Abstract
Main conclusion
The operation of 8HGO–ISY fusion enzymes can increase nepetalactol flux to iridoid biosynthesis, and the Gj8HGO–CrISY expression in Gardenia jasminoides indicates that seco-iridoids and closed-ring iridoids share a nepetalactol pool.
Abstract
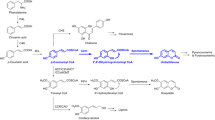
Nepetalactol is a common precursor of (seco)iridoids and their derivatives, which are a group of noncanonical monoterpenes. Functional characterization of an 8HGO (8-hydroxygeraniol oxidoreductase) from Catharanthus roseus, a seco-iridoids producing plant, has been reported; however, the 8HGO from G. jasminoides with plenty of closed-ring iridoids remains uninvestigated. In this work, a Gj8HGO was cloned and biochemically characterized. In addition, the relatively low production of nepetalactol in plants and engineered microbial host is likely to be attributed to the fact that Cr8HGO and CrISY (iridoid synthase) are substrate-promiscuous enzymes catalyzing unexpected substrates to the undesired products. Herein, a bifunctional enzyme consisting of an 8HGO fused to an ISY was designed for the proximity to the substrate and recycling of NADP+ and NADPH cofactor to reduce the undesired intermediate in the synthesis of nepetalactol. Of four fusion enzymes (i.e., Gj8HGO–GjISY, Gj8HGO–GjISY2, Gj8HGO–GjISY4, and Gj8HGO–CrISY), interestingly, only the last one can enable cascade reaction to form cis-trans-nepetalactol. Furthermore, we establish a reliable Agrobacterium-mediated transformation system. The expression of Gj8HGO–CrISY in G. jasminoides led to a significant enhancement of nepetalactol production, about 19-fold higher than that in wild-type plants, which further resulted in the twofold to fivefold increase of total iridoids and representative iridoid such as geniposide, indicating that seco-iridoids in C. roseus and closed-ring iridoids in G. jasminoides share a nepetalactol pool. All results suggest that 8HGO and ISY can be manipulated to maximize metabolic flux for nepetalactol and iridoid production.











Similar content being viewed by others
Data availability
All data included in this study are available upon request by contact with the corresponding author.
References
Allen DK, Libourel IG, Shachar-Hill Y (2009) Metabolic flux analysis in plants: coping with complexity. Plant Cell Environ 32(9):1241–1257. https://doi.org/10.1111/j.1365-3040.2009.01992.x
Awadasseid ALW, Liu Z, Qiao C, Pang J, Zhang G, Luo Y (2020) Characterization of Camptotheca acuminata 10-hydroxygeraniol oxidoreductase and iridoid synthase and their application in biological preparation of nepetalactol in Escherichia coli featuring NADP(+)-NADPH cofactors recycling. Int J Biol MaCromol 162:1076–1085
Billingsley JM, DeNicola AB, Barber JS, Tang MC, Horecka J, Chu A, Garg NK, Tang Y (2017) Engineering the biocatalytic selectivity of iridoid production in Saccharomyces cerevisiae. Metab Eng 44:117–125. https://doi.org/10.1016/j.ymben.2017.09.006
Campbell A, Bauchart P, Gold ND, Zhu Y, De Luca V, Martin VJ (2016) Engineering of a Nepetalactol-Producing Platform Strain of Saccharomyces cerevisiae for the Production of Plant Seco-Iridoids. ACS Synth Biol 5(5):405–414. https://doi.org/10.1021/acssynbio.5b00289
