Abstract
Main conclusion
Rhizosheath comprises soil that adheres firmly to roots. In this study, two ecotypes of switchgrass with different rhizosheath sizes after drought stress were analyzed which showed metabolic differences under drought conditions.
Abstract
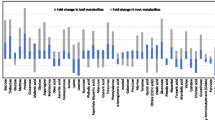
The rhizosheath comprises soil that adheres firmly to roots by a combination of root hairs and mucilage and may aid in root growth under soil drying. The aim of this work is to reveal the potential metabolites involved in rhizosheath formation under drought stress conditions. Panicum virgatum L. (switchgrass), which belongs to the Poaceae family, is an important biofuel and fodder crop in drought areas. Five switchgrass ecotypes (cv. Alamo, cv. Blackwake, cv. Summer, cv. Cave-in-Rock and cv. Kanlow) have a broad range of rhizosheath weight under drought conditions. For two selected ecotypes with contrast rhizosheath weight (cv. Alamo and cv. Kanlow), root hair length and density, lateral root number, root morphological parameters were measured, and real-time qRT-PCR was performed. Gas chromatography mass spectrophotometry (GC–MS) was used to determine the primary metabolites in the shoots and roots of selected ecotypes under drought stress conditions. The change trends of root hair length and density, lateral root number and related gene expression were consistent with rhizosheath weight in Alamo and Kanlow under drought and watered conditions. For root morphological parameters, Alamo grew deeper than Kanlow, while Kanlow exhibited higher values for other parameters. In this study, the levels of amino acids, sugars and organic acids were significantly changed in response to drought stress in two switchgrass ecotypes. Several metabolites including amino acids (arginine, isoleucine, methionine and cysteine) and sugars (kestose, raffinose, fructose, fucose, sorbose and xylose) in the large soil-sheathed roots of Alamo and Kanlow were significantly increased compared to small or no soil-sheathed roots of Alamo and Kanlow. Difference in rhizosheath size is reflected in the plant internal metabolites under drought stress conditions. Additionally, our results highlight the importance of using metabolite profiling and provide a better understanding of rhizosheath formation at the cellular level.







Similar content being viewed by others
References
Alqudah AM, Samarah NH, Mullen RE (2011) Alternative farming systems, biotechnology, drought stress and ecological fertilisation. Sustain Agric Rev 6:193–213. https://doi.org/10.1007/978-94-007-0186-1
Bailey C, Scholes M (1997) Rhizosheath occurrence in South African grasses. S Afr J Bot 63:484–490. https://doi.org/10.1016/S0254-6299(15)30803-6
Bao Y, Aggarwal P, Robbins NE, Sturrock CJ, Thompson MC, Tan HQ, Tham C, Duan LN, Rodriguez PL, Vernoux T, Mooney SJ, Bennett MJ, Dinneny JR (2014) Plant roots use a patterning mechanism to position lateral root branches toward available water. Proc Natl Acad Sci USA 111:9319–9324. https://doi.org/10.1073/pnas.1400966111
Barré P, Hallett PD (2009) Rheological stabilization of wet soils by model root and fungal exudates depends on clay mineralogy. Eur J Soil Sci 60:525–538. https://doi.org/10.1111/j.1365-2389.2009.01151.x
Baxter A, Mittler R, Suzuki N (2014) ROS as key players in plant stress signalling. J Exp Bot 65:1229–1240. https://doi.org/10.1093/jxb/ert375
Bengough AG, Bransby MF, Hans J, McKenna SJ, Roberts TJ, Valentine TA (2006) Root responses to soil physical conditions; growth dynamics from field to cell. J Exp Bot 57:437–447. https://doi.org/10.1093/jxb/erj003
Benjamin JG, Nielsen DC (2006) Water deficit effects on root distribution of soybean, field pea and chickpea. F Crop Res 97:248–253. https://doi.org/10.1016/j.fcr.2005.10.005
Bolouri-Moghaddam MR, Le Roy K, Xiang L, Rolland F, Van den Ende W (2010) Sugar signalling and antioxidant network connections in plant cells. FEBS J 277:2022–2037. https://doi.org/10.1111/j.1742-4658.2010.07633.x
Bowne JB, Erwin TA, Juttner J, Schnurbusch T, Langridge P, Bacic A, Roessner U (2012) Drought responses of leaf tissues from wheat cultivars of differing drought tolerance at the metabolite level. Mol Plant 5:418–429. https://doi.org/10.1093/mp/ssr114
