Abstract
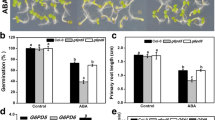
Arabidopsis nudix hydrolase 7 (Atnudt7) mutants exhibit reduced seed germination phenotype following after-ripening. The role of AtNUDT7 in seeds and during early stages of imbibition was examined. Seeds of Atnudt7-1 and Col-0 following 3 days of imbibition were used to profile changes in NADH- and ADP-ribose pyrophosphohydrolase enzyme activities, expression of nudix family genes closely related to AtNudt7, and AtNUDT7 protein levels. Changes in pyridine nucleotides, phytohormones, reactive oxygen species and poly(ADP-ribose) levels in after-ripened seeds and 1 day after imbibition were also analyzed. Changes in AtNUDT7 gene expression, protein levels and enzyme activities in WT seeds and during early stages of imbibition were correlated. Atnudt7-1 seeds lacked NADH pyrophosphohydrolase activity that led to very high catabolic redox charge. Abscisic acid (ABA) levels were higher in Atnudt7-1 mutant while salicylic acid, gibberellic acid, and reactive oxygen species (ROS) levels were higher in WT seeds. In Atnudt7-1, there was excess ROS accumulation 1 day after imbibition. PAR levels were significantly higher in Atnudt7-1 mutant when compared to WT during imbibition. Based on these observations, we conclude NADH pyrophosphohydrolase activity conferred by AtNUDT7 is important for NAD:NADH homeostasis in seeds. Perturbations to this key redox couple alter ABA and ROS levels in the seeds that in turn lowers germination.








Similar content being viewed by others
References
Aldasoro J, Nicolas G (1979) Change in the concentrations of glycolytic intermediates and adenosine phosphates during germination of seeds of Cicer arietinum L. Int J Biochem 10:947–950
Ames GR, King TA (1966) The assay and pH profile of lipoxidase. J Sci Food Agric 17:301–303
Bartsch M, Gobbato E, Bednarek P, Debey S, Schultze JL, Bautor J, Parker JE (2006) Salicylic acid-independent ENHANCED DISEASE SUSCEPTIBILITY1 signaling in Arabidopsis immunity and cell death is regulated by the monooxygenase FMO1 and the nudix hydrolase NUDT7. Plant Cell 18:1038–1051
Bazin J, Langlade N, Vincourt P, Arribat S, Balzergue S, El-Maarouf-Bouteau H, Bailly C (2011) Targeted mRNA oxidation regulates sunflower seed dormancy alleviation during dry after-ripening. Plant Cell 23:2196–2208
Beaudoin N, Serizet C, Gosti F, Giraudat J (2000) Interactions between abscisic acid and ethylene signaling cascades. Plant Cell 12:1103–1115
Bentsink L, Jowett J, Hanhart CJ, Koornneef M (2006) Cloning of DOG1, a quantitative trait locus controlling seed dormancy in Arabidopsis. Proc Natl Acad Sci 103:17042–17047
Bewley JD (1997) Seed germination and dormancy. Plant Cell 9:1055–1066
Botha FC, Potgeiter GT, Botha A-M (1992) Respiratory metabolism and gene expression during seed germination. J Plant Growth Regul 11:211–224
Carrera E, Holman T, Medhurst A, Dietrich D, Footitt S, Theodoulou FL, Holdsworth MJ (2008) Seed after-ripening is a discrete developmental pathway associated with specific gene networks in Arabidopsis. Plant J 53:214–224
Chen Q, Zhang B, Hicks LM, Wang S, Jez JM (2009) A liquid chromatography-tandem mass spectrometry-based assay for indole-3-acetic acid-amido synthetase. Anal Biochem 390:149–154
Chibani K, Ali-Rachedi S, Job C, Job D, Jullien M, Grappin P (2006) Proteomic analysis of seed dormancy in Arabidopsis. Plant Physiol 142:1493–1510
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Colville A, Alhattab R, Hu M, Labbe H, Xing T, Miki B (2011) Role of HD2 genes in seed germination and early seedling growth in Arabidopsis. Plant Cell Rep 10:1969–1979