Chen LP, Li MX, Yang ZQ, Tao WD, Wang P, Tian XY, Li XL, Wang WG (2020) Gardenia jasminoides Ellis: Ethnopharmacology, phytochemistry, and pharmacological and industrial applications of an important traditional Chinese medicine. J Ethnopharmacol 257:28. https://doi.org/10.1016/j.jep.2020.112829
Collu G, Unver N, Peltenburg-Looman AM, van der Heijden R, Verpoorte R, Memelink J (2001) Geraniol 10-hydroxylase, a cytochrome P450 enzyme involved in terpenoid indole alkaloid biosynthesis. FEBS Lett 508(2):215–220. https://doi.org/10.1016/s0014-5793(01)03045-9
Davies ME, Tsyplenkov D, Martin VJJ (2021) Engineering Yeast for De Novo Synthesis of the Insect Repellent Nepetalactone. ACS Synth Biol 10(11):2896–2903. https://doi.org/10.1021/acssynbio.1c00420
Dinda B, Debnath S, Harigaya Y (2007a) Naturally occurring iridoids. A review, part 1. Chem Pharm Bull 55(2):159–222. https://doi.org/10.1248/cpb.55.159
Dinda B, Debnath S, Harigaya Y (2007b) Naturally occurring secoiridoids and bioactivity of naturally occurring iridoids and secoiridoids. A review, part 2. Chem Pharm Bull 55(5):689–728. https://doi.org/10.1248/cpb.55.689
Dinda B, Debnath S, Banik R (2011) Naturally Occurring Iridoids and Secoiridoids. An Updated Review, Part 4. Chem Pharm Bull 59(7):803–833. https://doi.org/10.1248/cpb.59.803
Duan Y, Liu J, Du Y, Pei X, Li M (2021) Aspergillus oryzae Biosynthetic Platform for de Novo Iridoid Production. J Agric Food Chem 69(8):2501–2511. https://doi.org/10.1021/acs.jafc.0c06563
Fan XF, Lin L, Cui BX, Zhao TM, Mao LH, Song Y, Wang XY, Feng HJ, Yu QX, Zhang J, Jiang K, Cao XC, Wang BM, Sun C (2020) Therapeutic potential of genipin in various acute liver injury, fulminant hepatitis, NAFLD and other non-cancer liver diseases: More friend than foe. Pharmacol Res 159:9. https://doi.org/10.1016/j.phrs.2020.104945
Fellows R, Russo CM, Silva CS, Lee SG, Jez JM, Chisholm JD, Zubieta C, Nanao MH (2018) A multisubstrate reductase from Plantago major: structure-function in the short chain reductase superfamily. Sci Rep 8(1):14796. https://doi.org/10.1038/s41598-018-32967-1
Galvez M, Martin-Cordero C, Houghton PJ, Ayuso MJ (2005) Antioxidant activity of methanol extracts obtained from Plantago species. J Agric Food Chem 53(6):1927–1933. https://doi.org/10.1021/jf048076s
Geu-Flores F, Sherden NH, Courdavault V, Burlat V, Glenn WS, Wu C, Nims E, Cui Y, O’Connor SE (2012) An alternative route to cyclic terpenes by reductive cyclization in iridoid biosynthesis. Nature 492(7427):138–142. https://doi.org/10.1038/nature11692
Hallahan DL, West JM, Wallsgrove RM, Smiley DW, Dawson GW, Pickett JA, Hamilton JG (1995) Purification and characterization of an acyclic monoterpene primary alcohol:NADP+ oxidoreductase from catmint (Nepeta racemosa). Arch Biochem Biophys 318(1):105–112. https://doi.org/10.1006/abbi.1995.1210
Hernandez Lozada NJ, Hong B, Wood JC, Caputi L, Basquin J, Chuang L, Kunert M, Rodriguez Lopez CE, Langley C, Zhao D, Buell CR, Lichman BR, O’Connor SE (2022) Biocatalytic routes to stereo-divergent iridoids. Nat Commun 13(1):4718. https://doi.org/10.1038/s41467-022-32414-w