Boyer JS (1982) Plant productivity and environment. Science 218:443–448. https://doi.org/10.1126/science.218.4571.443
Brown LK, George TS, Thompson JA, Wright G, Lyon J, Dupuy L, Hubbard SF, White PJ (2012) What are the implications of variation in root hair length on tolerance to phosphorus deficiency in combination with water stress in barley (Hordeum vulgare)? Ann Bot 110:319–328. https://doi.org/10.1093/aob/mcs085
Budak H, Hussain B, Khan Z, Ozturk NZ, Ullah N (2015) From genetics to functional genomics: improvement in drought signaling and tolerance in wheat. Front Plant Sci 6:1–13. https://doi.org/10.3389/fpls.2015.01012
Carminati A, Vetterlein D, Koebernick N, Blaser S, Weller U, Vogel HJ (2013) Do roots mind the gap? Plant Soil 367:651–661. https://doi.org/10.1007/s11104-012-1496-9
Casartelli A, Riewe D, Hubberten HM, Altmann T, Hoefgen R, Heuer S (2018) Exploring traditional aus-type rice for metabolites conferring drought tolerance. Rice 11:1–16. https://doi.org/10.1186/s12284-017-0189-7
Cichon MJ, Riedl KM, Wan L, Thomas-Ahner JM, Francis DM, Clinton SK, Schwartz SJ (2017) Plasma metabolomics reveals steroidal alkaloids as novel biomarkers of tomato intake in mice. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.201700241
Cosgrove DJ (1999) Enzymes and other agents that enhance cell wall extensibility. Annu Rev Plant Physiol Plant Mol Biol 50:391–417. https://doi.org/10.1146/annurev.arplant.50.1.391
Czechowski T, Stitt M, Altmann T, Udvardi MK (2005) Genome-wide identification and testing of superior reference genes for transcript normalization. Society 139:5–17. https://doi.org/10.1104/pp.105.063743.1
Darrah PR (1993) The rhizosphere and plant nutrition: a quantitative approach. Plant Soil 155:1–20. https://doi.org/10.1007/BF00024980
Delhaize E, James RA, Ryan PR (2012) Aluminium tolerance of root hairs underlies genotypic differences in rhizosheath size of wheat (Triticum aestivum) grown on acid soil. N Phytol 195:609–619. https://doi.org/10.1111/j.1469-8137.2012.04183.x
Delhaize E, Rathjen TM, Cavanagh CR (2015) The genetics of rhizosheath size in a multiparent mapping population of wheat. J Exp Bot 66:4527–4536. https://doi.org/10.1093/jxb/erv223
Du ZY, Chen MX, Chen QF, Xiao S, Chye ML (2013) Overexpression of Arabidopsis acyl-CoA-binding protein ACBP2 enhances drought tolerance. Plant Cell Environ 36:300–314. https://doi.org/10.1111/j.1365-3040.2012.02574.x
Fernie AR, Schauer N (2009) Metabolomics-assisted breeding: a viable option for crop improvement? Trends Genet 25:39–48. https://doi.org/10.1016/j.tig.2008.10.010
Fiehn O (2002) Metabolomics–the link between genotypes and phenotypes. Plant Mol Biol 48:155–171. https://doi.org/10.1023/A:1013713905833
George TS, Brown LK, Ramsay L, White PJ, Newton AC, Bengough AG, Russell J, Thomas WTB (2014) Understanding the genetic control and physiological traits associated with rhizosheath production by barley (Hordeum vulgare). N Phytol 203:195–205. https://doi.org/10.1111/nph.12786
Green H, Kehinde O (1976) Spontaneous heritable changes leading to increased adipose conversion in 3T3 cells. Cell 7:105–113. https://doi.org/10.1016/0092-8674(76)90260-9
Haling RE, Richardson AE, Culvenor RA, Lambers H, Simpson RJ (2010) Root morphology, root-hair development and rhizosheath formation on perennial grass seedlings is influenced by soil acidity. Plant Soil 335:457–468. https://doi.org/10.1007/s11104-010-0433-z
Haling RE, Brown LK, Bengough AG, Young IM, Hallett PD, White PJ, George TS (2013) Root hairs improve root penetration, root-soil contact, and phosphorus acquisition in soils of different strength. J Exp Bot 64:3711–3721. https://doi.org/10.1093/jxb/ert200
Haling RE, Brown LK, Bengough AG, Valentine TA, White PJ, Young IM, George TS (2014) Root hair length and rhizosheath mass depend on soil porosity, strength and water content in barley genotypes. Planta 239:643–651. https://doi.org/10.1007/s00425-013-2002-1
