Abstract
Main conclusion
Based on the phenotypic, physiological and transcriptomic analysis, receptor-like kinase HAESA-like 1 was demonstrated to positively affect seed longevity in Arabidopsis.
Abstract
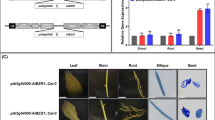
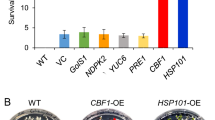
Seed longevity is very important for both genetic resource conservation and crop production. Receptor-like kinases (RLKs) are widely involved in plant growth, development and stress responses. However, the role of most RLKs, especially in seed longevity, is largely unknown. In this study, we report that Arabidopsis HAESA-like 1 (AtHSL1) positively regulated seed longevity. Disruption of HSL1 significantly decreased the germination rate to 50% at 7 days after cold stratification (DAC), compared with that of the wild type (93.5% at 7 DAC), after accelerated aging treatment. Expression of the HSL1 gene in hsl1 basically restored the defective phenotype (86.3%), while HSL1-overexpressing lines (98.3%) displayed slower accelerated aging than WT (93.5%). GUS staining revealed HSL1 was highly expressed universally, especially in young seedlings, mature seeds and embryos of imbibed seeds, and its expression could be induced by accelerated aging. No difference in the dyeing color and area of mucilage were identified between WT and hsl1. The soluble pectin content also was not different, while the adherent pectin content was significantly increased in hsl1. Global transcriptomics revealed that disruption of HSL1 mainly downregulated genes involved in trehalose synthesis, nucleotide sugar metabolism and protection and repair mechanisms. Therefore, an increase in adherent pectin content and downregulation of genes involved in trehalose synthesis may be the main reasons for decreasing seed longevity owing to disruption of HSL1 in Arabidopsis. Our work provides valuable information for understanding the function and mechanism of a receptor-like kinase, AtHSL1, in seed longevity.






Similar content being viewed by others
Data availability
The RNA-seq raw data were stored on the NCBI Read Archive database under the accession number PRJNA793755, and will be made publicly accessible after the publication of the manuscript. Other datasets and plant materials generated during the study are available from the corresponding author upon request.
Abbreviations
- DAC:
-
Days after cold stratification
- DEGs:
-
Differentially expressed genes
- GGLT:
-
Golgi nucleotide sugar transporter 3
- HAE:
-
HAESA
- HSL1:
-
HAESA-like 1
- LRR:
-
Leucine-rich repeat
- RHM:
-
Rhamnose synthase
- RLKs:
-
Receptor like kinases
- SSPs:
-
Seed storage proteins
References
Arsovski AA, Haughn GW, Western TL (2010) Seed coat mucilage cells of Arabidopsis thaliana as a model for plant cell wall research. Plant Signal Behav 5(7):796–801. https://doi.org/10.4161/psb.5.7.11773
Bailly C, El-Maarouf-Bouteau H, Corbineau F (2008) From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. C R Biol 331(10):806–814. https://doi.org/10.1016/j.crvi.2008.07.022
Benaroudj N, Lee DH, Goldberg AL (2001) Trehalose accumulation during cellular stress protects cells and cellular proteins from damage by oxygen radicals. J Biol Chem 276(26):24261–24267. https://doi.org/10.1074/jbc.M101487200
Blumenkrantz N, Asboe-Hansen G (1973) New method for quantitative determination of uronic acids. Anal Biochem 54:484–489
Bueso E, Muñoz-Bertomeu J, Campos F, Brunaud V, Martínez L, Sayas E, Ballester P, Yenush L, Serrano R (2014) Arabidopsis Thaliana Homeobox25 uncovers a role for gibberellins in seed longevity. Plant Physiol 164(2):999–1010. https://doi.org/10.1104/pp.113.232223
Buitink J, Leprince O (2008) Postgenomic analysis of desiccation tolerance. J Soc Biol 202(3):213–222. https://doi.org/10.1051/jbio:2008027
Chen D, Li Y, Fang T, Shi X, Chen X (2016) Specific roles of tocopherols and tocotrienols in seed longevity and germination tolerance to abiotic stress in transgenic rice. Plant Sci 244:31–39. https://doi.org/10.1016/j.plantsci.2015.12.005
Cho SK, Larue CT, Chevalier D, Wang H, Jinn TL, Zhang S, Walker JC (2008) Regulation of floral organ abscission in Arabidopsis thaliana. Proc Natl Acad Sci USA 105(40):15629–15634. https://doi.org/10.1073/pnas.0805539105