Hu YM, Liu WD, Malwal SR, Zheng YY, Feng XX, Ko TP, Chen CC, Xu ZX, Liu MX, Han X, Gao J, Oldfield E, Guo RT (2015) Structures of Iridoid Synthase from Cantharanthus roseus with Bound NAD(+), NADPH, or NAD(+)/10-Oxogeranial: Reaction Mechanisms. Angew Chem-Int Edit 54(51):15478–15482. https://doi.org/10.1002/anie.201508310
Ikeda H, Esaki N, Nakai S, Hashimoto K, Uesato S, Soda K, Fujita T (1991) Acyclic monoterpene primary alcohol:NADP+ oxidoreductase of Rauwolfia serpentina cells: the key enzyme in biosynthesis of monoterpene alcohols. J Biochem 109(2):341–347
Jornvall H, Hoog JO, Persson B (1999) SDR and MDR: completed genome sequences show these protein families to be large, of old origin, and of complex nature. FEBS Lett 445(2–3):261–264. https://doi.org/10.1016/s0014-5793(99)00130-1
Kavanagh KL, Jornvall H, Persson B, Oppermann U (2008) Medium- and short-chain dehydrogenase/reductase gene and protein families : the SDR superfamily: functional and structural diversity within a family of metabolic and regulatory enzymes. Cell Mol Life Sci CMLS 65(24):3895–3906. https://doi.org/10.1007/s00018-008-8588-y
Kim BH, Park KS, Chang IM (2009) Elucidation of Anti-inflammatory Potencies of Eucommia ulmoides Bark and Plantago asiatica Seeds. J Med Food 12(4):764–769. https://doi.org/10.1089/jmf.2008.1239
Kouda R, Yakushiji F (2020) Recent Advances in Iridoid Chemistry: Biosynthesis and Chemical Synthesis. Chem-Asian J 15(22):3771–3783. https://doi.org/10.1002/asia.202001034
Krithika R, Srivastava PL, Rani B, Kolet SP, Chopade M, Soniya M, Thulasiram HV (2015) Characterization of 10-Hydroxygeraniol Dehydrogenase from Catharanthus roseus Reveals Cascaded Enzymatic Activity in Iridoid Biosynthesis. Sci Rep 5:6. https://doi.org/10.1038/srep08258
Lindner S, Geu-Flores F, Brase S, Sherden NH, O’Connor SE (2014) Conversion of Substrate Analogs Suggests a Michael Cyclization in Iridoid Biosynthesis. Chem Biol 21(11):1452–1456. https://doi.org/10.1016/j.chembiol.2014.09.010
Miettinen K, Dong L, Navrot N, Schneider T, Burlat V, Pollier J, Woittiez L, van der Krol S, Lugan R, Ilc T, Verpoorte R, Oksman-Caldentey KM, Martinoia E, Bouwmeester H, Goossens A, Memelink J, Werck-Reichhart D (2014) The seco-iridoid pathway from Catharanthus roseus (vol 5, 3606, 2014). Nat Commun 5:1. https://doi.org/10.1038/ncomms5175
Monterrey DT, Ayuso-Fernandez I, Oroz-Guinea I, Garcia-Junceda E (2022) Design and biocatalytic applications of genetically fused multifunctional enzymes. Biotechnol Adv 60:108016. https://doi.org/10.1016/j.biotechadv.2022.108016
Munkert J, Pollier J, Miettinen K, Van Moerkercke A, Payne R, Muller-Uri F, Burlat V, O’Connor SE, Memelink J, Kreis W, Goossens A (2015) Iridoid Synthase Activity Is Common among the Plant Progesterone 5 beta-Reductase Family. Mol Plant 8(1):136–152. https://doi.org/10.1016/j.molp.2014.11.005
Navale GR, Sharma P, Said MS, Ramkumar S, Dharne MS, Thulasiram HV, Shinde SS (2019) Enhancing epi-cedrol production in Escherichia coli by fusion expression of farnesyl pyrophosphate synthase and epi-cedrol synthase. Eng Life Sci 19(9):606–616. https://doi.org/10.1002/elsc.201900103
Nguyen TD, O’Connor SE (2020) The Progesterone 5beta-Reductase/Iridoid Synthase Family: A Catalytic Reservoir for Specialized Metabolism across Land Plants. ACS Chem Biol 15(7):1780–1787. https://doi.org/10.1021/acschembio.0c00220