Harrigan GG, Stork LAG, Riordan SG, Ridley WP, Maclsaac S, Halle SC, Orth R, Rau D, Smith RG, Wen L, Brown WE, Riley R, Sun D, Modiano S, Pester T, Lund A, Nelson D (2007) Metabolite analyses of grain from maize hybrids grown in the United States under drought and watered conditions during the 2002 field season. J Agric Food Chem 55:6169–6176. https://doi.org/10.1021/jf070493s
Henry RJ, Darbyshire B (1980) Sucrose: sucrose fructosyltransferase and fructan: fructan fructosyltransferase from allium cepa. Phytochemistry 19:1017–1020
Hinsinger P, Gobran GR, Gregory PJ, Wenzel WW (2005) Rhizosphere geometry and heterogeneity arising from root-mediated physical and chemical processes. N Phytol 168:293–303. https://doi.org/10.1111/j.1469-8137.2005.01512.x
Jacobs A, Lunde C, Bacic A, Tester M, Roessner U (2007) The impact of constitutive heterologous expression of a moss Na+ transporter on the metabolomes of rice and barley. Metabolomics 3:307–317. https://doi.org/10.1007/s11306-007-0056-4
Kang Y, Khan S, Ma X (2009) Climate change impacts on crop yield, crop water productivity and food security—a review. Prog Nat Sci USA 19:1665–1674. https://doi.org/10.1016/j.pnsc.2009.08.001
Kopka J, Schauer N, Krueger S, Birkemeyer C, Usadel B, Bergmuller E, Dormann P, Weckwerth W, Gibon Y, Stitt M, Willmitzer L, Fernie AR, Steinhauser D (2005) GMD@CSB.DB: the Golm metabolome database. Bioinformatics 21:1635–1638. https://doi.org/10.1093/bioinformatics/bti236
Lanzinger A, Frank T, Reichenberger G, Herz M, Engel KH (2015) Metabolite profiling of barley grain subjected to induced drought stress: responses of free amino acids in differently adapted cultivars. J Agric Food Chem 63:4252–4261. https://doi.org/10.1021/acs.jafc.5b01114
Lesk C, Rowhani P, Ramankutty N (2016) Influence of extreme weather disasters on global crop production. Nature 529:84–87. https://doi.org/10.1038/nature16467
Lisec J, Schauer N, Kopka J, Willmitzer L, Fernie AR (2006) Gas chromatography mass spectrometry-based metabolite profiling in plants. Nat Protoc 1:387–396. https://doi.org/10.1038/nprot.2006.59
Liu TY, Ye NH, Song T, Cao YY, Gao B, Zhang D, Zhu FY, Chen MX, Zhang YJ, Xu WF, Zhang JH (2019) Rhizosheath formation and involvement in foxtail millet (Setaria italica) root growth under drought stress. J Integr Plant Biol 61(4):449–462. https://doi.org/10.1111/jipb.12716
Luedemann A, Strassburg K, Erban A, Kopka J (2008) TagFinder for the quantitative analysis of gas chromatography-mass spectrometry (GC-MS) based metabolite profiling experiments. Bioinformatics 24:732–737
Malamy JE (2005) Intrinsic and environmental response pathways that regulate root system architecture. Plant Cell Environ 28:67–77. https://doi.org/10.1111/j.1365-3040.2005.01306.x
McCully M (1995) How do real roots work? (Some new views of root structure). Plant Physiol 109:1–6. https://doi.org/10.1104/pp.109.1.1
Mibei EK, Owino WO, Ambuko J, Giovannoni JJ, Onyango AN (2018) Metabolomic analyses to evaluate the effect of drought stress on selected African eggplant accessions. J Sci Food Agric 98:205–216. https://doi.org/10.1002/jsfa.8458
Muller B, Stosser M, Tardieu F (1998) Spatial distributions of tissue expansion and cell division rates are related to irradiance and to sugar content in the growing zone of maize roots. Plant Cell Environ 21:149–158. https://doi.org/10.1046/j.1365-3040.1998.00263.x
Nam KH, Shin HJ, Pack IS, Park JH, Kim HB, Kim CG (2016) Metabolomic changes in grains of well-watered and drought-stressed transgenic rice. J Sci Food Agric 96:807–814. https://doi.org/10.1002/jsfa.7152
Othman AA, Amer WM, Fayez M, Hegazi NA (2004) Rhizosheath of sinai desert plants is a potential repository for associative diazotrophs. Microbiol Res 159:285–293. https://doi.org/10.1016/j.micres.2004.05.004
Pollock CJ, Housley TL (1993) The extraction and assay of 1-kestose: sucrose fructosyl transferase from leaves of wheat. Plant Physiol 102:537–539. https://doi.org/10.1104/pp.102.2.537
Praba ML, Cairns JE, Babu RC, Lafitte HR (2009) Identification of physiological traits underlying cultivar differences in drought tolerance in rice and wheat. J Agron Crop Sci 195:30–46. https://doi.org/10.1111/j.1439-037X.2008.00341.x
Price SR (1911) The roots of some North African desert-grasses. N Phytol 10:328–340
