Abstract.
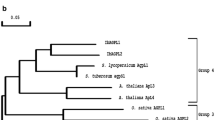
Polymerase chain reaction-based methodology was used to obtain a cDNA clone (MAL2) from potato (Solanum tuberosum L.) with the sequence characteristics of an α-glucosidase. Phylogenetic analysis of the deduced polypeptide encoded by this cDNA demonstrated that the most similar sequences were α-glucosidases and α-xylosidases of plant origin. The MAL2 cDNA was expressed in Escherichia coli and the recombinant MAL2 protein was affinity-purified. MAL2 catalysed the hydrolysis of a range of maltooligomers and p-nitrophenyl-α-D-glucopyranoside with a pH optimum of 5.5–5.7. The substrate with the lowest K m value was maltotetraose (3.7 mM). The MAL2 expression product did not catalyse the hydrolysis of xyloglucan oligosaccharides, p-nitrophenyl-α-D-xylopyranoside or gelatinised potato starch. MAL2 was down-regulated in transgenic potato plants using an antisense approach. In several independent transgenic antisense lines, MAL2 expression was severely down-regulated. Despite this, no decrease in total extractable α-glucosidase and α-xylosidase activity could be detected in tissues from the transgenic plants. In glasshouse trials, no visible phenotype, change in tuber yield or carbohydrate content was associated with MAL2 down-regulation. The implications of these results are discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Taylor, M.A., Ross, H.A., McRae, D. et al. Copy-DNA cloning and characterisation of a potato α-glucosidase: expression in Esherichia coli and effects of down-regulation in transgenic potato. Planta 213, 258–264 (2001). https://doi.org/10.1007/s004250000502
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s004250000502