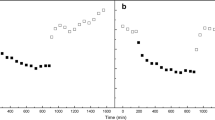
Abstract
Physcomitrella patens is a model bryophyte representing an early land plant in the green plant lineage. This organism possesses many advantages as a model organism. Its genome has been sequenced, its predominant life cycle stage is the haploid gametophyte, it is readily transformable and it can integrate transformed DNA into its genome by homologous recombination. One limitation for the use of P. patens in photosynthesis research is its reported inability to grow photoheterotrophically, in the presence of sucrose and the Photosystem II inhibitor 3-(3,4-dichlorophenyl)-1,1-dimethylurea, which prevents linear photosynthetic electron transport. In this communication we describe the facile isolation of a P. patens strain which can grow photoheterotrophically. Additionally, we have examined a number of photosynthetic parameters for this strain grown under photoautotrophic, mixotrophic (in the presence of sucrose) and photoheterotrophic conditions, as well as the 3-(3,4-dichlorophenyl)-1,1-dimethylurea-inhibited state. The ability to grow P. patens photoheterotrophically should significantly facilitate its use in photosynthetic studies.






Similar content being viewed by others
References
Anderson SL, McIntosh L (1991) Light-activated heterotrophic growth of the cyanobacterium Synechocystis sp. strain PCC 6803: a blue-light-requiring process. J Bacteriol 173:2761–2767
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15
Bouyoub A, Vernotte C, Astier C (1993) Functional analysis of the two homologous psbA gene copies in Synechocystis PCC 6714 and PCC 6803. Plant Mol Biol 21:249–258
Bricker TM, Putnam-Evans C, Wu J (1998) Mutagenesis in the study of the structure and function of Photosystem II. Meth Enzymol 297:320–337
Burnap RL, Shen J-R, Jursinic PA, Inoue Y, Sherman LA (1992) Oxygen yield and thermoluminescence characteristics of a cyanobacterium lacking the manganese-stabilizing protein of Photosystem II. Biochemistry 31:7404–7410
Cove DJ (2005) The moss Physcomitrella patens. Annu Rev Genet 39:339–358
Cove DJ, Schild A, Ashton NW, Hartmann E (1978) Genetic and physiological studies of the effect of light on the development of the moss, Physcomitrella patens. Photochem Photobiol 27:249–254
Cove DJ, Bezanilla M, Harries P, Quatrano RS (2006) Mosses as model systems for the study of metabolism and development. Annu Rev Plant Biol 57:497–520
Cove DJ, Perroud P-F, Charron AJ, McDanie SFl, Khandelwal A, Quatrano RS (2009) Culturing the Moss Physcomitrella patens. Cold Spring Harb Protoc. doi:10.1101/pdb.prot5136
Dal Bosco C, Lezhneva L, Biehl A, Leister D, Strotmann H, Wanner G, Meurer J (2004) Inactivation of the chloroplast ATP synthase gamma subunit results in high non-photochemical fluorescence quenching and altered nuclear gene expression in Arabidopsis thaliana. J Biol Chem 279:1060–1069
Dalla-Chiesa M, Friso G, Deak Z, Vass I, Barber J, Nixon PJ (1997) Reduced turnover of the D1 polypeptide and photoactivation of electron transfer in novel herbicide resistant mutants of Synechocystis sp. PCC 6803. Eur J Biochem 248(3):731–740
de Vitry C, Olive J, Drapier D, Recouvreur M, Wollman FA (1989) Post-translational events leading to the assembly of Photosystem II protein complex a study using photosynthesis mutants from Chlamydomonas reinhardtii. J Cell Biol 109:991–1006
Delepelaire P, Chua N (1979) Lithium dodecyl sulfate/polyacrylamide gel electrophoresis of thylakoid membranes at 4 °C: characterizations of two additional chlorophyll a-protein complexes. Proc Natl Acad Sci (USA) 76:111–115
Drapier D, Girard-Bascou J, Wollman F-A (1992) Evidence for nuclear control of the expression of the atpA and atpB chloroplast genes in Chlamydomonas. Plant Cell 4:283–295
Erickson JM, Rahire M, Bennoun P, Delepelaire P, Diner B, Rochaix JD (1984) Herbicide resistance in Chlamydomonas reinhardtii results from a mutation in the chloroplast gene for the 32 kilodalton protein of Photosystem II. Proc Natl Acad Sci USA 81(12):3617–3621
