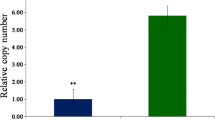
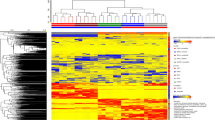
Abstract
Main conclusion
Stemphylium botryosum alters lentil secondary metabolism and differentially affects resistant and susceptible genotypes. Untargeted metabolomics identifies metabolites and their potential biosynthetic pathways that play a crucial role in resistance to S. botryosum.
Abstract
The molecular and metabolic processes that mediate resistance to stemphylium blight caused by Stemphylium botryosum Wallr. in lentil are largely unknown. Identifying metabolites and pathways associated with Stemphylium infection may provide valuable insights and novel targets to breed for enhanced resistance. The metabolic changes following infection of four lentil genotypes by S. botryosum were investigated by comprehensive untargeted metabolic profiling employing reversed-phase or hydrophilic interaction liquid chromatography (HILIC) coupled to a Q-Exactive mass spectrometer. At the pre-flowering stage, plants were inoculated with S. botryosum isolate SB19 spore suspension and leaf samples were collected at 24, 96 and 144 h post-inoculation (hpi). Mock-inoculated plants were used as negative controls. After analyte separation, high-resolution mass spectrometry data was acquired in positive and negative ionization modes. Multivariate modeling revealed significant treatment, genotype and hpi effects on metabolic profile changes that reflect lentil response to Stemphylium infection. In addition, univariate analyses highlighted numerous differentially accumulated metabolites. By contrasting the metabolic profiles of SB19-inoculated and mock-inoculated plants and among lentil genotypes, 840 pathogenesis-related metabolites were detected including seven S. botryosum phytotoxins. These metabolites included amino acids, sugars, fatty acids and flavonoids in primary and secondary metabolism. Metabolic pathway analysis revealed 11 significant pathways including flavonoid and phenylpropanoid biosynthesis, which were affected upon S. botryosum infection. This research contributes to ongoing efforts toward a comprehensive understanding of the regulation and reprogramming of lentil metabolism under biotic stress, which will provide targets for potential applications in breeding for enhanced disease resistance.









Similar content being viewed by others
Data availability
All the data supporting the findings of this study are included in this published article and its supplementary information files. Raw data used are available on request from the corresponding author.
Abbreviations
- ASCA:
-
Analysis of variance simultaneous component analysis
- ChEBI:
-
Chemical entities of biological interest
- ESI:
-
Electrospray ionization
- FooDB:
-
Food database
- HILIC:
-
Hydrophilic interaction liquid chromatography
- HMDB:
-
Human metabolome database
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- NHST:
-
Non-host-specific toxin
- PFP:
-
Pentafluorophenyl
- RH:
-
Relative humidity
- ROS:
-
Reactive oxygen species
References
Agrios GN (2005) Parasitism and disease development. In: Agrios GN (ed) Plant pathology. Elsevier Academic press, San Diego, pp 77–104
Allwood JW, Clarke A, Goodacre R, Mur LAJ (2010) Dual metabolomics: a novel approach to understanding plant-pathogen interactions. Phytochemistry 71:590–597. https://doi.org/10.1016/j.phytochem.2010.01.006
Aly AH, Debbab A, Edrada-Ebel RA et al (2010) Protein kinase inhibitors and other cytotoxic metabolites from the fungal endophyte Stemphylium botryosum isolated from Chenopodium album. Mycosphere 1(2):153–162
Andersen B, Frisvad JC (2004) Natural occurrence of fungi and fungal metabolites in moldy tomatoes. J Agric Food Chem 52:7507–7513. https://doi.org/10.1021/jf048727k
