matteson PG, Desai J, Korstanje R, Lazar G, Borsuk TE, Rollins J, Kadambi S, Joseph J, Rahman T, Wink J, Benayed R, Paigen B, Millonig JH (2008) The orphan G protein-coupled receptor, Gpr161, encodes the vacuolated lens locus and controls neurulation and lens development. Proc Natl Acad Sci USA 105:2088–2093
Offermanns S, Mancino V, Revel JP, Simon MI (1997) Vascular system defects and impaired cell chemokinesis as a result of Ga13 deficiency. Science 275:533–5367
Okubo T, Ishikawa Y (2009) Brain. In: Kinoshita M, Murata K, Naruse K, Tanaka M (eds) Medaka: biology, management, and experimental protocols. Wiley-Blackwell, Iowa, pp 180–187
Romo X, Pasten P, Martinez S, Soto X, Lara P, de Arellano AR, Torrejon M, Montecino M, Hinrichs MV, Olate J (2008) xRic-8 is a GEF for Gsalpha and participates in maintaining meiotic arrest in Xenopus laevis oocytes. J Cell Physiol 214:673–680
Ruppel KM, Willison D, Kataoka H, Wang A, Zheng YW, Cornelissen I, Yin L, Xu SM, Coughlin SR (2005) Essential role for Ga13 in endothelial cells during embryonic development. Proc Natl Acad Sci U S A 102:8281–8286
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters PJ, Clevers H (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459:262–265
Scott IC, Masri B, D’Amico LA, Jin SW, Jungblut B, Wehman AM, Baier H, Audigier Y, Stainier DY (2007) The G protein-coupled receptor agtrl1b regulates early development of myocardial progenitors. Dev Cell 12:403–413
Snippert HJ, Haegebarth A, Kasper M, Jaks V, van Es JH, Barker N, van de Wetering M, van den Born M, Begthel H, Vries RG, Stange DE, Toftgard R, Clevers H (2010) LGR6 marks stem cells in the hair follicle that generate all cell lineages of the skin. Science 327:1385–1389
Shima A, Mitani H (2004) Medaka as a research organism: past, present and future. Mech Dev 121:599–604
Van Loy T, Vandersmissen HP, Van Hiel MB, Poels J, Verlinden H, Badisco L, Vassart G, Vanden Broeck J (2008) Comparative genomics of leucine-rich repeats containing G protein-coupled receptors and their ligands. Gen Comp Endocrinol 155:14–21
Van Hiel MB, Vandersmissen HP, Van Loy T, Vanden Broeck J (2011) An evolutionary comparison of leucine-rich repeat containing G protein-coupled receptors reveals a novel LGR subtype. Peptides. 2011 Nov 10
Wakamatsu Y, Pristyazhnyuk S, Kinoshita M, Tanaka M, Ozato K (2001) The see-through medaka: a fish model that is transparent throughout life. Proc Natl Acad Sci U S A 98:10046–10050
Wallace KN, Pack M (2003) Unique and conserved aspects of gut development in zebrafish. Dev Biol 255:12–29
Yasuoka A, Hirose Y, Yoda H, Aihara Y, Suwa H, Niwa K, Sasado T, Morinaga C, Deguchi T, Henrich T, Iwanami N, Kunimatsu S, Abe K, Kondoh H, Furutani-Seiki M (2004) Mutations affecting the formation of posterior lateral line system in Medaka, Oryzias latipes. Mech Dev 121:729–738
Zeng XX, Wilm TP, Sepich DS, Solnica-Krezel L (2007) Apelin and its receptor control heart field formation during zebrafish gastrulation. Dev Cell 12:391–402
Acknowledgments
We thank Y. Ishikawa for providing Hd-rR and Y. Wakamatsu for providing STII through the National BioResource Project. We also thank Y. Jyosaki for maintaining the fish and sectioning, K. Shizuma and Y. Nogami for help with RT-PCR.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: M. Hammerschmidt
Rights and permissions
About this article
Cite this article
Deguchi, T., Kawasaki, T., Ohnishi, H. et al. Identification and developmental expression of leucine-rich repeat-containing G protein-coupled receptor 6 (lgr6) in the medaka fish, Oryzias latipes . Dev Genes Evol 222, 217–227 (2012). https://doi.org/10.1007/s00427-012-0403-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00427-012-0403-y