Frank W, Dekker EL, Reski R (2005) Molecular tools to study Physcomitrella patens. Plant Biol 7:220–227
Galloway RE, Mets L (1982) Non-mendelian inheritance of 3-(3,4-dichlorophenyl)-1,1-dimethylurea resistant thylakoid membrane properties in Chlamydomonas reinhardii. Plant Physiol 70:1673–1677
Gatenby AA, Rothstein SJ, Nomura M (1989) Translational coupling of the maize chloroplast atpB and atpE genes. Proc Natl Acad Sci (USA) 86:4066–4070
Hueber FM (1961) Hepaticites devonicus : a new fossil liverwort from the Devonian of New York. Ann Miss Bot Gard 48:125–132
Joliot P, Barbieri G, Chabaud R (1969) Un nouveau modele des centres photochimique du systeme II. Photochem Photobiol 10:309–329
Jursinic PA (1979) Flash-yield pattern for photosynthetic oxygen evolution in Chlorella and chloroplasts as a function of excitation intensity. Arch Biochem Biophys 196:484–492
Kok B, Forbush B, McGloin M (1970) Cooperation of charges in photosynthetic oxygen evolution: I. A linear four step mechanism. Photochem Photobiol 11:457–475
Pakrasi HB, Diner BA, Williams JGK, Arntzen CJ (1989) Deletion mutagenesis of the cytochrome b 559 protein inactivates the reaction center of Photosystem II. Plant Cell 1(6):591–598
Reinders A, Sivitz AB, Ward JM (2012) Evolution of plant sucrose uptake transporters. Front Plant Sci 3:22–31
Rensing SA, Lang D, Zimmer AD, Terry A, Salamov A, Shapiro H, Nishiyama T, Perroud P-F, Lindquist EA, Kamisugi Y, Tanahashi T, Sakakibara K, Fujita T, Oishi K, Shin-I T, Kuroki Y, Toyoda A, Suzuki Y, S-i Hashimoto, Yamaguchi K, Sugano S, Kohara Y, Fujiyama A, Anterola A, Aoki S, Ashton N, Barbazuk WB, Barker E, Bennetzen JL, Blankenship R, Cho SH, Dutcher SK, Estelle M, Fawcett JA, Gundlach H, Hanada K, Heyl A, Hicks KA, Hughes J, Lohr M, Mayer K, Melkozernov A, Murata T, Nelson DR, Pils B, Prigge M, Reiss B, Renner T, Rombauts S, Rushton PJ, Sanderfoot A, Schween G, Shiu S-H, Stueber K, Theodoulou FL, Tu H, Van de Peer Y, Verrier PJ, Waters E, Wood A, Yang L, Cove D, Cuming AC, Hasebe M, Lucas S, Mishler BD, Reski R, Grigoriev IV, Quatrano RS, Boore JL (2008) The Physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 319:64–69
Rochaix J-D (1987) Molecular genetics of chloroplasts and mitochondria in the unicellular green alga Chlamydomonas. FEMS Microbiol Rev 46:13–34
Schaefer DG, Zrÿd J-P (1997) Efficient gene targeting in the moss Physcomitrella patens. Plant J 11:1195–1206
Strasser RJ, Srivastava A, Tsimilli-Michael M (2000) The fluorescence transient as a tool to characterize and screen photosynthetic samples. In: Yunus M, Pathre U, Mohanty P (eds) Probing photosynthesis: mechanism, regulation and adaptation. Taylor and Francis, London, pp 443–480
Thornton LE, Keren N, Ohad I, Pakrasi HB (2005) Physcomitrella patens and Ceratodon purpureus, mosses as model organisms in photosynthesis studies. Photosynth Res 83:87–96
Vermaas WFJ, Williams JGK, Arntzen CJ (1987) Sequencing and modification of psbB, the gene encoding the CP 47 protein of Photosystem II in the cyanobacterium Synechocystis 6803. Plant Mol Biol 8:317–326
Williams JGK (1988) Construction of specific mutations in Photosystem II photosynthetic reaction center by genetic engineering methods in Synechocystis 6803. Methods Enzymol 167:766–778
Yi X, Hargett SR, Frankel LK, Bricker TM (2009) The PsbP protein, but not the PsbQ protein, is required for normal thylakoid membrane architecture in Arabidopsis thaliana. FEBS Lett 583:2142–2147
Acknowledgments
This work was supported by the Division of Chemical Sciences, Geosciences, and Biosciences, Office of Basic Energy Sciences of the U.S. Department of Energy through Grant DE-FG02-98ER20310 to T.M.B and L.K.F and Grant DE-FG02-03ER1540 to S.M.T.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bricker, T.M., Bell, A.J., Tran, L. et al. Photoheterotrophic growth of Physcomitrella patens . Planta 239, 605–613 (2014). https://doi.org/10.1007/s00425-013-2000-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-013-2000-3