Avni A, Anderson JD, Holland N, Rochaix JD, Gromet-Elhanan Z, Edelman M (1992) Tentoxin sensitivity of chloroplasts determined by codon 83 of β subunit of proton-ATPase. Science 257:1245–1247. https://doi.org/10.1126/science.1387730
Barash I, Karr AL, Strobel GA (1975) Isolation and characterization of Stemphylin, a chromone glucoside from Stemphylium botryosum. Plant Physiol 55:646–651. https://doi.org/10.1104/pp.55.4.646
Barash I, Pupkin G, Netzer D, Kashman Y (1982) A Novel Enolic β-Ketoaldehyde phytotoxin produced by Stemphylium botryosum f. sp. lycopersici. Plant Physiol 69:23–27. https://doi.org/10.1104/pp.69.1.23
Bazghaleh N, Prashar P, Purves RW, Vandenberg A (2018) Polyphenolic composition of lentil roots in response to infection by Aphanomyces euteiches. Front Plant Sci 9:1–12. https://doi.org/10.3389/fpls.2018.01131
Benedict WG (1954) Stemphylium leafspot of alfalfa in Ontario. Plant Dis Report 38:27–29
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Royal Stat Soc Ser B 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Berger S, Sinha AK, Roitsch T (2007) Plant physiology meets phytopathology: plant primary metabolism and plant-pathogen interactions. J Exp Bot 58:4019–4026. https://doi.org/10.1093/jxb/erm298
Bhadauria V, Ramsay L, Bett EK, Banniza S (2017) QTL mapping reveals genetic determinants of fungal disease resistance in the wild lentil species Lens ervoides. Sci Rep 7(1):1–9. https://doi.org/10.1038/s41598-017-03463-9
Bollina V, Kushalappa AC, Choo TM, Dion Y, Rioux S (2011) Identification of metabolites related to mechanisms of resistance in barley against Fusarium graminearum, based on mass spectrometry. Plant Mol Biol 77:355–370. https://doi.org/10.1007/s11103-011-9815-8
Boudet AM (1998) A new view of lignification. Trends Plant Sci 3:67–71. https://doi.org/10.1016/S1360-1385(97)01176-X
Cao Z, Li L, Kapoor K, Banniza S (2019) Using a transcriptome sequencing approach to explore candidate resistance genes against stemphylium blight in the wild lentil species Lens ervoides. BMC Plant Biol 19:1–16. https://doi.org/10.1186/s12870-019-2013-6
Caudillo-Ruiz KB (2016) Characterization of the stemphylium blight pathogens and their effect on lentil yield. Dissertation, University of Saskatchewan
Chapman JM, Muhlemann JK, Gayomba SR, Muday GK (2019) RBOH-dependent ROS synthesis and ROS scavenging by plant specialized metabolites to modulate plant development and stress responses. Chem Res Toxicol 32:370–396. https://doi.org/10.1021/acs.chemrestox.9b00028
Cheynier V, Comte G, Davies KM, Lattanzio V, Martens S (2013) Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol Biochem 72:1–20. https://doi.org/10.1016/j.plaphy.2013.05.009
Chiriac ER, Chiţescu CL, Borda D et al (2020) Comparison of the polyphenolic profile of Medicago sativa L. and Trifolium pratense L. sprouts in different germination stages using the UHPLC-Q exactive hybrid quadrupole orbitrap high-resolution mass spectrometry. Molecules 25:2321. https://doi.org/10.3390/molecules25102321
Couran F, Antignac JP, Dervilly-Pinel G, Le Bizec B (2014) Basics of mass spectrometry based metabolomics. Proteomics 14:2369–2388. https://doi.org/10.1002/pmic.201400255
Csepregi K, Hideg É (2018) Phenolic compound diversity explored in the context of photo-oxidative stress protection. Phytochem Anal 29:129–136. https://doi.org/10.1002/pca.2720
Djoumbou Feunang Y, Eisner R, Knox C et al (2016) ClassyFire: automated chemical classification with a comprehensive, computable taxonomy. J Cheminform 8:1–20. https://doi.org/10.1186/s13321-016-0174-y
Elessawy FM, Vandenberg A, El-Aneed A, Purves RW (2021) An untargeted metabolomics approach for correlating pulse crop seed coat polyphenol profiles with antioxidant capacity and iron chelation ability. Molecules 26:3833. https://doi.org/10.3390/molecules26133833