Rabe B (1990) Stress physiology: the function significance of the accumulation of nitrogen containing compounds. J Hort Sci 65:231–243
Roessner U (2001) Metabolic profiling allows comprehensive phenotyping of genetically or environmentally modified plant systems. Plant Cell 13:11–29. https://doi.org/10.1105/tpc.13.1.11
Shelden MC, Dias DA, Jayasinghe NS, Bacic A, Roessner U (2016) Root spatial metabolite profiling of two genotypes of barley (Hordeum vulgare L.) reveals differences in response to short-term salt stress. J Exp Bot 67:3731–3745. https://doi.org/10.1093/jxb/erw059
Shiomi N, Kido H, Kiriyama S (1985) Purification and properties of sucrose: sucrose 1F-β-D-fructosyltransferase in onion seeds. Phytochemistry 24:25–28
Steinfath M, Groth D, Lisec J, Selbig J (2008) Metabolite profile analysis: from raw data to regression and classification. Physiol Plant 132:150–161. https://doi.org/10.1111/j.1399-3054.2007.01006.x
Templer SE, Ammon A, Pscheidt D, Ciobotea O, Schuy C, McCollum C, Sonnewald U, Hanemann A, Forster J, Ordon F, von Korff M, Voll LM (2017) Metabolite profiling of barley flag leaves under drought and combined heat and drought stress reveals metabolic QTLs for metabolites associated with antioxidant defense. J Exp Bot 68:1697–1713. https://doi.org/10.1093/jxb/erx038
Ullah N, Yüce M, Neslihan Öztürk Gökçe Z, Budak H (2017) Comparative metabolite profiling of drought stress in roots and leaves of seven Triticeae species. BMC Genom 18:1–12. https://doi.org/10.1186/s12864-017-4321-2
Unno Y, Okubo K, Wasaki J, Shinano T, Osaki M (2005) Plant growth promotion abilities and microscale bacterial dynamics in the rhizosphere of Lupin analysed by phytate utilization ability. Environ Microbiol 7:396–404. https://doi.org/10.1111/j.1462-2920.2004.00701.x
Van’t Hof J (1968) The action of IAA and kinetin on the mitotic cycle of proliferative and stationary phase excised root meristems. Exp Cell Res 51:167–176. https://doi.org/10.1016/0014-4827(68)90167-5
Vermeer J, McCully ME (1982) The rhizosphere in Zea: new insight into its structure and development. Planta 156:45–61. https://doi.org/10.1007/BF00393442
Vinocur B, Altman A (2005) Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Curr Opin Biotechnol 16:123–132. https://doi.org/10.1016/j.copbio.2005.02.001
Wang Y, Gapstur SM, Carter BD, Hartman TJ, Stevens VL, Gaudet MM, McCullough ML (2018) Novel potential biomarkers of habitual food intake in a cross-sectional study of postmenopausal women. J Nutr 146:932–943. https://doi.org/10.1093/jn/nxy027
Watt M, Mccully ME, Canny MJ (1994) Formation and stabilization of rhizosheaths of Zea mays L. (effect of soil water content). Plant Physiol 106:179–186. https://doi.org/10.1104/pp.106.1.179
White PJ, George TS, Dupuy LX, Valentine T (2013a) Root traits for infertile soils. Front Plant Sci 4:1–7. https://doi.org/10.3389/fpls.2013.00193
White PJ, George TS, Gregory PJ, Bengough AG, Hallett PD, McKenzie BM (2013b) Matching roots to their environment. Ann Bot 112:207–222. https://doi.org/10.1093/aob/mct123
Wright L, Turhollow A (2010) Switchgrass selection as a “model” bioenergy crop: a history of the process. Biomass Bioenerg 34:851–868. https://doi.org/10.1016/j.biombioe.2010.01.030
Young IM (1995) Variation in moisture contents between bulk soil and the rhizosheath of wheat (Triticum aestivum L. cv. Wembley). N Phytol 130:135–139. https://doi.org/10.1111/j.1469-8137.1995.tb01823.x
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2017YFD0301502), the Natural Science Foundation of Hunan Province (2019JJ50263), the National Natural Science Foundation of China (NSFC 31771701), the Shenzhen Virtual University Park Support Scheme to CUHK Shenzhen Research Institute and the Hong Kong Research Grant Council (AoE/M-05/12, AoE/M-403/16, GRF 14160516, 14177617, 12100318).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, TY., Chen, MX., Zhang, Y. et al. Comparative metabolite profiling of two switchgrass ecotypes reveals differences in drought stress responses and rhizosheath weight. Planta 250, 1355–1369 (2019). https://doi.org/10.1007/s00425-019-03228-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-019-03228-w