Debeaujon I, Koornneef M (2000) Gibberellin requirement for Arabidopsis seed germination is determined both by testa characteristics and embryonic abscisic acid. Plant Physiol 122:415–424
Debeaujon I, Leon-Kloosterziel KM, Koornneef M (2000) Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis. Plant Physiol 122:403–414
Dunn CA, O’Handley SF, Frick DN, Bessman MJ (1999) Studies on the ADP-ribose pyrophosphatase subfamily of the nudix hydrolases and tentative identification of trgB, a gene associated with tellurite resistance. J Biol Chem 274:32318–32324
Ehrenshaft M, Brambl R (1990) Respiration and mitochondrial biogenesis in germinating embryos of maize. Plant Physiol 93:295–304
El-Maarouf-Boteau H, Bailly C (2008) Oxidative signaling in seed germination and dormancy. Plant Signal Behav 3:175–182
Finch-Savage WE, Leubner-Metzger G (2006) Seed dormancy and the control of germination. New Phytol 171:501–523
Gallais S, Crescenzo M-APD, Laval-Martin DL (1998) Pyridine nucleotides and redox charges during germination of non-dormant and dormant caryopses of Avena sativa L. J Plant Physiol 153:664–669
Ge X, Xia Y (2008) The role of AtNUDT7, a nudix hydrolase, in the plant defense response. Plant Signal Behav 3:119–120
Ge X, Li GJ, Wang SB, Zhu H, Zhu T, Wang X, Xia Y (2007) AtNUDT7, a negative regulator of basal immunity in Arabidopsis, modulates two distinct defense response pathways and is involved in maintaining redox homeostasis. Plant Physiol 145:204–215
Ghassemian M, Nambara E, Cutler S, Kawaide H, Kamiya Y, McCourt P (2000) Regulation of abscisic acid signaling by the ethylene response pathway in arabidopsis. Plant Cell 12:1117–1126
Gonzalez-Guzman M, Apostolova N, Belles JM, Barrero JM, Piqueras P, Ponce MR, Micol JL, Serrano R, Rodriguez PL (2002) The short-chain alcohol dehydrogenase ABA2 catalyzes the conversion of xanthoxin to abscisic aldehyde. Plant Cell 14:1833–1846
Hayashi M, Takahashi H, Tamura K, Huang JR, Yu LH, Kawai-Yamada M, Tezuka T, Uchimiya H (2005) Enhanced dihydroflavonol-4-reductase activity and NAD homeostasis leading to cell death tolerance in transgenic rice. Proc Natl Acad Sci 102:7020–7025
Henderson JT, Li HC, Rider SD, Mordhorst AP, Romero-Severson J, Cheng JC, Robey J, Sung ZR, de Vries SC, Ogas J (2004) PICKLE acts throughout the plant to repress expression of embryonic traits and may play a role in gibberellin-dependent responses. Plant Physiol 134:995–1005
Holdsworth MJ, Bentsink L, Soppe WJ (2008) Molecular networks regulating Arabidopsis seed maturation, after-ripening, dormancy and germination. New Phytol 179:33–54
Hunt L, Gray JE (2009) The relationship between pyridine nucleotides and seed dormancy. New Phytol 181:62–70
Hunt L, Lerner F, Ziegler M (2004) NAD—new roles in signalling and gene regulation in plants. New Phytol 163:31–44
Hunt L, Holdsworth MJ, Gray JE (2007) Nicotinamidase activity is important for germination. Plant J 51:341–351
Ishikawa K, Ogawa T, Hirosue E, Nakayama Y, Harada K, Fukusaki E, Yoshimura K, Shigeoka S (2009) Modulation of the poly(ADP-ribosyl)ation reaction via the Arabidopsis ADP-ribose/NADH pyrophosphohydrolase, AtNUDX7, is involved in the response to oxidative stress. Plant Physiol 151:741–754
Jambunathan N, Mahalingam R (2006) Analysis of Arabidopsis growth factor gene 1 (GFG1) encoding a nudix hydrolase during oxidative signaling. Planta 224:1–11
Jambunathan N, Penaganti A, Tang Y, Mahalingam R (2010) Modulation of redox homeostasis under suboptimal conditions by Arabidopsis nudix hydrolase 7. BMC Plant Biol 10:173