Debeaujon I, Léon-Kloosterziel KM, Koornneef M (2000) Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis. Plant Physiol 122(2):403–414. https://doi.org/10.1104/pp.122.2.403
Dekkers BJW, Willems L, Bassel GW, Bolderen-Veldkamp RP, Ligterink W, Hilhorst HWM, Bentsink L (2012) Identification of reference genes for RT–qPCR expression analysis in Arabidopsis and tomato seeds. Plant Cell Physiol. https://doi.org/10.1093/pcp/pcr113
Huang J, Wijeratne AJ, Tang C, Zhang T, Fenelon RE, Owen HA, Zhao D (2016) Ectopic expression of TAPETUM DETERMINANT1 affects ovule development in Arabidopsis. J Exp Bot 67(5):1311–1326. https://doi.org/10.1093/jxb/erv523
Iturriaga G, Cushman MAF, Cushman JC (2006) An EST catalogue from the resurrection plant Selaginella lepidophylla reveals abiotic stress-adaptive genes. Plant Sci 170(6):1173–1184. https://doi.org/10.1016/j.plantsci.2006.02.004
Jinn T-L, Stone JM, Walker JC (2000) HAESA, an Arabidopsis leucine-rich repeat receptor kinase, controls floral organ abscission. Genes Dev 14(1):108–117. https://doi.org/10.1101/gad.14.1.108
Kumpf RP, Shi CL, Larrieu A, Sto IM, Butenko MA, Peret B, Riiser ES, Bennett MJ, Aalen RB (2013) Floral organ abscission peptide IDA and its HAE/HSL2 receptors control cell separation during lateral root emergence. Proc Natl Acad Sci USA 110(13):5235–5240. https://doi.org/10.1073/pnas.1210835110
Li J, Wen J, Lease KA, Doke JT, Tax FE, Walker JC (2002) BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110(2):213–222. https://doi.org/10.1016/S0092-8674(02)00812-7
Liu XS, Liang CC, Hou SG, Wang X, Chen DH, Shen JL, Zhang W, Wang M (2020) The LRR-RLK protein HSL3 regulates stomatal closure and the drought stress response by modulating hydrogen peroxide homeostasis. Front Plant Sci 11:1789. https://doi.org/10.3389/fpls.2020.548034
Lunn JE, Delorge I, Figueroa CM, Van Dijck P, Stitt M (2014) Trehalose metabolism in plants. Plant J 79(4):544–567. https://doi.org/10.1111/tpj.12509
Mendu V, Griffiths JS, Persson S, Stork J, Downie AB, Voiniciuc C, Haughn GW, DeBolt S (2011) Subfunctionalization of cellulose synthases in seed coat epidermal cells mediates secondary radial wall synthesis and mucilage attachment. Plant Physiol 157(1):441–453. https://doi.org/10.1104/pp.111.179069
Meng XZ, Zhou JG, Tang J, Li B, de Oliveira Marcos VV, Chai J, He P, Shan L (2016) Ligand-induced receptor-like kinase complex regulates floral organ abscission in Arabidopsis. Cell Rep 14(6):1330–1338. https://doi.org/10.1016/j.celrep.2016.01.023
Mohnen D (2008) Pectin structure and biosynthesis. Curr Opin Plant Biol 11(3):266–277. https://doi.org/10.1016/j.pbi.2008.03.006
Niederhuth CE, Patharkar OR, Walker JC (2016) Transcriptional profiling of the Arabidopsis abscission mutant hae hsl2 by RNA-Seq. BMC Genom 14(1):37–37. https://doi.org/10.1186/1471-2164-14-37
Oka T, Nemoto T, Jigami Y (2007) Functional analysis of Arabidopsis thaliana RHM2/MUM4, a multidomain protein involved in UDP-D-glucose to UDP-L-rhamnose conversion. J Biol Chem 282(8):5389–5403. https://doi.org/10.1074/jbc.M610196200
Patharkar OR, Walker JC (2015) Floral organ abscission is regulated by a positive feedback loop. Proc Natl Acad Sci USA 112(9):2906–2911. https://doi.org/10.1073/pnas.1423595112
Patharkar OR, Walker JC (2016) Core mechanisms regulating developmentally timed and environmentally triggered abscission. Plant Physiol 172(1):510–520. https://doi.org/10.1104/pp.16.01004
Pellizzaro A, Neveu M, Lalanne D, Ly VuB, Kanno Y, Seo M, Leprince O, Buitink J (2020) A role for auxin signaling in the acquisition of longevity during seed maturation. New Phytol 225(1):284–296. https://doi.org/10.1111/nph.16150
Pitorre D, Llauro C, Jobet E, Guilleminot J, Brizard J-P, Delseny M, Lasserre E (2010) RLK7, a leucine-rich repeat receptor-like kinase, is required for proper germination speed and tolerance to oxidative stress in Arabidopsis thaliana. Planta 232(6):1339–1353. https://doi.org/10.1007/s00425-010-1260-4
Prieto-Dapena P, Rl C, Cn A, Jordano J (2006) Improved resistance to controlled deterioration in transgenic seeds. Plant Physiol 142(3):1102–1112. https://doi.org/10.1104/pp.106.087817