FAO (2022) Food and Agriculture Organization of the United Nations (FAO). FAOSTAT Database. Rome, Italy Available at: http://www.fao.org/faostat/en/#data/QI
Feussner I, Polle A (2015) What the transcriptome does not tell—proteomics and metabolomics are closer to the plants’ patho-phenotype. Curr Opin Plant Biol 26:26–31. https://doi.org/10.1016/j.pbi.2015.05.023
Fiehn O (2001) Combining genomics, metabolome analysis, and biochemical modelling to understand metabolic networks. Comp Funct Genomics 2:155–168. https://doi.org/10.1002/cfg.82
Fiehn O (2002) Metabolomics—the link between genotypes and phenotypes. Plant Mol Biol 48:155–171. https://doi.org/10.1016/j.mimet.2012.08.011
Fonseca S, Chini A, Hamberg M et al (2009) (+)-7-iso-Jasmonoyl-L-isoleucine is the endogenous bioactive jasmonate. Nat Chem Biol 5:344–350. https://doi.org/10.1038/nchembio.161
Ghosh S, Narula K, Sinha A et al (2016) Proteometabolomic analysis of transgenic tomato overexpressing oxalate decarboxylase uncovers novel proteins potentially involved in defense mechanism against Sclerotinia. J Proteomics 143:242–253. https://doi.org/10.1016/j.jprot.2016.04.047
Gika HG, Wilson ID, Theodoridis GA (2014) LC–MS-based holistic metabolic profiling. Problems, limitations, advantages, and future perspectives. J Chromatogr B 966:1–6. https://doi.org/10.1016/j.jchromb.2014.01.054
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227. https://doi.org/10.1146/annurev.phyto.43.040204.135923
Govrin EM, Levine A (2000) The hypersensitive response facilitates plant infection by the necrotrophic pathogen Botrytis cinerea. Curr Biol 10:751–757. https://doi.org/10.1016/S0960-9822(00)00560-1
Heuberger AL, Robison FM, Lyons SMA, Broeckling CD, Prenni JE (2014) Evaluating plant immunity using mass spectrometry-based metabolomics workflows. Front Plant Sci 5:1–11. https://doi.org/10.3389/fpls.2014.00291
Huang JS (2001) Plant pathogenesis and resistance: Biochemistry and physiology of plant-microbe interactions. Kluwer Academic Publishers, Boston
Khazaei H, Subedi M, Nickerson M, Martínez-Villaluenga C, Frias J, Vandenberg A (2019) Seed protein of lentils: current status, progress, and food applications. Foods 8:391. https://doi.org/10.3390/foods8090391
Klassen A, Faccio AT, Canuto GAB et al (2017) Metabolomics: Definitions and significance in systems biology. In: Sussulini A (ed) Metabolomics: from fundamentals to clinical applications. Springer, Cham, pp 3–17. https://doi.org/10.1007/978-3-319-47656-8_1
Laluk K, Mengiste T (2010) Necrotroph attacks on plants: wanton destruction or covert extortion? Arab B 8:e0136. https://doi.org/10.1199/tab.0136
Lattanzio V, Lattanzio VMT, Cardinali A (2006) Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. In: Imperato F (ed) Phytochemistry: advances in research. Research Signpost, India, pp 23–67
Lee M, Jeon HS, Kim SH et al (2019) Lignin-based barrier restricts pathogens to the infection site and confers resistance in plants. EMBO J 38:1–17. https://doi.org/10.15252/embj.2019101948
Liang X, Rollins JA (2018) Mechanisms of broad host range necrotrophic pathogenesis in Sclerotinia sclerotiorum. Phytopathology 108:1128–1140. https://doi.org/10.1094/PHYTO-06-18-0197-RVW
Manulis S, Kashman Y, Netzert D, Barash I (1984) Phytotoxins from Stemphylium botryosum: structural determination of stemphyloxin II, production in culture and interaction with iron. Phytochemistry 23:2193–2198. https://doi.org/10.1016/S0031-9422(00)80518-X
Nueda MJ, Conesa A, Westerhuis JA et al (2007) Discovering gene expression patterns in time course microarray experiments by ANOVA-SCA. Bioinformatics 23:1792–1800. https://doi.org/10.1093/bioinformatics/btm251