Qin L, Zhu Y, Ding Z, Zhang X, Ye S, Zhang R (2016) Structure of iridoid synthase in complex with NADP(+)/8-oxogeranial reveals the structural basis of its substrate specificity. J Struct Biol 194(2):224–230. https://doi.org/10.1016/j.jsb.2016.02.010
Sandholu AS, Mujawar SP, Ramakrishnan K, Thulasiram HV, Kulkarni K (2020) Structural studies on 10-hydroxygeraniol dehydrogenase: A novel linear substrate-specific dehydrogenase from Catharanthus roseus. Proteins 88(9):1197–1206. https://doi.org/10.1002/prot.25891
Schmidt K, Petersen J, Munkert J, Egerer-Sieber C, Hornig M, Muller YA, Kreis W (2018) PRISEs (progesterone 5beta-reductase and/or iridoid synthase-like 1,4-enone reductases): Catalytic and substrate promiscuity allows for realization of multiple pathways in plant metabolism. Phytochemistry 156:9–19. https://doi.org/10.1016/j.phytochem.2018.08.012
Shanmugam MK, Shen HY, Tang FR, Arfuso F, Rajesh M, Wang LZ, Kumar AP, Bian JS, Goh BC, Bishayee A, Sethi G (2018) Potential role of genipin in cancer therapy. Pharmacol Res 133:195–200. https://doi.org/10.1016/j.phrs.2018.05.007
Xia Q, Dong JY, Li L, Wang Q, Liu YX, Wang QM (2018) Discovery of Glycosylated Genipin Derivatives as Novel Antiviral, Insecticidal, and Fungicidal Agents. J Agric Food Chem 66(6):1341–1348. https://doi.org/10.1021/acs.jafc.7b05861
Xu C, Ye P, Wu QW, Liang SC, Wei WK, Yang JF, Chen WW, Zhan RT, Ma DM (2022) Identification and functional characterization of three iridoid synthases in Gardenia jasminoides. Planta 255(3):15. https://doi.org/10.1007/s00425-022-03824-3
Ye P, Liang SC, Wang XM, Duan LX, Jiang FYY, Yang JF, Zhan RT, Ma DM (2019) Transcriptome analysis and targeted metabolic profiling for pathway elucidation and identification of a geraniol synthase involved in iridoid biosynthesis from Gardenia jasminoides. Ind Crop Prod 132:48–58. https://doi.org/10.1016/j.indcrop.2019.02.002
Yee DA, DeNicola AB, Billingsley JM, Creso JG, Subrahmanyam V, Tang Y (2019) Engineered mitochondrial production of monoterpenes in Saccharomyces cerevisiae. Metab Eng 55:76–84. https://doi.org/10.1016/j.ymben.2019.06.004
Acknowledgements
This work was supported by the Guangdong Provincial Rural Revitalization Strategy (agricultural science and technology innovation and extension system construction) special project-Guangdong modern southern medicine industry technology system innovation team (2020KJ148) and Guangdong Pearl River Talents Plan (2017GC010368).
Author information
Authors and Affiliations
Contributions
DM designed the experiments. HL, SL, MZ, and WS performed the experiments. CX and WW analyzed the data. DM and HL wrote the manuscript revised the manuscript. RZ supervised this project.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by De-Yu Xie.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, H., Liang, S., Zhu, M. et al. A fused hybrid enzyme of 8-hydroxygeraniol oxidoreductase (8HGO) from Gardenia jasminoides and iridoid synthase (ISY) from Catharanthus roseus significantly enhances nepetalactol and iridoid production. Planta 259, 62 (2024). https://doi.org/10.1007/s00425-023-04287-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04287-w