Probert R, Adams J, Coneybeer J, Crawford A, Hay F (2007) Seed quality for conservation is critically affected by pre-storage factors. Aust J Bot. https://doi.org/10.1071/BT06046
Qian P, Song W, Yokoo T, Minobe A, Wang G, Ishida T, Sawa S, Chai J, Kakimoto T (2018) The CLE9/10 secretory peptide regulates stomatal and vascular development through distinct receptors. Nat Plants 4(12):1071–1081. https://doi.org/10.1038/s41477-018-0317-4
Rautengarten C, Ebert B, Moreno I, Temple H, Herter T, Link B, Donas-Cofre D, Moreno A, Saez-Aguayo S, Blanco F, Mortimer JC, Schultink A, Reiter WD, Dupree P, Pauly M, Heazlewood JL, Scheller HV, Orellana A (2014) The Golgi localized bifunctional UDP-rhamnose/UDP-galactose transporter family of Arabidopsis. Proc Natl Acad Sci USA 111(31):11563–11568. https://doi.org/10.1073/pnas.1406073111
Renard J, Martinez-Almonacid I, Sonntag A, Molina I, Moya-Cuevas J, Bissoli G, Munoz-Bertomeu J, Faus I, Ninoles R, Shigeto J, Tsutsumi Y, Gadea J, Serrano R, Bueso E (2020) PRX2 and PRX25, peroxidases regulated by COG1, are involved in seed longevity in Arabidopsis. Plant Cell Environ 43(2):315–326. https://doi.org/10.1111/pce.13656
Sattler SE, Gilliland LU, Magallanes-Lundback M, Pollard M, DellaPenna D (2004) Vitamin E is essential for seed longevity and for preventing lipid peroxidation during germination. Plant Cell 16(6):1419–1432. https://doi.org/10.1105/tpc.021360
Schröder M, Kaufman RJ (2005) ER stress and the unfolded protein response. Mutat Res-Fund Mol M 569(1):29–63. https://doi.org/10.1016/j.mrfmmm.2004.06.056
Shi D, Wang J, Hu R, Zhou G, O’Neill MA, Kong Y (2017) Boron-bridged RG-II and calcium are required to maintain the pectin network of the Arabidopsis seed mucilage ultrastructure. Plant Mol Biol 94(3):267–280. https://doi.org/10.1007/s11103-017-0606-8
Shiu SH, Bleecker AB (2001) Receptor-like kinases from Arabidopsis form a monophyletic gene family related to animal receptor kinases. Proc Natl Acad Sci USA 98(19):10763–10768. https://doi.org/10.1073/pnas.181141598
Usadel BR, Kuschinsky AM, Rosso MG, Eckermann N, Pauly M (2004) RHM2 is involved in mucilage pectin synthesis and is required for the development of the seed coat in Arabidopsis. Plant Physiol 134(1):286–295. https://doi.org/10.1104/pp.103.034314
Wang X, Hou S, Wu Q, Lin M, Acharya BR, Wu D, Zhang W (2017) IDL6-HAE/HSL2 impacts pectin degradation and resistance to Pseudomonas syringae pv tomato DC3000 in Arabidopsis leaves. Plant J 89(2):250–263. https://doi.org/10.1111/tpj.13380
Western TL, Young DS, Dean GH, Tan WL, Samuels AL, Haughn GW (2004) MUCILAGE-MODIFIED4 encodes a putative pectin biosynthetic enzyme developmentally regulated by APETALA2, TRANSPARENT TESTA GLABRA1, and GLABRA2 in the Arabidopsis seed coat. Plant Physiol 134(1):296–306. https://doi.org/10.1104/pp.103.035519
Wu X, Ning F, Hu X, Wang W (2017) Genetic modification for improving seed vigor is transitioning from model plants to crop plants. Front Plant Sci 8:8. https://doi.org/10.3389/fpls.2017.00008
Zhou W, Chen F, Luo X, Dai Y, Yang Y, Zheng C, Yang W, Shu K (2020) A matter of life and death: molecular, physiological, and environmental regulation of seed longevity. Plant Cell Environ 43(2):293–302. https://doi.org/10.1111/pce.13666
Zhu Q, Shao Y, Ge S, Zhang M, Zhang T, Hu X, Liu Y, Walker J, Zhang S, Xu J (2019) A MAPK cascade downstream of IDA-HAE/HSL2 ligand-receptor pair in lateral root emergence. Nat Plants 5(4):414–423. https://doi.org/10.1038/s41477-019-0396-x
Zinsmeister J, Lalanne D, Terrasson E, Chatelain E, Vandecasteele C, Vu BL, Dubois-Laurent C, Geoffriau E, Signor CL, Dalmais M, Gutbrod K, Dörmann P, Gallardo K, Bendahmane A, Buitink J, Leprince O (2016) ABI5 Is a regulator of seed maturation and longevity in legumes. Plant Cell 28(11):2735–2754. https://doi.org/10.1105/tpc.16.00470
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 32070349 and No. 31870304), and the creative group project of the Rice Industry Technological System of Tianjin (No. ITTRRS20211000-02).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, D., Guo, H., Chen, S. et al. Receptor-like kinase HAESA-like 1 positively regulates seed longevity in Arabidopsis. Planta 256, 21 (2022). https://doi.org/10.1007/s00425-022-03942-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-022-03942-y