Oliveros JC (2015) Venny. An interactive tool for comparing lists with Venn’s diagrams. Available at: https://bioinfogp.cnb.csic.es/tools/venny/index.html. Accessed 31 Mar 2022
Olsen KJK, Rossman A, Andersen B (2018) Metabolite production by species of Stemphylium. Fungal Biol 122:172–181. https://doi.org/10.1016/j.funbio.2017.12.012
Pang Z, Zhou G, Ewald J et al (2022) Using MetaboAnalyst 5.0 for LC–HRMS spectra processing, multi-omics integration and covariate adjustment of global metabolomics data. Nat Protoc 6:743–760. https://doi.org/10.1038/s41596-022-00710-w
Park SW, Li W, Viehhauser A et al (2013) Cyclophilin 20–3 relays a 12-oxo-phytodienoic acid signal during stress responsive regulation of cellular redox homeostasis. Proc Natl Acad Sci USA 110:9559–9564. https://doi.org/10.1073/pnas.1218872110
Parker D, Beckmann M, Zubair H et al (2009) Metabolomic analysis reveals a common pattern of metabolic re-programming during invasion of three host plant species by Magnaporthe grisea. Plant J 59:723–737. https://doi.org/10.1111/j.1365-313X.2009.03912.x
Pedras MSC, Chumala PB, Jin W, Islam MS, Hauck DW (2009) The phytopathogenic fungus Alternaria brassicicola: Phytotoxin production and phytoalexin elicitation. Phytochemistry 70:394–402. https://doi.org/10.1016/j.phytochem.2009.01.005
Petrussa E, Braidot E, Zancani M et al (2013) Plant flavonoids-biosynthesis, transport and involvement in stress responses. Int J Mol Sci 14:14950–14973. https://doi.org/10.3390/ijms140714950
Pharr DM, Stoop JMH, Williamson JD, Feusi MES, Massel MO, Conkling MA (1995) The dual role of mannitol as osmoprotectant and photoassimilate in celery. HortScience 30:1182–1188. https://doi.org/10.21273/HORTSCI.30.6.1182
Picart-Armada S, Fernández-Albert F, Vinaixa M, Yanes O, Perera-Lluna A (2018) FELLA: an R package to enrich metabolomics data. BMC Bioinformat 19:1–9. https://doi.org/10.1186/s12859-018-2487-5
Podder R, Banniza S, Vandenberg A (2013) Screening of wild and cultivated lentil germplasm for resistance to stemphylium blight. Plant Genet Resour 11:26–35. https://doi.org/10.1017/S1479262112000329
Rotem J, Cohen Y, Wahl I (1966) A new tomato foliage disease in Israel caused by Stemphylium botryosum. Can J Plant Sci 46:265–270
Ruttkies C, Schymanski EL, Wolf S, Hollender J, Neumann S (2016) MetFrag relaunched: Incorporating strategies beyond in silico fragmentation. J Cheminform 8:1–16. https://doi.org/10.1186/s13321-016-0115-9
Sardans J, Peñuelas J, Rivas-Ubach A (2011) Ecological metabolomics: overview of current developments and future challenges. Chemoecology 21:191–225. https://doi.org/10.1007/s00049-011-0083-5
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–26. https://doi.org/10.1155/2012/217037
Smilde AK, Jansen JJ, Hoefsloot HCJ, Lamers RJAN, van der Greef J, Timmerman ME (2005) ANOVA-simultaneous component analysis (ASCA): A new tool for analyzing designed metabolomics data. Bioinformatics 21:3043–3048. https://doi.org/10.1093/bioinformatics/bti476
Solfrizzo M, Strange RN, Sabia C, Visconti A (1994) Production of a toxin stemphol by Stemphylium species. Nat Toxins 2:14–18. https://doi.org/10.1002/nt.2620020104
Solomon PS, Waters ODC, Oliver RP (2007) Decoding the mannitol enigma in filamentous fungi. Trends Microbiol 15:257–262. https://doi.org/10.1016/j.tim.2007.04.002
Stoop J, Williamson J, Mason Pharr D (1996) Mannitol metabolism in plants: a method for coping with stress. Trends Plant Sci 1:139–144. https://doi.org/10.1016/S1360-1385(96)80048-3
Subedi M, Bazghaleh N, Caudillo-Ruiz K, Vandenberg A (2021) Stemphylium blight reduces milling qualities of lentil. Agron J 113:1909–1919. https://doi.org/10.1002/agj2.20551
Sumner LW, Amberg A, Barrett D et al (2007) Proposed minimum reporting standards for chemical analysis. Metabolomics 3:211–221. https://doi.org/10.1007/s11306-007-0082-2