Joo JH, Wang SY, Chen JG, Jones AM, Fedoroff NV (2005) Different signaling and cell death roles of heterotrimeric G protein alpha and beta subunits in the arabidopsis oxidative stress response to ozone. Plant Cell 17:957–970
Kanno Y, Jikumaru Y, Hanada A, Nambara E, Abrams SR, Kamiya Y, Seo M (2010) Comprehensive hormone profiling in developing Arabidopsis seeds: examination of the site of ABA biosynthesis, ABA transport and hormone interactions. Plant Cell Physiol 51:1988–2001
Kennedy RA, Rumpho ME, Fox TC (1992) Anaerobic metabolism in plants. Plant Physiol 100:1–6
Koornneef M, Bentsink L, Hilhorst H (2002) Seed dormancy and germination. Curr Opin Plant Biol 5:33–36
Kucera B, Cohn MA, Leubner-Metzger G (2005) Plant hormone interactions during seed dormancy release and germination. Seed Sci Res 15:281–307
Kwak JM, Mori IC, Pei ZM, Leonhardt N, Torres MA, Dangl JL, Bloom RE, Bodde S, Jones JDG, Schroeder JI (2003) NADPH oxidase AtrbohD and AtrbohF genes function in ROS-dependent ABA signaling in Arabidopsis. EMBO J 22:2623–2633
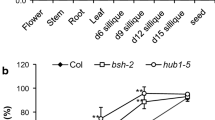
Liu Y, Koornneef M, Soppe WJ (2007) The absence of histone H2B monoubiquitination in the Arabidopsis hub1 (rdo4) mutant reveals a role for chromatin remodeling in seed dormancy. Plant Cell 19:433–444
Mahalingam R, Jambunathan N, Penaganti A (2007) Pyridine nucleotide homeostasis in plant development and stress. Int J Plant Dev Biol 1:194–201
Muller K, Carstens AC, Linkies A, Torres MA, Leubner-Metzger G (2009) The NADPH-oxidase AtrbohB plays a role in Arabidopsis seed after-ripening. New Phytol 184:885–897
Murata Y, Pei ZM, Mori IC, Schroeder J (2001) Abscisic acid activation of plasma membrane Ca2+ channels in guard cells requires cytosolic NAD(P)H and is differentially disrupted upstream and downstream of reactive oxygen species production in abi1-1 and abi2-1 protein phosphatase 2C mutants. Plant Cell 13:2513–2523
Nakabayashi K, Bartsch M, Xiang Y, Miatton E, Pellengahr S, Yano R, Seo M, Soppe WJ (2012) The time required for dormancy release in Arabidopsis is determined by DELAY OF GERMINATION1 protein levels in freshly harvested seeds. Plant Cell 24:2826–2838
Naredo MEB, Juliano AB, Lu BR, De Guzman F, Jackson MT (1998) Responses to seed dormancy-breaking treatments in rice species (Oryza L.). Seed Sci Tech 26:675–689
Nonogaki H, Bassel GW, Bewley JD (2010) Germination—still a mystery. Plant Sci 179:574–581
Ogawa M, Hanada A, Yamauchi Y, Kuwahara A, Kamiya Y, Yamaguchi S (2003) Gibberellin biosynthesis and response during Arabidopsis seed germination. Plant Cell 15:1591–1604
Ogawa T, Ueda Y, Yoshimura K, Shigeoka S (2005) Comprehensive analysis of cytosolic nudix hydrolases in Arabidopsis thaliana. J Biol Chem 280:25277–25283
Olejnik K, Kraszewska E (2005) Cloning and characterization of an Arabidopsis thaliana nudix hydrolase homologous to the mammalian GFG protein. Biochim Biophys Acta 1752:133–141
Oracz K, El-Maarouf Bouteau H, Farrant JM, Cooper K, Belghazi M, Job C, Job D, Corbineau F, Bailly C (2007) ROS production and protein oxidation as a novel mechanism for seed dormancy alleviation. Plant J 50:452–465
Overmyer K, Brosche M, Kangasjarvi J (2003) Reactive oxygen species and hormonal control of cell death. Trends Plant Sci 8:335–342
Penfield S, King J (2009) Towards a systems biology approach to understanding seed dormancy and germination. Proc Biol Sci 276:3561–3569