Taylor P, Lindbeck K, Chen W, Ford R (2007) Lentil diseases. In: Yadav SS, McNeil DL, Stevenson PC (eds) Lentil: An ancient crop for modern times. Springer, Dordrecht, pp 291–313. https://doi.org/10.1007/978-1-4020-6313-8
Tugizimana F, Piater L, Dubery I (2013) Plant metabolomics: a new frontier in phytochemical analysis. S Afr J Sci 109:18–20. https://doi.org/10.1590/sajs.2013/20120005
Tullu A, Bett K, Banniza S, Vail S, Vandenberg A (2013) Widening the genetic base of cultivated lentil through hybridization of Lens culinaris “Eston” and L. ervoides accession IG 72815. Can J Plant Sci 93:1037–1047. https://doi.org/10.4141/CJPS2013-072
van Kan JAL (2006) Licensed to kill: the lifestyle of a necrotrophic plant pathogen. Trends Plant Sci 11:247–253. https://doi.org/10.1016/j.tplants.2006.03.005
Vis DJ, Westerhuis JA, Smilde AK, van der Greef J (2007) Statistical validation of megavariate effects in ASCA. BMC Bioinformat 8:1–8. https://doi.org/10.1186/1471-2105-8-322
Vogt T (2010) Phenylpropanoid biosynthesis. Mol Plant 3:2–20. https://doi.org/10.1093/mp/ssp106
Walton JD (1996) Host-selective toxins: agents of compatibility. Plant Cell 8:1723–1733. https://doi.org/10.1105/tpc.8.10.1723
Wang X, Jiang N, Liu J, Liu W, Wang GL (2014) The role of effectors and host immunity in plant–necrotrophic fungal interactions. Virulence 5:722–732. https://doi.org/10.4161/viru.29798
Wasternack C, Song S (2017) Jasmonates: Biosynthesis, metabolism, and signaling by proteins activating and repressing transcription. J Exp Bot 68:1303–1321. https://doi.org/10.1093/jxb/erw443
Williams B, Kabbage M, Kim HJ, Britt R, Dickman MB (2011) Tipping the balance: Sclerotinia sclerotiorum secreted oxalic acid suppresses host defenses by manipulating the host redox environment. PLoS Pathog 7:1002107. https://doi.org/10.1371/journal.ppat.1002107
Wolf S, Schmidt S, Müller-Hannemann M, Neumann S (2010) In silico fragmentation for computer assisted identification of metabolite mass spectra. BMC Bioinformatics 11:148. https://doi.org/10.1186/1471-2105-11-148
Xia J, Wishart DS (2011) Web-based inference of biological patterns, functions and pathways from metabolomic data using MetaboAnalyst. Nat Protoc 6:743–760. https://doi.org/10.1038/nprot.2011.319
Xia J, Wishart DS, Valencia A (2011) MetPA: A web-based metabolomics tool for pathway analysis and visualization. Bioinformatics 27:2342–2344. https://doi.org/10.1093/bioinformatics/btq418
Yan J, Li S, Gu M et al (2016) Endogenous bioactive jasmonate is composed of a set of (+)-7-iso-JA-amino acid conjugates. Plant Physiol 172:2154–2164. https://doi.org/10.1104/pp.16.00906
Zeilinger S, Gupta V, Dahm TES et al (2016) Friends or foes? Emerging insights from fungal interactions with plants. FEMS Microbiol Rev 40:182–207. https://doi.org/10.1093/femsre/fuv045
Acknowledgements
The authors thank Fatma Elessawy for her assistance during sample preparation and valuable suggestions. The authors also thank Bryn Shurmer and Stephan Briere for their internal review of this manuscript. This project was funded from multiple sources including the Agricultural Development Fund of the Saskatchewan Ministry of Agriculture, Saskatchewan Pulse Growers and the Natural Sciences and Engineering Research Council of Canada Industrial Research Chair Program.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Communicated by Anastasios Melis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Adobor, S., Banniza, S., Vandenberg, A. et al. Untargeted profiling of secondary metabolites and phytotoxins associated with stemphylium blight of lentil. Planta 257, 73 (2023). https://doi.org/10.1007/s00425-023-04105-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04105-3