Powell AD, Dulson J, Bewley JD (1984) Changes in germination and respiratory potential of embryos of dormant grand rapids lettuce seeds during long-term imbibed storage, ad related changes in the endosperm. Planta 162:40–45
Rajjou L, Gallardo K, Debeaujon I, Vandekerckhove J, Job C, Job D (2004) The effect of alpha-amanitin on the Arabidopsis seed proteome highlights the distinct roles of stored and neosynthesized mRNAs during germination. Plant Physiol 134:1598–1613
Ribeiro JM, Carloto A, Costas MJ, Cameselle JC (2001) Human placenta hydrolases active on free ADP-ribose: an ADP-sugar pyrophosphatase and a specific ADP-ribose pyrophosphatase. Biochim Biophys Acta 1526:86–94
Russell L, Larner V, Kurup S, Bougourd S, Holdsworth M (2000) The Arabidopsis COMATOSE locus regulates germination potential. Development 127:3759–3767
Salon C, Raymond P, Pradet A (1988) Quantification of carbon fluxes through the tricarboxylic acid cycle in early germinating lettuce embryos. J Biol Chem 263:12278–12287
Sarath G, Hou G, Baird LM, Mitchell RB (2007) Reactive oxygen species, ABA and nitric oxide interactions on the germination of warm-season C4-grasses. Planta 226:697–708
Steber CM, Cooney SE, McCourt P (1998) Isolation of the GA-response mutant sly1 as a suppressor of ABI1-1 in Arabidopsis thaliana. Genetics 149:509–521
Torres MA, Onouchi H, Hamada S, Machida C, Hammond-Kosack KE, Jones JDG (1998) Six Arabidopsis thaliana homologues of the human respiratory burst oxidase (gp91(phox)). Plant J 14:365–370
Wang M, Heimovaara-Dijkstra S, Van Duijn B (1995) Modulation of germination of embryos isolated from dormant and nondormant barley grains by manipulation of endogenous abscisic levels. Planta 195:586–592
Winter D, Vinegar B, Nahal H, Ammar R, Wilson GV, Provart NJ (2007) An “electronic fluorescent pictograph” browser for exploring and analyzing large-scale biological data sets. PLoS One 2:e718
Zhang Y, Zhang B, Yan D, Dong W, Yang W, Li Q, Zeng L, Wang J, Wang L, Hicks LM, He Z (2011) Two Arabidopsis cytochrome P450 monooxygenases, CYP714A1 and CYP714A2, function redundantly in plant development through gibberellin deactivation. Plant J 67:342–353
Acknowledgments
This research was supported partially by National Science Foundation under Grant No. EPS-081431. We thank Dr. Jane Parker (Max Planck Institute for Plant Breeding Research) for providing us the Atnudt7-2 mutant seeds. We thank Dr. Leslie Hicks, Donald Danforth Plant Sciences Center, Saint Louis, MO for assistance with the phytohormone analysis. We thank Dr. Yixing Wang for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM_1 Expression of AtNUDT7 protein in after-ripened seeds of Atnudt7-1 complementation lines. A. Ten micrograms of protein extract from seeds of Col-0, and the two complementation lines (Atnudt7-1t23, Atnudt7-1t37) were used to prepare the blot, followed by hybridization with AtNUDT7 polyclonal antibodies. B. Coomassie blue staining of membrane showing protein loading.
ESM_2 Coomassie blue stained membranes showing protein loading. The same membranes were used for western hybridizations with AtNUDT7 polyclonal antibodies shown in Fig. 4.
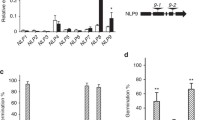
ESM_3 eFP browser analysis of Atnudt7 gene expression in various tissues and developmental stages.
ESM_4 Primer sequences used for RT-PCR analysis.
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zeng, X., Li, YF. & Mahalingam, R. Arabidopsis nudix hydrolase 7 plays a role in seed germination. Planta 239, 1015–1025 (2014). https://doi.org/10.1007/s00425-014-2035-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-014-2035-0